

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (5): 899.doi: 10.7503/cjcu20141138
收稿日期:2014-12-29
出版日期:2015-05-10
发布日期:2015-04-13
作者简介:联系人简介: 郑敏燕, 女, 博士, 教授, 主要从事功能材料研究.E-mail: 基金资助:
ZHENG Minyan*( ), WEI Yongsheng, GENG Wei
), WEI Yongsheng, GENG Wei
Received:2014-12-29
Online:2015-05-10
Published:2015-04-13
Contact:
ZHENG Minyan
E-mail:zmy71@126.com
Supported by:摘要:
以4-烷基环己基甲酸(C1~C3)、 4-烷基苯甲酸(C1~C5)和对羟基偶氮苯甲酸为主要原料, 通过室温一步酯化反应, 合成了2个系列8个不对称酯基偶氮苯甲酸类多官能团液晶化合物. 目标产物的结构、 液晶性及光敏性采用红外光谱(IR)、 核磁共振谱(NMR)、 质谱(MS)、 元素分析、 示差扫描量热分析(DSC)、 热台偏光显微镜(HS-POM)和紫外-可见光谱(UV-Vis)等表征. 利用循环伏安法(CV)测定了目标分子的前线轨道能级. 测试结果表明, 8个化合物均有液晶性, 在甲醇溶液中显现出光敏性, 在液晶态也观察到光顺反异构化. 端烷基碳原子数对化合物紫外吸收峰Ⅱ(偶氮结构π-π*带)、 HOMO能级、 LUMO能级和能隙Eg存在奇偶效应影响. 烷基为奇碳数化合物的Eg低于相邻烷基为偶碳数化合物. 这与目标分子紫外光谱变化趋势相吻合, 即Eg较小的化合物在紫外光谱中峰Ⅱ的吸收波长较长(能量较低), 反之亦然.
中图分类号:
TrendMD:
郑敏燕, 魏永生, 耿薇. 不对称三环偶氮苯甲酸液晶的合成、光敏性及电化学性质. 高等学校化学学报, 2015, 36(5): 899.
ZHENG Minyan, WEI Yongsheng, GENG Wei. Synthesis, Photosensitive and Electrochemical Properties of Asymmetric Liquid Crystals Based on Tri-ring Azo-benzoic Acid†. Chem. J. Chinese Universities, 2015, 36(5): 899.
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | C | H | ||||||||
| 3a | Orange needle | 82.4 | 211.2—212.6 | 3200—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559, 1541, 1449(m, ArH), 1428, 1276, 1222, 1201, 1178, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 807(w, 1,4-Ar), 688 [w,(CH2)n] | 366 (366.41) | 68.94 (68.84) | 6.30 (6.05) | 7.83 (7.65) | ||
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), ν/cm-1 | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
| N | C | H | ||||||||
| 3b | Orange needle | 80.2 | 221.7—222.8 | 3200—2500(m, O—H), 2950, 2930, 2864(s, C—H), 1755, 1697(vs, C═O), 1603(m, N═N), 1563, 1499(m, ArH), 1427, 1290, 1223, 1200, 1141, 1121, 1009(vs, C—O—C), 868(s, trans-R—N═N—R), 809(w, 1, 4-Ar), 690[w,(CH2)n] | 380 (380.44) | 69.67 (69.46) | 6.54 (6.36) | 7.49 (7.36) | ||
| 3c | Orange needle | 84.0 | 208.9—209.5 | 3000—2500(m, O—H), 2960, 2931(s, C—H), 1755, 1680(vs, C═O), 1603(m, N═N), 1592, 1500, 1449(m, ArH), 1428, 1290, 1224, 1201, 1142, 1122, 1101, 1076, 1010(vs, C—O—C), 869(s, trans-R—N═N—R), 808(w, 1, 4-Ar), 765 [w,(CH2)n] | 394 (394.46) | 70.22 (70.03) | 6.73 (6.64) | 7.15 (7.10) | ||
| 3d | Orange needle | 81.1 | 198.0—199.7 | 3000—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559(m, ArH), 1498, 1428, 1276, 1222, 1201, 1178, 1141, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 812(w, 1, 4-Ar), 776 [w,(CH2)n] | 360 (360.11) | 70.14 (69.99) | 4.60 (4.48) | 7.93 (7.77) | ||
