

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (7): 1276.doi: 10.7503/cjcu20150980
• Analytical Chemistry • Previous Articles Next Articles
ZOU Haimin1,2, ZHOU Chen1, SUN Chengjun1, LI Yongxin1, YANG Xiaosong2, WEN Jun2, ZENG Hongyan1,*( )
)
Received:2015-12-23
Online:2016-07-10
Published:2016-06-02
Contact:
ZENG Hongyan
E-mail:zhm504532@163.com
CLC Number:
TrendMD:
ZOU Haimin, ZHOU Chen, SUN Chengjun, LI Yongxin, YANG Xiaosong, WEN Jun, ZENG Hongyan. Simultaneous Determination of 7 Components in Functional Food for Anti-hangover and Hepatoprotection by Capillary Electrophoresis†[J]. Chem. J. Chinese Universities, 2016, 37(7): 1276.
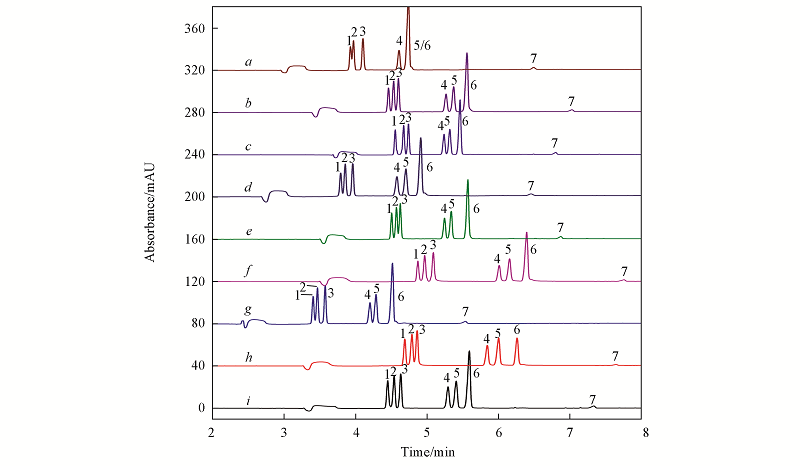
Fig.1 Electrophoretograms of 7 components in mixed standard solution with orthogonal experiment of L9(34) a—i represent to different orthogonal combination corresponding to Table S1 in supplemental file. 1. EGC; 2. C; 3. EC;
| Compound | Regression equation | Correlation coefficient | Linear range/(μg·mL-1) | LOD/(μg·g-1) | LOQ/(μg·g-1) |
|---|---|---|---|---|---|
| EGC | y=3049.1x-13.5 | 0.9995 | 0.09—100 | 0.55 | 1.83 |
| C | y=4294.2x+17.9 | 0.9994 | 0.06—100 | 0.34 | 1.13 |
| EC | y=4892.8x+27.4 | 0.9991 | 0.05—100 | 0.26 | 0.87 |
| EGCG | y=3343.5x-33.1 | 0.9993 | 0.12—100 | 0.79 | 2.64 |
| ECG | y=5521.2x+22.7 | 0.9989 | 0.08—100 | 0.63 | 2.09 |
| DMY | y=3940.3x-15.5 | 0.9993 | 0.16—100 | 1.00 | 3.32 |
| GA | y=897.2x+11.9 | 0.9992 | 0.35—200 | 2.22 | 7.39 |
Table 1 Regression equation, linear range, LOD and LOQ of 7 components
| Compound | Regression equation | Correlation coefficient | Linear range/(μg·mL-1) | LOD/(μg·g-1) | LOQ/(μg·g-1) |
|---|---|---|---|---|---|
| EGC | y=3049.1x-13.5 | 0.9995 | 0.09—100 | 0.55 | 1.83 |
| C | y=4294.2x+17.9 | 0.9994 | 0.06—100 | 0.34 | 1.13 |
| EC | y=4892.8x+27.4 | 0.9991 | 0.05—100 | 0.26 | 0.87 |
| EGCG | y=3343.5x-33.1 | 0.9993 | 0.12—100 | 0.79 | 2.64 |
| ECG | y=5521.2x+22.7 | 0.9989 | 0.08—100 | 0.63 | 2.09 |
| DMY | y=3940.3x-15.5 | 0.9993 | 0.16—100 | 1.00 | 3.32 |
| GA | y=897.2x+11.9 | 0.9992 | 0.35—200 | 2.22 | 7.39 |
| Compound | Intra-day precision(RSD, %) | Inter-day precision(RSD, %) | ||
|---|---|---|---|---|
| Retention time | Area | Retention time | Area | |
| EGC | 0.41 | 1.3 | 0.92 | 1.9 |
| C | 0.32 | 1.5 | 1.0 | 2.3 |
