

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (3): 388.doi: 10.7503/cjcu20190631
• 庆祝《高等学校化学学报》复刊40周年专栏 • 上一篇 下一篇
高其龙1,梁二军1,邢献然2,陈骏3,*
收稿日期:2019-12-06
出版日期:2020-03-10
发布日期:2020-01-07
通讯作者:
陈骏
作者简介:陈 骏, 男, 博士, 研究员, 主要从事固体化学研究. E-mail: junchen@ustb.edu.cn
基金资助:GAO Qilong1,LIANG Erjun1,XING Xianran2,CHEN Jun3,*
Received:2019-12-06
Online:2020-03-10
Published:2020-01-07
Contact:
Jun CHEN
Supported by:摘要:
负热膨胀化合物可调控材料的热膨胀系数, 在复合材料、 精密仪器等方面具有重要的应用前景, 成为近年来化学、 物理和材料工程领域的研究热点之一. 因其晶体结构主要由—M—CN—M—含有双原子氰根(CN)链组成, 许多普鲁士蓝类化合物呈现反常的热膨胀性质. 本文综述了普鲁士蓝类负热膨胀化合物结构、 热膨胀机制与系数调控等方面的研究进展. 以氰根配体数量为分类主线, 将具有反常热膨胀性的氰根配体化合物分为氰化物、 六氰基和八氰基普鲁士蓝类化合物等进行介绍, 从局域结构和平均结构角度分析了N和C原子的横向振动对负热膨胀贡献的角度解释了机理, 从客体离子或分子嵌入的方法分析了热膨胀调控原理, 并对新型普鲁士蓝类负热膨胀化合物的设计及应用进行了展望.
中图分类号:
TrendMD:
高其龙, 梁二军, 邢献然, 陈骏. 普鲁士蓝类化合物负热膨胀及机理. 高等学校化学学报, 2020, 41(3): 388.
GAO Qilong, LIANG Erjun, XING Xianran, CHEN Jun. Negative Thermal Expansion in Prussian Blue Analogues †. Chem. J. Chinese Universities, 2020, 41(3): 388.
| Compound | 106αa/K-1 | 106αc/K-1 | 106αl/K-1 | ΔT/K | Crystal | Ref. |
|---|---|---|---|---|---|---|
| Au(CN) | 61.6 | -8.9 | 95—490 | Hexagonal | [ | |
| Ag(CN) | 70.8 | -24.8 | 95—490 | Trigonal | [ | |
| HT-Cu(CN) | 84.1 | -30.7 | 95—490 | Trigonal | [ | |
| Ni(CN)2 | -6.5 | 61.8 | 28—300 | Trigonal | [ | |
| Hg(CN)2 | 22.8 | 71.3 | 100—395 | Tetragonal | [ | |
| HgCN(NO3) | 26.1 | -23.0 | 100—395 | Hexagonal | [ | |
| Zn[Au(CN)2]2 | 36.9 | -57.5 | 100—775 | Hexagonal | [ | |
| Zn[Au(CN)2]2-xAgCN | 4.07 | -21.7 | 100—375 | Hexagonal | [ | |
| Cd(CN)2 | -20.4 | 150—375 | Cubic | [ | ||
| Zn(CN)2 | -16.9 | 25—375 | Cubic | [ | ||
| Single-network Cd(CN)2 | -33.5 | 170—375 | Cubic | [ | ||
| Cd(CN)2-0.64CCl4 | -16.9 | 240—375 | Cubic | [ | ||
| Cd(CN)2-0.75CCl4 | -5.7 | 200—375 | Cubic | [ | ||
| Cd(CN)2-CCl4 | 10.0 | 100—240 | Cubic | [ | ||
| Me4NCuZn(CN)4 | 0.67 | 218—368 | Cubic | [ |
Table 1 Structures and thermal expansion properties of metal cyanides
| Compound | 106αa/K-1 | 106αc/K-1 | 106αl/K-1 | ΔT/K | Crystal | Ref. |
|---|---|---|---|---|---|---|
| Au(CN) | 61.6 | -8.9 | 95—490 | Hexagonal | [ | |