| 3e | Orange needle | 80.7 | 200.4—201.9 | 3000—2500(m, O—H), 3097, 3071, 2968, 2853(s, C—H), 1735, 1685(vs, C═O), 1602(m, N═N), 1559, 1541, 1496, 1458(m, ArH), 1266, 1223, 1195, 1177, 1142, 1068, 1009(vs, C—O—C), 882(s, trans-R—N═N—R), 811(w, 1,4-Ar), 766 [w,(CH2)n] | 374 (374.39) | 70.77 (70.58) | 4.90 (4.85) | 7.66 (7.48) | ||
| 3f | Orange needle | 85.2 | 199.5—200.9 | 3100—2500(m, O—H), 2958, 2929, 2871(s, C—H), 1734, 1684(vs, C═O), 1604(m, N═N), 1495, 1464(m, ArH), 1309, 1267, 1223, 1196, 1141, 1099, 1061, 1010(vs, C—O—C), 882(s, trans-R—N═N—R), 831(w, 1, 4-Ar), 776 [w,(CH2)n] | 388 (388.42) | 71.33 (71.12) | 5.30 (5.19) | 7.39 (7.21) | ||
| 3g | Orange needle | 88.6 | 186.3—187.8 | 3000—2500(m, O—H), 2956, 2927, 2857(s, C—H), 1734, 1681(vs, C═O), 1603(m, N═N), 1595, 1495(m, ArH), 1416, 1379, 1270, 1223, 1198, 1176, 1141, 1100, 1068, 1010(vs, C—O—C), 946, 880(s, trans-R—N═N—R), 846(w, 1, 4-Ar), 775 [w,(CH2)n] | 402 (402.44) | 71.78 (71.63) | 5.66 (5.51) | 6.75 (6.96) | ||
| 3h | Orange needle | 84.3 | 187.1—188.5 | 3000—2500(m, O—H), 2929, 2857(s, C—H), 1786, 1685(vs, C═O), 1602(m, N═N), 1541, 1468(m, ArH), 1417, 1358, 1287, 1255, 1220, 1142, 1101, 1041, 1008(vs, C—O—C), 943, 882(s, trans-R—N═N—R), 817(w, 1, 4-Ar), 721 [w,(CH2)n] | 416 (416.47) | 72.33 (72.10) | 5.99 (5.81) | 6.88 (6.73) | ||
Table 1 Appearance, yields, melting points, IR, MS data and elemental analysis of target compounds
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| N | C | H | ||||||||
| 3a | Orange needle | 82.4 | 211.2—212.6 | 3200—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559, 1541, 1449(m, ArH), 1428, 1276, 1222, 1201, 1178, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 807(w, 1,4-Ar), 688 [w,(CH2)n] | 366 (366.41) | 68.94 (68.84) | 6.30 (6.05) | 7.83 (7.65) | ||
| Compd. | Appea- rance | Yield (%) | m.p./℃ | IR(KBr), ν/cm-1 | MS([M+], calcd.), m/z | Elemental analysis(%, calcd.) | ||||
| N | C | H | ||||||||
| 3b | Orange needle | 80.2 | 221.7—222.8 | 3200—2500(m, O—H), 2950, 2930, 2864(s, C—H), 1755, 1697(vs, C═O), 1603(m, N═N), 1563, 1499(m, ArH), 1427, 1290, 1223, 1200, 1141, 1121, 1009(vs, C—O—C), 868(s, trans-R—N═N—R), 809(w, 1, 4-Ar), 690[w,(CH2)n] | 380 (380.44) | 69.67 (69.46) | 6.54 (6.36) | 7.49 (7.36) | ||
| 3c | Orange needle | 84.0 | 208.9—209.5 | 3000—2500(m, O—H), 2960, 2931(s, C—H), 1755, 1680(vs, C═O), 1603(m, N═N), 1592, 1500, 1449(m, ArH), 1428, 1290, 1224, 1201, 1142, 1122, 1101, 1076, 1010(vs, C—O—C), 869(s, trans-R—N═N—R), 808(w, 1, 4-Ar), 765 [w,(CH2)n] | 394 (394.46) | 70.22 (70.03) | 6.73 (6.64) | 7.15 (7.10) | ||