| EC | 0.54 | 1.9 | 1.3 | 2.8 |
| EGCG | 0.52 | 2.5 | 1.5 | 3.7 |
| ECG | 0.61 | 2.1 | 1.7 | 3.9 |
| DMY | 0.68 | 2.3 | 1.2 | 3.2 |
| GA | 0.77 | 1.7 | 1.4 | 2.6 |
Table 2 Intra-day precision and inter-day precision(n=6)
| Compound | Intra-day precision(RSD, %) | Inter-day precision(RSD, %) | ||
|---|---|---|---|---|
| Retention time | Area | Retention time | Area | |
| EGC | 0.41 | 1.3 | 0.92 | 1.9 |
| C | 0.32 | 1.5 | 1.0 | 2.3 |
| EC | 0.54 | 1.9 | 1.3 | 2.8 |
| EGCG | 0.52 | 2.5 | 1.5 | 3.7 |
| ECG | 0.61 | 2.1 | 1.7 | 3.9 |
| DMY | 0.68 | 2.3 | 1.2 | 3.2 |
| GA | 0.77 | 1.7 | 1.4 | 2.6 |
| Compound | Background/(μg·mL-1) | Added/(μg·mL-1) | Found/(μg·mL-1) | Recovery(%) | RSD(%) |
|---|---|---|---|---|---|
| EGC | 8.27 | 5.00 | 13.0 | 93.9 | 2.7 |
| 10.00 | 17.9 | 95.7 | 2.3 | ||
| 20.00 | 27.4 | 95.4 | 2.9 | ||
| C | ND | 5.00 | 4.66 | 93.1 | 3.2 |
| 10.00 | 9.48 | 94.8 | 2.7 | ||
| 20.00 | 19.3 | 96.5 | 2.6 | ||
| EC | 5.88 | 5.00 | 10.4 | 94.1 | 2.5 |
| 10.00 | 15.4 | 97.8 | 2.7 | ||
| 20.00 | 26.1 | 102.1 | 1.8 | ||
| EGCG | 27.23 | 10.00 | 37.0 | 96.4 | 2.2 |
| 20.00 | 46.6 | 96.2 | 1.4 | ||
| 40.00 | 69.4 | 104.9 | 1.7 | ||
| ECG | 10.30 | 5.00 | 14.8 | 93.4 | 3.1 |
| 10.00 | 19.2 | 91.4 | 2.8 | ||
| 20.00 | 29.6 | 97.5 | 3.0 | ||
| DMY | ND | 5.00 | 4.60 | 92.1 | 3.2 |
| 10.00 | 9.35 | 93.5 | 2.6 | ||
| 20.00 | 18.3 | 91.6 | 2.2 | ||
| GA | ND | 20.00 | 20.7 | 103.3 | 2.3 |
| 40.00 | 39.2 | 98.1 | 1.8 | ||
| 80.00 | 81.8 | 102.2 | 1.7 |
Table 3 Results of the spiked recovery tests*(n=6)
| Compound | Background/(μg·mL-1) | Added/(μg·mL-1) | Found/(μg·mL-1) | Recovery(%) | RSD(%) |
|---|---|---|---|---|---|
| EGC | 8.27 | 5.00 | 13.0 | 93.9 | 2.7 |
| 10.00 | 17.9 | 95.7 | 2.3 | ||
| 20.00 | 27.4 | 95.4 | 2.9 | ||
| C | ND | 5.00 | 4.66 | 93.1 | 3.2 |
| 10.00 | 9.48 | 94.8 | 2.7 | ||
| 20.00 | 19.3 | 96.5 | 2.6 | ||
| EC | 5.88 | 5.00 | 10.4 | 94.1 | 2.5 |
| 10.00 | 15.4 | 97.8 | 2.7 | ||
| 20.00 | 26.1 | 102.1 | 1.8 | ||
| EGCG | 27.23 | 10.00 | 37.0 | 96.4 | 2.2 |
| 20.00 | 46.6 | 96.2 | 1.4 | ||
| 40.00 | 69.4 | 104.9 | 1.7 | ||
| ECG | 10.30 | 5.00 | 14.8 | 93.4 | 3.1 |
| 10.00 | 19.2 | 91.4 | 2.8 | ||
| 20.00 | 29.6 | 97.5 | 3.0 | ||
| DMY | ND | 5.00 | 4.60 | 92.1 | 3.2 |
| 10.00 | 9.35 | 93.5 | 2.6 | ||
| 20.00 | 18.3 | 91.6 | 2.2 | ||
| GA | ND | 20.00 | 20.7 | 103.3 | 2.3 |