| Ag(CN) | 70.8 | -24.8 | 95—490 | Trigonal | [ | |
| HT-Cu(CN) | 84.1 | -30.7 | 95—490 | Trigonal | [ | |
| Ni(CN)2 | -6.5 | 61.8 | 28—300 | Trigonal | [ | |
| Hg(CN)2 | 22.8 | 71.3 | 100—395 | Tetragonal | [ | |
| HgCN(NO3) | 26.1 | -23.0 | 100—395 | Hexagonal | [ | |
| Zn[Au(CN)2]2 | 36.9 | -57.5 | 100—775 | Hexagonal | [ | |
| Zn[Au(CN)2]2-xAgCN | 4.07 | -21.7 | 100—375 | Hexagonal | [ | |
| Cd(CN)2 | -20.4 | 150—375 | Cubic | [ | ||
| Zn(CN)2 | -16.9 | 25—375 | Cubic | [ | ||
| Single-network Cd(CN)2 | -33.5 | 170—375 | Cubic | [ | ||
| Cd(CN)2-0.64CCl4 | -16.9 | 240—375 | Cubic | [ | ||
| Cd(CN)2-0.75CCl4 | -5.7 | 200—375 | Cubic | [ | ||
| Cd(CN)2-CCl4 | 10.0 | 100—240 | Cubic | [ | ||
| Me4NCuZn(CN)4 | 0.67 | 218—368 | Cubic | [ |
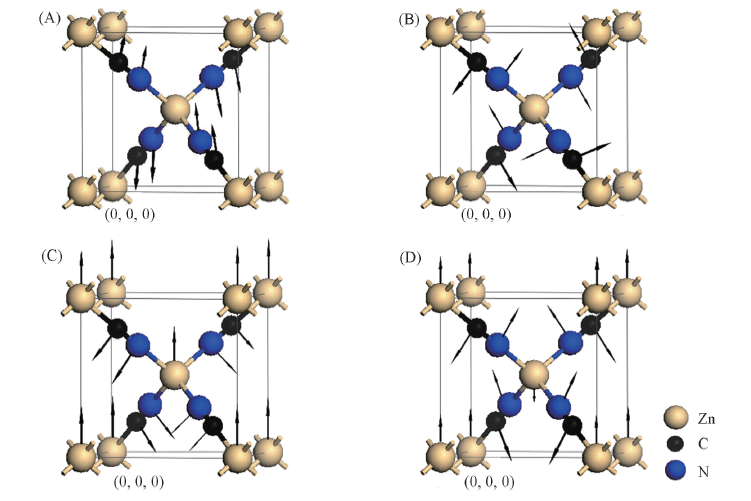
Fig.2 Typical vibrational manners of the phonon modes contributing to NTE in Zn(CN)2 of 47 cm-1(A), 263 cm-1(B), 189 cm-1(C) and 356 cm-1(D) [44] Copyright 2008, Chinese Physical Society.
| Compound | 106αV/K-1 | ΔT/K | Ref. | Compound | 106αV/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|---|---|---|
| FeCo(CN)6 | -4.41 | 4.2—300 | [ | YFe(CN)6 | -33.7 | 300—525 | [ |
| GaFe(CN)6 | -12 | 100—475 | [ | Mn3[Co(CN)6]2 | -87.6 | 123—300 | [ |
| FeFe(CN)6 | -12.8 | 100—475 | [ | Fe3[Co(CN)6]2 | -58.8 | 123—300 | [ |
| CdPt(CN)6 | -30.06 | 100—240 | [ | Co3[Co(CN)6]2 | -119.1 | 123—300 | [ |
| MnPt(CN)6 | -19.74 | 100—300 | [ | Ni3[Co(CN)6]2 | -90.0 | 123—300 | [ |
| FePt(CN)6 | -12 | 100—315 | [ | Cu3[Co(CN)6]2 | -60.0 | 123—300 | [ |
| ZnPt(CN)6 | -10.59 | 100—400 | [ | Zn3[Co(CN)6]2 | -89.1 | 123—300 | [ |
| CoPt(CN)6 | -4.8 | 100—350 | [ | Fe3[Fe(CN)6]2 | -29.7 | 123—300 | [ |
| NiPt(CN)6 | -3.06 | 100—330 | [ | Cu3[Fe(CN)6]2 | -59.7 | 123—300 | [ |
| CuPt(CN)6 | -4.71 | 100—400 | [ | Zn3[Fe(CN)6]2 | -118.8 | 123—300 | [ |
| ErCo(CN)6 | -27 | 100—500 | [ | Cs0.7Ni[Fe(CN)6]0.9·2.9H2O | -1.2 | 100—300 | [ |
| LaCo(CN)6 | -43.86 | 100—500 | [ | Cs0.97Cu[Fe(CN)6]0.99·1.1H2O | -6.3 | 100—300 | [ |
| SmCo(CN)6 | -37.38 | 100—500 | [ | Cs0.91Zn[Fe(CN)6]0.97·0.4H2O | -12.3 | 100—300 | [ |
| HoCo(CN)6 | -30.15 | 100—500 | [ | CsCd[Fe(CN)6]·0.5H2O | -26.4 | 100—300 | [ |