| 3d | Orange needle | 81.1 | 198.0—199.7 | 3000—2500(m, O—H), 3069, 2994, 2922, 2882(s, C—H), 1734, 1684(vs, C═O), 1602(m, N═N), 1594, 1559(m, ArH), 1498, 1428, 1276, 1222, 1201, 1178, 1141, 1074, 1010(vs, C—O—C), 879(s, trans-R—N═N—R), 812(w, 1, 4-Ar), 776 [w,(CH2)n] | 360 (360.11) | 70.14 (69.99) | 4.60 (4.48) | 7.93 (7.77) | ||
| 3e | Orange needle | 80.7 | 200.4—201.9 | 3000—2500(m, O—H), 3097, 3071, 2968, 2853(s, C—H), 1735, 1685(vs, C═O), 1602(m, N═N), 1559, 1541, 1496, 1458(m, ArH), 1266, 1223, 1195, 1177, 1142, 1068, 1009(vs, C—O—C), 882(s, trans-R—N═N—R), 811(w, 1,4-Ar), 766 [w,(CH2)n] | 374 (374.39) | 70.77 (70.58) | 4.90 (4.85) | 7.66 (7.48) | ||
| 3f | Orange needle | 85.2 | 199.5—200.9 | 3100—2500(m, O—H), 2958, 2929, 2871(s, C—H), 1734, 1684(vs, C═O), 1604(m, N═N), 1495, 1464(m, ArH), 1309, 1267, 1223, 1196, 1141, 1099, 1061, 1010(vs, C—O—C), 882(s, trans-R—N═N—R), 831(w, 1, 4-Ar), 776 [w,(CH2)n] | 388 (388.42) | 71.33 (71.12) | 5.30 (5.19) | 7.39 (7.21) | ||
| 3g | Orange needle | 88.6 | 186.3—187.8 | 3000—2500(m, O—H), 2956, 2927, 2857(s, C—H), 1734, 1681(vs, C═O), 1603(m, N═N), 1595, 1495(m, ArH), 1416, 1379, 1270, 1223, 1198, 1176, 1141, 1100, 1068, 1010(vs, C—O—C), 946, 880(s, trans-R—N═N—R), 846(w, 1, 4-Ar), 775 [w,(CH2)n] | 402 (402.44) | 71.78 (71.63) | 5.66 (5.51) | 6.75 (6.96) | ||
| 3h | Orange needle | 84.3 | 187.1—188.5 | 3000—2500(m, O—H), 2929, 2857(s, C—H), 1786, 1685(vs, C═O), 1602(m, N═N), 1541, 1468(m, ArH), 1417, 1358, 1287, 1255, 1220, 1142, 1101, 1041, 1008(vs, C—O—C), 943, 882(s, trans-R—N═N—R), 817(w, 1, 4-Ar), 721 [w,(CH2)n] | 416 (416.47) | 72.33 (72.10) | 5.99 (5.81) | 6.88 (6.73) | ||
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
|---|---|
| 3a | 0.90(s, 3H, CH3), 1.40—2.97(m, 10H, 10cyclohexyl-H), 7.53, 7.97, 8.01, 8.23(d, J=8.0, 8.4, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.96(s, 1H, COOH) |
| 3b | 0.89(t, 3H, J=7.6 Hz, CH3), 1.25—1.26(m, 2H, CH2), 1.25—2.18(m, 10H, 10cyclohexyl-H), 7.27, 7.64, 7.80, 8.25(d, J=8.0, 9.2, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.81(s, 1H, COOH) |
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
| 3c | 0.88(t, 3H, J=7.2 Hz, CH3), 1.20—1.27(m, 2H, CH2), 1.24(m, 2H, CH2), 1.57—2.60(m, 10H, 10cyclohexyl-H), 7.30, 7.48, 8.01, 8.31(d, J=8.0, 8.4, 8.4, 8.0 Hz, 2H of each, 8Ph-H), 12.73(s, 1H, COOH) |
| 3d | 2.41(s, 3H, CH3), 7.39, 7.41, 8.01, 8.04, 8.32, 8.41(d, J=7.2, 7.2, 8.0, 8.0, 7.2, 7.2 Hz, 2H of each, 12Ph-H), 13.11(s, 1H, COOH) |
| 3e | 1.27(t, J=7.2 Hz, 3H, CH3), 2.74(q, J=7.2 Hz, 2H, CH2), 7.29, 7.35, 7.41, 8.03, 8.12, 8.25(d, J=8.0, 8.4, 8.4, 8.0, 8.0, 8.0 Hz, 2H of each, 12Ph-H), 12.91(s, 1H, COOH) |
| 3f | 0.97(t, 3H, J=7.2 Hz, CH3), 1.70(m, 2H, CH2), 2.68(t, J=7.2 Hz, 2H, CH2), 7.32, 7.34, 7.98, 8.01, 8.11, 8.35(d, J=8.0, 8.4, 8.0, 8.0, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 12.86(s, 1H, COOH) |