| 40.00 | 39.2 | 98.1 | 1.8 | ||
| 80.00 | 81.8 | 102.2 | 1.7 |
| Sample | Formulation | EGC | C | EC | EGCG | ECG | DMY | GA |
|---|---|---|---|---|---|---|---|---|
| 1 | Tea bag | ND | ND | ND | 0.338 | 0.241 | 2.230 | ND |
| 2 | Tea bag | 0.213 | ND | 0.148 | 0.684 | 0.259 | ND | ND |
| 3 | Tea | ND | ND | ND | 0.981 | 0.231 | ND | ND |
| 4 | Tea | 0.054 | 0.389 | 0.964 | 1.590 | 0.455 | ND | 0.117 |
| 5 | Liquid | 0.433 | 0.120 | 0.315 | 0.092 | 0.746 | ND | ND |
| 6 | Powder | ND | 0.925 | 1.030 | 0.607 | 0.711 | 0.292 | 1.090 |
| 7 | Liquid | ND | ND | 0.045 | 0.524 | 0.104 | ND | ND |
| 8 | Tablet | ND | 0.053 | ND | 0.256 | ND | ND | 0.496 |
Table 4 Contents of the target components in 8 samples*(mg/g, n=3)
| Sample | Formulation | EGC | C | EC | EGCG | ECG | DMY | GA |
|---|---|---|---|---|---|---|---|---|
| 1 | Tea bag | ND | ND | ND | 0.338 | 0.241 | 2.230 | ND |
| 2 | Tea bag | 0.213 | ND | 0.148 | 0.684 | 0.259 | ND | ND |
| 3 | Tea | ND | ND | ND | 0.981 | 0.231 | ND | ND |
| 4 | Tea | 0.054 | 0.389 | 0.964 | 1.590 | 0.455 | ND | 0.117 |
| 5 | Liquid | 0.433 | 0.120 | 0.315 | 0.092 | 0.746 | ND | ND |
| 6 | Powder | ND | 0.925 | 1.030 | 0.607 | 0.711 | 0.292 | 1.090 |
| 7 | Liquid | ND | ND | 0.045 | 0.524 | 0.104 | ND | ND |
| 8 | Tablet | ND | 0.053 | ND | 0.256 | ND | ND | 0.496 |
| [1] | Svoboda P., VlćckováH. , NovákováL. , J. Pharm. Biomed. Anal. , 2015, 114, 62—70 |
| [2] | Ye L. Y., Wang H. J., Duncan S. E., Eigel W. N., O’Keefe S. F., Food Chem., 2015, 172, 416—422 |
| [3] | Charpe T. W., Rathod V. K., Food Bioprod. Process,2015, 93, 51—57 |
| [4] | Shabkhiz M. A., Eikani M. H., Bashiri Sadr Z., Golmohammad F., Food Chem., 2016, 210, 396—401 |
| [5] | Liu G. Q., Dong J., Wang H., Wan L. R., Duan Y.S., Chen S. Z., Chem. J. Chinese Universities,2009, 30(8), 1566—1570(刘国强, 董静, 王弘, 万乐人, 端裕树, 陈世忠. 高等学校化学学报, 2009, 30(8), 1566—1570) |
| [6] | Anissi J., El Hassouni M., Ouardaoui A., Sendide K., Food Chem., 2014, 150, 438—447 |
| [7] | Oh J., Jo H., Cho A. R., Kim S. J., Hanet J., Food Control,2013, 31(2), 403—409 |
| [8] | Ou X. H., Ye Y., Huang Q. J., Liu H. G., Song Y. F., Nat. Prod. Res. Dev., 2013, 25(2), 245—248(欧贤红, 叶勇, 黄秋洁, 刘华钢, 宋云飞. 天然产物研究与开发, 2013, 25(2), 245—248) |
| [9] | Fu Y., Chen J., Li Y. J., Zheng Y. F., Li P., Food Chem., 2013, 141(2), 1063—1071 |