| LuCo(CN)6 | -27.15 | 100—500 | [ | Rb0.78Fe[Fe(CN)6]0.83·2.8H2O | -6.3 | 100—300 | [ |
| YCo(CN)6 | -31.59 | 100—500 | [ | Rb0.64Zn[Fe(CN)6]0.88·2.3H2O | -17.7 | 100—300 | [ |
Table 2 Negative thermal expansion properties in hexacyano-Prussian blue analogues
| Compound | 106αV/K-1 | ΔT/K | Ref. | Compound | 106αV/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|---|---|---|
| FeCo(CN)6 | -4.41 | 4.2—300 | [ | YFe(CN)6 | -33.7 | 300—525 | [ |
| GaFe(CN)6 | -12 | 100—475 | [ | Mn3[Co(CN)6]2 | -87.6 | 123—300 | [ |
| FeFe(CN)6 | -12.8 | 100—475 | [ | Fe3[Co(CN)6]2 | -58.8 | 123—300 | [ |
| CdPt(CN)6 | -30.06 | 100—240 | [ | Co3[Co(CN)6]2 | -119.1 | 123—300 | [ |
| MnPt(CN)6 | -19.74 | 100—300 | [ | Ni3[Co(CN)6]2 | -90.0 | 123—300 | [ |
| FePt(CN)6 | -12 | 100—315 | [ | Cu3[Co(CN)6]2 | -60.0 | 123—300 | [ |
| ZnPt(CN)6 | -10.59 | 100—400 | [ | Zn3[Co(CN)6]2 | -89.1 | 123—300 | [ |
| CoPt(CN)6 | -4.8 | 100—350 | [ | Fe3[Fe(CN)6]2 | -29.7 | 123—300 | [ |
| NiPt(CN)6 | -3.06 | 100—330 | [ | Cu3[Fe(CN)6]2 | -59.7 | 123—300 | [ |
| CuPt(CN)6 | -4.71 | 100—400 | [ | Zn3[Fe(CN)6]2 | -118.8 | 123—300 | [ |
| ErCo(CN)6 | -27 | 100—500 | [ | Cs0.7Ni[Fe(CN)6]0.9·2.9H2O | -1.2 | 100—300 | [ |
| LaCo(CN)6 | -43.86 | 100—500 | [ | Cs0.97Cu[Fe(CN)6]0.99·1.1H2O | -6.3 | 100—300 | [ |
| SmCo(CN)6 | -37.38 | 100—500 | [ | Cs0.91Zn[Fe(CN)6]0.97·0.4H2O | -12.3 | 100—300 | [ |
| HoCo(CN)6 | -30.15 | 100—500 | [ | CsCd[Fe(CN)6]·0.5H2O | -26.4 | 100—300 | [ |
| LuCo(CN)6 | -27.15 | 100—500 | [ | Rb0.78Fe[Fe(CN)6]0.83·2.8H2O | -6.3 | 100—300 | [ |
| YCo(CN)6 | -31.59 | 100—500 | [ | Rb0.64Zn[Fe(CN)6]0.88·2.3H2O | -17.7 | 100—300 | [ |
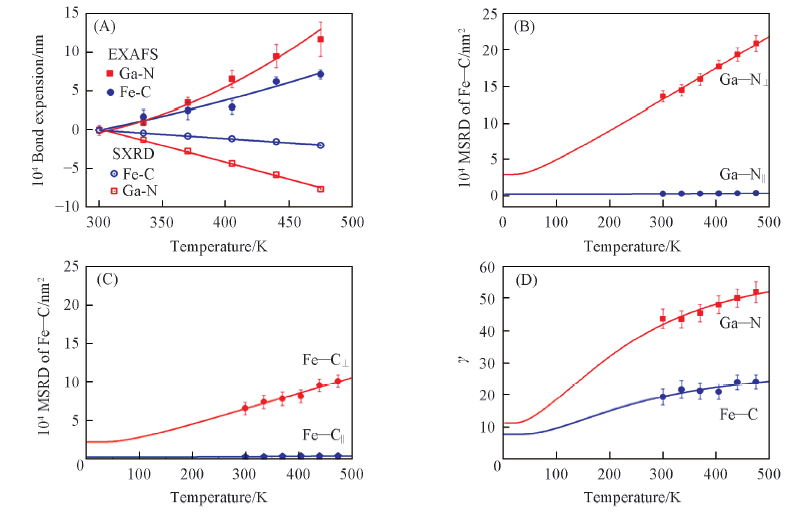
Fig.5 Comparison between the “apparent” and the “true” Ga—N and Fe—C bond lengths in GaFe(CN)6 as a function of temperature(A), Ga—N(B) and Fe—C(C) perpendicular(⊥) and parallel(‖) MSRDs of Ga—N atomic pairs of GaFe(CN)6 as a function of temperature, anisotropy of the relative thermal vibrations of the Ga—N and Fe—C atomic pairs of GaFe(CN)6 determined by EXAFS(D)[48] Copyright 2018, American Chemical Society.