| [10] | Wang X.Y., Zhang H, Chen L. L., Shan L. H., Fan G. W., Gao X. M., J. Ethnopharmacol., 2013, 150(3), 781—790 |
| [11] | Boudesocque L., Dorat J., Pothier J., Gueiffer A., Gueiffier C. E., Food Chem., 2013, 139, 866—871 |
| [12] | Soleas G. J., Yan J., Goldberg D. M., J. Chromatogr. B,2001, 757(1), 161—172 |
| [13] | Liao W. C., Lin Y. H., Chang T. M., Huang W. Y., Food Chem., 2012, 132(4), 2188—2193 |
| [14] | Rahim A. A., Nofrizal S., Saad B., Food Chem., 2014, 147, 262—268 |
| [15] | Peres R. G., Tonin F. G., Tavares M. F. M., Amaya D. B. R., Food Chem., 2011, 127(2), 651—655 |
| [16] | Zhang S., Dong S. Q., Chi L. Z., He P. G., Wang Q. J., Fang Y. Z., Talanta,2008, 76(4), 780—784 |
| [17] | Vinatoru M., Ultrason. Sonochem., 2001, 8(3), 303—313 |
| [18] | Morelli L. L. L., Prado M. A., Ultrason. Sonochem., 2012, 19(6), 1144—1149 |
| [19] | Lun S. Y., Leung L. K., Huang Y., Chen Z. Y., Food Chem., 2003, 83(2), 189—195 |
| [20] | Ananingsih V. K., Sharma A., Zhou W. B., Food Res. Int., 2013, 50(2), 469—479 |
| [21] | Xu J. J., Yao M. J., Wu M. C., Food Sci., 2008, 29(11), 622—625(徐静娟, 姚茂君, 邬敏辰. 食品科学, 2008, 29(11), 622—625) |
| [1] | HUANG Chibao, KANG Shuai, PAN Qi, LYU Guoling. Carbazole-derived Dicyanostilbene Two-photon Fluorescence Probe for Lipid Raft [J]. Chem. J. Chinese Universities, 2021, 42(8): 2443. |
| [2] | MA Zhuoyuan, WANG Dayang. Status and Prospect of Surface Wettability of Molecular Self-assembled Monolayers [J]. Chem. J. Chinese Universities, 2021, 42(4): 1031. |
| [3] | KUANG Xiaojun, YI Jingwei, FANG Xiaoxia, LAI Dongmei, XU Hong. Preparation of Water-soluble Coumarin Fluorescent Substrate and Its Application in Droplet Based Digital Detection [J]. Chem. J. Chinese Universities, 2021, 42(11): 3537. |
| [4] | WANG Baichun, YUAN Yuxin, YAN Yinghua, DING Chuanfan, TANG Keqi. Glucose-6-phosphate Functionalized Hydrophilic Magnetic Probe: a Dual-purpose Affinity Material for Effective Separation and Enrichment of Glycopeptides/Phosphopeptides [J]. Chem. J. Chinese Universities, 2021, 42(10): 3062. |
| [5] | SHEN Yang, ZHU Fang, SHEN Wanwan, FAN Qianqian, LI Yiwen, CHENG Yiyun. Structure-function Relationship of Plant Polyphenols for Promoted siRNA Delivery † [J]. Chem. J. Chinese Universities, 2020, 41(4): 633. |
| [6] | AI Wanpeng, SONG Shiyao, BAI Yu, LIU Huwei. Development and Applications of Mass Spectrometry in Reaction Monitoring [J]. Chem. J. Chinese Universities, 2020, 41(12): 2598. |
| [7] | CHENG Qinwen, YUAN Bo, ZHU Xiangdong, ZHANG Kai, ZHANG Xingdong. Surface Elemental Compositions and in Vitro Bioactivities of the PEEK Materials with Various Sulfonation and Alkali Treatments [J]. Chem. J. Chinese Universities, 2019, 40(8): 1757. |
| [8] | ZHANG Huan, DONG Xiaoyan. Effects of EGCG on Amyloid β-Protein Fibrillogenesis and Cytotoxicity at Different pH Values† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2534. |
| [9] | HU Jingwei, CHENG Rui, DING Shunmin, XU Mengmeng, XU Lu, XIAO Weiming, ZHOU Penghui, ZHANG Ning. Diphenylphosphine Functionalized Graphene Oxide Supported Palladium Complex Catalyzed Heck Coupling Reaction† [J]. Chem. J. Chinese Universities, 2017, 38(9): 1627. |
| [10] | LIU Yuming, TIAN Lijun, HU Dong, NIE Jianbing. yntheses and Anti-cholinesterase Activity of 4-N-Phenylaminoquinoline Derivatives † [J]. Chem. J. Chinese Universities, 2017, 38(3): 392. |
| [11] | SHEN Xiaoqin, LI Zhi, WANG Ganglin, WANG Li, SUN Quanhong, LUO Xucheng, MA Nan. Logic and Reversible Dual DNA Detection Based on the Assembly of Graphene Oxide and DNA-templated Quantum Dots† [J]. Chem. J. Chinese Universities, 2017, 38(12): 2176. |
| [12] | YANG Zihui,LI Beibei,YE Jiao,HU Aixi. Synthesis and Herbicidal Activity of 2-(4-Arylxoyphenoxy)propionamide Derivatived from Benzofuranol† [J]. Chem. J. Chinese Universities, 2016, 37(8): 1442. |
| [13] | HUANG Chibao, ZHANG Daohai, ZENG Boping, LIU Qibin, CHEN Huashi, KANG Shuai, CHEN Xiaoyuan. Two-photon Fluorescent Labeling Kit for the Real-time Dynamic Biopsy of CA19-9† [J]. Chem. J. Chinese Universities, 2016, 37(4): 638. |
| [14] | ZHANG Xiaopeng, LI Zhengwei, WANG Yan, NIU Xueli, ZHANG Guisheng. Selenium-catalyzed Carbonylation of 4-Aminopyridine with Nitro Aromatics to 4-Pyridinylureas† [J]. Chem. J. Chinese Universities, 2016, 37(10): 1804. |
| [15] | ZHANG Jing, CHEN Weixiao, ZHANG Wei, DUAN Ying, ZHU Yuxiu, ZHU Yaxian, ZHANG Yong. Interaction of 1-Hydroxypyrene with BSA Using Fluorescence Anisotropy and Synchronous Fluorescence Analysis Methods† [J]. Chem. J. Chinese Universities, 2015, 36(8): 1511. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||