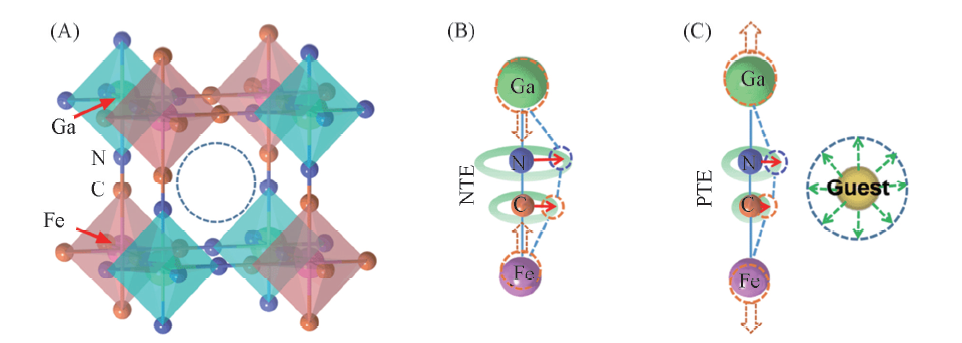
Fig.6 Sketch of the cubic structure of GaFe(CN)6 consisting of alternating GaN6 and FeC6 octahedra(A), the negative thermal expansion of GaFe(CN)6 derived from the transverse vibrations of CN cyanogen ions(B) and the transverse vibrations of CN cyanogen ions hindered by guest ions or molecules(C)[57] (A) The empty space indicated by the dash circle can host guest ions or molecules. Copyright 2018, American Chemical Society.
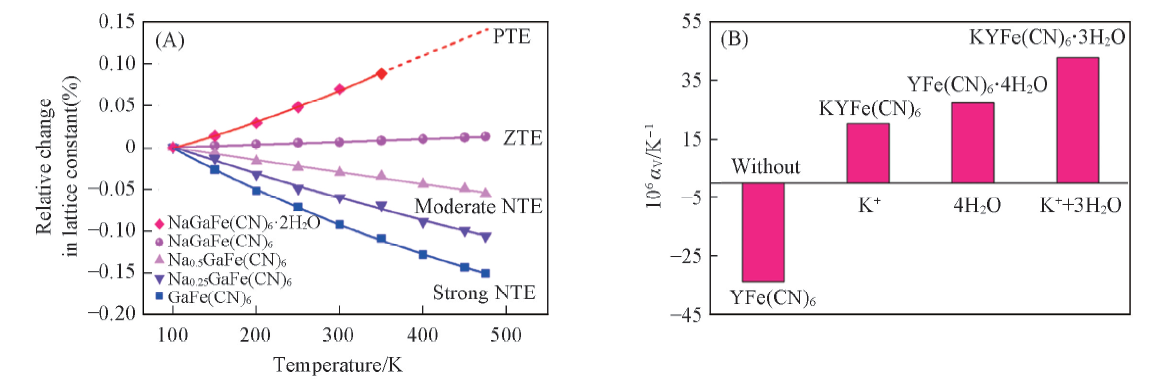
Fig.7 Temperature dependence of the relative change in lattice constant of GaFe(CN)6-based compounds(A)[57] and the coefficient of thermal expansion for YFe(CN)6-based materials within different guests(B)[28] (A) Copyright 2018, American Chemical Society; (B) copyright 2017, Wiley.
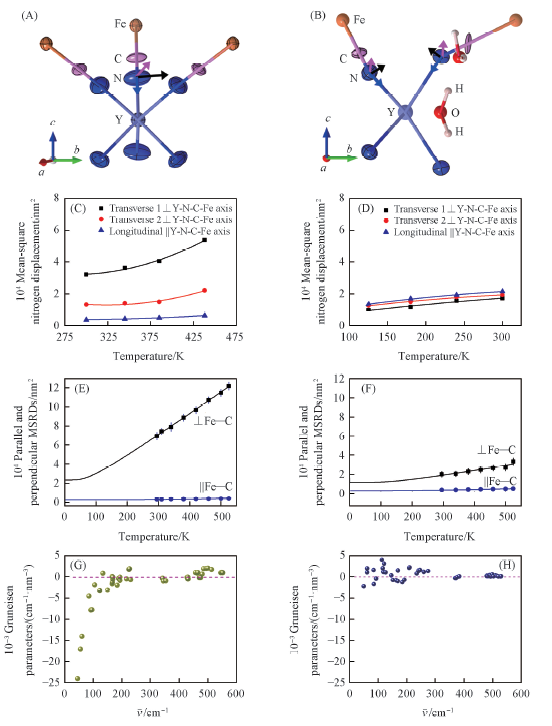
Fig.8 ADPs depiction and its temperature dependence for N atoms in YFe(CN)6(A,C) and YFe(CN)6·4H2O(B,D), respectively, the temperature dependence of MSRDs of the Fe—C atomic and the frequency dependence of mode Grüneisen parameters in YFe(CN)6(E,G) and KYFe(CN)6(F,H), respectively[28] Copyright 2017, Wiley.
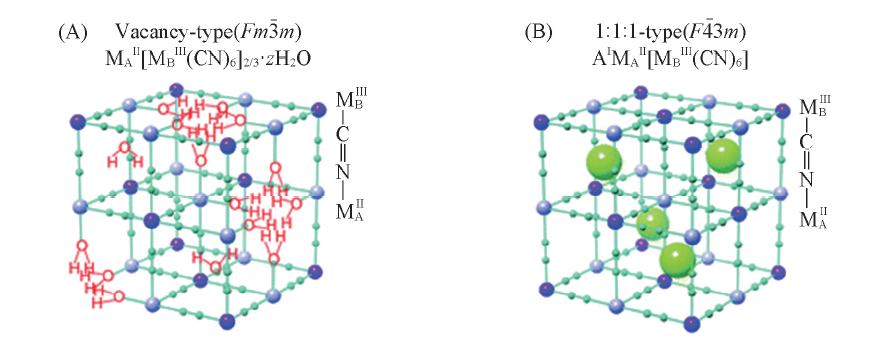
Fig.9 Schematic crystal structures of Prussian blue analogues MAⅡ[MBⅢ(CN)6]2/3·zH2O(A) and AⅠMⅡ[MⅢ(CN)6](B) [59] Copyright 2011, Royal Society of Chemistry.
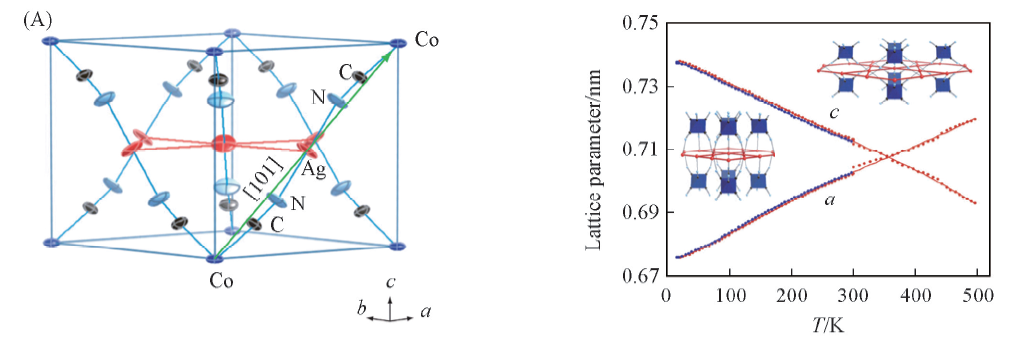
Fig.10 Structure of Ag3Co(CN)6, the strongest bonding interactions, which occur within Co—CN—Ag—NC—Co linkages, all lie parallel to the crystallographic [101] directions(A) and the lattice parameters for Ag3Co(CN)6 as a function of temperature(B)[20] (B) Inset show the sketch of large changes for Ag…Ag and [Co(CN)6]…[Co(CN)6] unit. Copyright 2008, Science.
| Compound | 106a/K-1 | 106c/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|
| H3Co(CN)6 | 12.0(4) | -8.8(3) | 4—300 | [ |
| D3Co(CN)6 | 17.4(6) | -5.04(24) | 8—304 | [ |
| Cu3Co(CN)6 | 25.4(5) | -43.5(8) | 100—596 | [ |
| Ag3Co(CN)6 | 145.9(6) | -122.1(3) | 16—500 | [ |
| Ag3Fe(CN)6 | 124.0(10) | -113(3) | 8—310 | [ |
| In[Au(CN)2]3 | 86.1—83.9 | -62.2—-63.3 | 100—395 | [ |
| In[Ag(CN)2]3 | 104—106 | -85—-83 | 100—395 | [ |
| KCd[Au(CN)2]3 | 72.9—71.4 | -56.1—-57.1 | 100—395 | [ |
| KCd[Ag(CN)2]3 | 76.6—74.9 | -64.3—-65.6 | 100—395 | [ |
| KNi[Au(CN)2]3 | 59.7—58.7 | -42.2—-42.7 | 100—395 | [ |
| KMn[Ag(CN)2]3 | 61(2) | -60(3) | 100—395 | [ |
Table 3 Thermal expansion prepoerties of Ag3Co(CN)6-typed PBAs
| Compound | 106a/K-1 | 106c/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|
| H3Co(CN)6 | 12.0(4) | -8.8(3) | 4—300 | [ |
| D3Co(CN)6 | 17.4(6) | -5.04(24) | 8—304 | [ |
| Cu3Co(CN)6 | 25.4(5) | -43.5(8) | 100—596 | [ |
| Ag3Co(CN)6 | 145.9(6) | -122.1(3) | 16—500 | [ |
| Ag3Fe(CN)6 | 124.0(10) | -113(3) | 8—310 | [ |
| In[Au(CN)2]3 | 86.1—83.9 | -62.2—-63.3 | 100—395 | [ |
| In[Ag(CN)2]3 | 104—106 | -85—-83 | 100—395 | [ |
| KCd[Au(CN)2]3 | 72.9—71.4 | -56.1—-57.1 | 100—395 | [ |
| KCd[Ag(CN)2]3 | 76.6—74.9 | -64.3—-65.6 | 100—395 | [ |
| KNi[Au(CN)2]3 | 59.7—58.7 | -42.2—-42.7 | 100—395 | [ |
| KMn[Ag(CN)2]3 | 61(2) | -60(3) | 100—395 | [ |
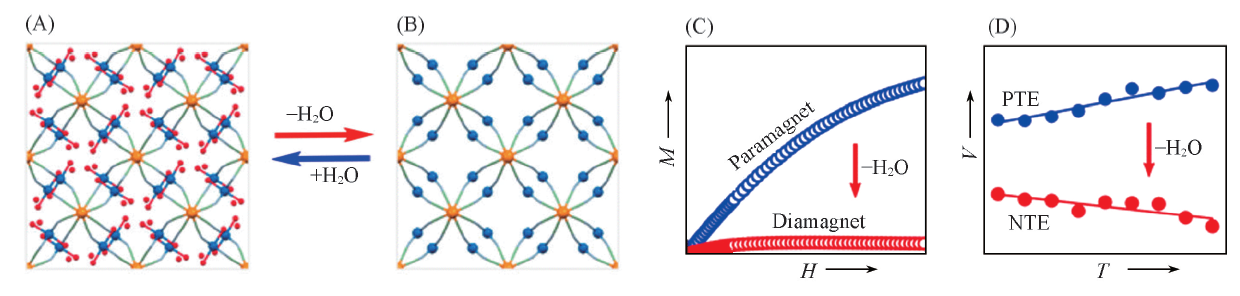
Fig.11 Structure schematic of Ni2Ⅱ[WⅣ(CN)8]·6H2O(A) and Ni2Ⅱ[WⅣ(CN)8](B), the comparison physical property of Ni2Ⅱ[WⅣ(CN)8]·6H2O and Ni2Ⅱ[WⅣ(CN)8] in magnetism(C) and thermal expansion(D) [32] Copyright 2016, American Chemical Society.
| [1] | Chen J., Hu L., Deng J., Xing X ., Chem. Soc. Rev., 2015, 44, 3522— 3567 |
| [2] | Pryde A. K. A., Hammonds K. D., Dove M. T., Heine V., Gale J. D., Warren M. C ., J. Phys. Condens. Matter., 1996, 8, 10973— 10982 |
| [3] | Ernst G., Broholm C., Kowach G. R., Ramirez A. P ., Nature, 1998, 396, 56— 57 |
| [4] | Ramirez A. P., Kowach G. R ., Phys. Rev. Lett., 1998, 80, 4903— 4906 |
| [5] | Yuan B., Liu X., Song W., Cheng Y., Liang E., Chao M ., Phys. Lett. A, 2014, 378, 3397— 3401 |
| [6] | Chen D., Yuan B., Cheng Y., Ge X., Jia Y., Liang E., Chao M ., Phys. Lett. A, 2016, 380, 4070— 4074 |
| [7] | Huang L. P., Kieffer J ., Phys. Rev. Lett., 2005, 95, 215901 |
| [8] | Yamamura Y., Ikeuchi S., Saito K ., Chem. Mater., 2009, 21, 3008— 3016 |
| [9] | Qiu L., Bao Y., Wen S., Liang E. J ., Chinese Phys. B, 2012, 21, 046501 |
| [10] | Liu Y., Zhang N., Jia Y., Sun Q., Chao M ., Phys. Lett. A, 2015, 379, 2756— 2760 |
| [11] | Liang E. J ., Chinese Phys. Lett., 2013, 30, 126502 |
| [12] | Chang D., Yu W., Sun Q., Jia Y ., Phys. Chem. Chem. Phys., 2017, 19,2067—2072 |
| [13] | Woodcock D. A., Lightfoot P., Villaescusa L. A., Diaz-Cabanas M. J ., Chem. Mater., 1999, 11, 2508— 2514 |
| [14] | Ge X., Liu X., Cheng Y., Yuan B., Chen D., Chao M., Liang E. J ., J. Appl. Phys., 2016, 120, 205101 |
| [15] | Carey T., Corma A., Rey F., Tang C. C., Hriljac J. A., Anderson P. A ., Chem. Commun., 2012, 48, 5829— 5831 |
| [16] | Goodwin A. L., Chapman K. W., Kepert C. J ., J. Am. Chem. Soc., 2005, 127, 17980— 17981 |
| [17] | Hibble S. J., Chippindale A. M., Pohl A. H., Hannon A. C ., Angew. Chem. Int. Ed., 2007, 46, 7116— 7118 |
| [18] | Margadonna S., Prassides K., Fitch A. N ., J. Am. Chem. Soc., 2005, 126, 15390— 15391 |
| [19] | Duyker S. G., Peterson V. K., Kearley G. J., Anibal J. R., Kepert C. J ., Angew. Chem., 2013, 125, 5374— 5378 |
| [20] | Goodwin A. L., Calleja M., Conterio M. J., Dove M. T., Evans J. S. O., Keen D. A., Peters L., Tucker M. G ., Science, 2008, 319, 794— 797 |
| [21] | Li M., Li Y., Wang C. Y., Sun Q ., Chinese Phys. Lett., 2019, 36, 066301 |
| [22] | Liu Z., Liu C., Li Q., Chen J., Xing X ., Phys. Chem. Chem. Phys., 2017, 19, 24436— 24439 |
| [23] | Dubbeldam D., Walton K. S., Ellis D. E., Snurr R. Q ., Angew. Chem. Int. Ed., 2007, 119, 4580— 4583 |
| [24] | Wang L., Yuan P. F., Wang F., Sun Q., Liang E. J., Jia Y., Guo Z. X ., Phys. Lett. A, 2014, 378, 2906— 2909 |
| [25] | Hu L., Chen J., Fan L., Ren Y., Xing X ., J. Am. Chem. Soc., 2014, 136, 13566— 13569 |
| [26] | Hu L., Chen J., Sanson A Wu H., Rodriguez C. G., Olivi L., Ren Y., Fan L., Deng J., Xing X ., J. Am. Chem. Soc., 2016, 138, 8320— 8323 |
| [27] | Chang D., Niu C. Y., Huang X., Sun Q., Cho J. H., Jia Y ., Phys. Rev. B, 2017, 95, 104101 |
| [28] | Gao Q. L., Chen J., Sun Q., Chang D. H., Huang Q. Z., Wu H., Sanson A., Milazzo R., Zhu H., Li Q., Liu Z. N., Deng J. X., Xing X. R ., Angew. Chem. Int. Ed., 2017, 56, 9023— 9028 |
| [29] | Mary T. A., Evans J. S. O., Vogt T., Sleight A. W ., Science, 1996, 272, 90— 92 |
| [30] | Liu Y., Jia Y., Sun Q., Liang E. J ., Phys. Lett. A, 2015, 379, 54— 59 |
| [31] | Buser H. J., Schwarzenbach D., Petter W., Ludi A ., Inorg. Chem., 1977, 16, 2704— 2710 |
| [32] | Reczyński M., Chorazy S., Nowicka B., Sieklucka B., Ohkoshi S ., Inorg. Chem., 2016, 56, 179— 185 |
| [33] | Tokoro H., Nakagawa K., Imoto K., Hakoe F., Ohkoshi S ., Chem. Mater., 2012, 24, 1324— 1330 |
| [34] | Chippindale A. M., Hibble S. J., Bilbé E. J., Marelli E., Hannon A. C., Allain C., Pansu R., Hartl F ., J. Am. Chem. Soc., 2012, 134, 16387— 16400 |
| [35] | Goodwin A. L., Kepert C. J ., Phys. Rev. B, 2005, 71, 140301 |
| [36] | Williams D. J., Partin D. E., Lincoln F. J., Kouvetakis J., O'Keeffe M ., J. Solid State Chem., 1997, 134, 164— 169 |
| [37] | Hibble S. J., Hannon A. C., Cheyne S. M ., Inorg. Chem., 2003, 34, 4724— 4730 |
| [38] | Korčok J. L., Leznoff D. B ., Polyhedron, 2013, 52, 72— 77 |
| [39] | Goodwin A. L., Kennedy B. J., Kepert C. J ., J. Am. Chem. Soc., 2009, 131, 6334— 6335 |
| [40] | Phillips A. E., Goodwin A. L., Halder G. J., Southon P. D., Kepert C. J ., Angew. Chem. Int. Ed., 2008, 47, 1396— 1399 |
| [41] | Phillips A. E., Halder G. J., Chapman K. W., Goodwin A. L., Kepert C. J ., J. Am. Chem. Soc., 2009, 132, 10— 11 |
| [42] | Zwanziger J. W ., Phys. Rev. B, 2007, 76, 052102 |
| [43] | Ding P., Liang E. J., Jia Y., Du Z ., J. Phys. Condens. Matter, 2008, 20, 275224 |
| [44] | Du Z. Y., Liang E. J., Ding P., Wang J. P., Xu E. M ., J. Light Scattering, 2008, 20( 2), 145— 150 |
| ( 杜志勇, 梁二军, 丁佩, 王俊平, 徐二明 . 光散射学报, 2008, 20( 2), 145— 150) | |
| [45] | Hibble S. J., Chippindale A. M., Marelli E., Kroeker S., Michaelis V. K., Greer B. J., Aguiar P. M., Bilbé E. J., Barney E. R., Hannon A. C ., J. Am. Chem. Soc., 2013, 135, 16478— 16489 |
| [46] | Chapman K. W., Peter J. C ., J. Am. Chem. Soc., 2007, 129, 10090— 10091 |
| [47] | Ovens J. S., Leznoff D. B ., Inorg. Chem., 2017, 56, 7332— 7343 |
| [48] | Gao Q., Shi N., Sun Q., Sanson A., Milazzo R., Carnera A., Zhu H., Lapidus S. H., Ren Y., Huang Q., Chen J., Xing X ., Inorg. Chem., 2018, 57, 10918— 10924 |
| [49] | Shi N., Gao Q., Sanson A., Li Q., Fan L., Ren Y., Olivi L., Chen J., Xing X ., Dalton T., 2019, 48, 3658— 3663 |
| [50] | Chapman K. W., Chupas P. J., Kepert C. J ., J. Am. Chem. Soc., 2010, 37, 7009— 7014 |
| [51] | Pretsch T., Chapman K. W., Halder G. J., Kepert C. J ., Chem. Commun., 2006, ( 17), 1857— 1859 |
| [52] | Adak S., Daemen L. L., Hartl M., Williams D., Summerhill J., Nakotte H ., J. Solid State Chem., 2011, 184, 2854— 2861 |
| [53] | Matsuda T., Kim J. E., Ohoyama K., Moritomo Y ., Phys. Rev. B, 2009, 79, 172302 |
| [54] | Sanson A ., Chem. Mater., 2014, 26, 3716— 3720 |
| [55] | Chapman K. W., Chupas P. J., Kepert C. J ., J. Am. Chem. Soc., 2005, 127, 15630— 15636 |
| [56] | Vaccari M., Grisenti R., Fornasini P., Rocca F., Sanson A ., Phys. Rev. B, 2007, 75, 184307 |
| [57] | Gao Q., Shi N., Sanson A., Sun Y., Milazzo R., Olivi L., Chen J., Xing X ., Inorg. Chem., 2018, 57, 14027— 14030 |
| [58] | Chen J., Gao Q. L., Sanson A., Jiang X. X., Huang Q. Z., Carnera A., Rodriguez C. G., Olivi L., Wang L., Hu L., Lin K., Ren Y., Lin Z. S., Wang C., Gu L., Deng J. X., Attfield J. P., Xing X. R ., Nat. Commun. 2017, 8, 14441 |
| [59] | Tokoro H., Ohkoshi S. I ., Dalton T., 2011, 40, 6825— 6833 |
| [60] | Adak S., Daemen L. L., Nakotte H ., J. Phys Conf. Ser., 2010, 251, 012007 |
| [61] | Matsuda T., Tokoro H., Hashimoto K., Ohkoshi S. I ., Dalton T., 2006, 42, 5046— 5050 |
| [62] | Korcok J. L., Katz M. J., Leznoff D. B ., J. Am. Chem. Soc., 2009, 131, 4866— 4871 |
| [63] | Wang L., Wang C., Luo H., Sun Y ., J. Phys. Chem. C, 2017, 121, 333— 341 |
| [64] | Cairns A. B., Thompson A. L., Tucker M. G., Haines J., Goodwin A. L ., J. Am. Chem. Soc., 2012, 134, 4454— 4456 |
| [65] | Keen D. A., Dove M. T., Evans J. S., Goodwin A. L., Peters L., Tucker M. G ., J. Phys-Condens Mat., 2010, 22, 404202 |
| [66] | Sapnik A. F., Liu X., Boström H. L. B., Coates C. S., Overy A. R., Reynolds E. M., Tkatchenko A., Goodwin A. L ., J. Solid State Chem., 2018, 258, 298— 306 |
| [1] | 施耐克, 张娅, SANSON Andrea, 王蕾, 陈骏. Zn(NCN)单轴的负热膨胀性及机理研究[J]. 高等学校化学学报, 2022, 43(6): 20220124. |
| [2] | 任玉英 周鼎 郭婧 施鹰 谢建军 谢治 贺博. Nd3+在Lu2O3纳米粉体和透明陶瓷中的局域结构研究[J]. 高等学校化学学报, 2011, 32(1): 32. |
| [3] | 刘芹芹, 杨娟, 孙秀娟, 程晓农 . 不同形貌ZrWMoO8粉体的制备、表征及其负热膨胀特性[J]. 高等学校化学学报, 2007, 28(3): 397. |
| [4] | 郭振亚, 王敬中, 王凯旋, 周达明, 项寿鹤, 李赫咺. 直接法合成ZSM-5型分子筛的物理化学表征[J]. 高等学校化学学报, 1990, 11(8): 800. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||