| 3g | 0.90(t, 3H, J=7.2 Hz, CH3), 1.29—1.37(m, 2H, CH2), 1.54—1.65(m, 2H, CH2), 2.66(t, J=7.2 Hz, 2H, CH2), 7.29, 7.36, 7.97, 8.02, 8.09, 8.22(d, J=8.4, 8.8, 8.4, 8.8, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.01(s, 1H, COOH) |
| 3h | 0.90(t, 3H, J=6.8 Hz, CH3), 1.25—1.44(m, 6H, CH2CH2CH2), 1.64(t, 2H, J=7.2 Hz, CH2), 7.28, 7.32, 8.03, 8.06, 8.22, 8.28(d, J=8.4, 8.4, 8.0, 8.4, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.02(s, 1H, COOH) |
Table 2 1H NMR data for target compounds
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
|---|---|
| 3a | 0.90(s, 3H, CH3), 1.40—2.97(m, 10H, 10cyclohexyl-H), 7.53, 7.97, 8.01, 8.23(d, J=8.0, 8.4, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.96(s, 1H, COOH) |
| 3b | 0.89(t, 3H, J=7.6 Hz, CH3), 1.25—1.26(m, 2H, CH2), 1.25—2.18(m, 10H, 10cyclohexyl-H), 7.27, 7.64, 7.80, 8.25(d, J=8.0, 9.2, 8.8, 8.0 Hz, 2H of each, 8Ph-H), 12.81(s, 1H, COOH) |
| Compd. | 1H NMR(400 MHz, CDCl3), δ |
| 3c | 0.88(t, 3H, J=7.2 Hz, CH3), 1.20—1.27(m, 2H, CH2), 1.24(m, 2H, CH2), 1.57—2.60(m, 10H, 10cyclohexyl-H), 7.30, 7.48, 8.01, 8.31(d, J=8.0, 8.4, 8.4, 8.0 Hz, 2H of each, 8Ph-H), 12.73(s, 1H, COOH) |
| 3d | 2.41(s, 3H, CH3), 7.39, 7.41, 8.01, 8.04, 8.32, 8.41(d, J=7.2, 7.2, 8.0, 8.0, 7.2, 7.2 Hz, 2H of each, 12Ph-H), 13.11(s, 1H, COOH) |
| 3e | 1.27(t, J=7.2 Hz, 3H, CH3), 2.74(q, J=7.2 Hz, 2H, CH2), 7.29, 7.35, 7.41, 8.03, 8.12, 8.25(d, J=8.0, 8.4, 8.4, 8.0, 8.0, 8.0 Hz, 2H of each, 12Ph-H), 12.91(s, 1H, COOH) |
| 3f | 0.97(t, 3H, J=7.2 Hz, CH3), 1.70(m, 2H, CH2), 2.68(t, J=7.2 Hz, 2H, CH2), 7.32, 7.34, 7.98, 8.01, 8.11, 8.35(d, J=8.0, 8.4, 8.0, 8.0, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 12.86(s, 1H, COOH) |
| 3g | 0.90(t, 3H, J=7.2 Hz, CH3), 1.29—1.37(m, 2H, CH2), 1.54—1.65(m, 2H, CH2), 2.66(t, J=7.2 Hz, 2H, CH2), 7.29, 7.36, 7.97, 8.02, 8.09, 8.22(d, J=8.4, 8.8, 8.4, 8.8, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.01(s, 1H, COOH) |
| 3h | 0.90(t, 3H, J=6.8 Hz, CH3), 1.25—1.44(m, 6H, CH2CH2CH2), 1.64(t, 2H, J=7.2 Hz, CH2), 7.28, 7.32, 8.03, 8.06, 8.22, 8.28(d, J=8.4, 8.4, 8.0, 8.4, 8.0, 8.4 Hz, 2H of each, 12Ph-H), 13.02(s, 1H, COOH) |
| Entry | Compd. | λmax/nm | f(cis)t(%) | Time of isomerization/min | ||||
|---|---|---|---|---|---|---|---|---|
| Trans isomer | Cis isomer | |||||||
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | Solution | Mesophase | |||
| 1 | 3a | 233 | 347 | 235 | 354 | 11 | 40 | 50 |
| 3b | 223 | 330 | 223 | 327 | 55 | 40 | 70 | |
| 3c | 230 | 334 | 229 | 335 | 38 | 50 | 80 | |
| 2 | 3d | 238 | 340 | 238 | 334 | 37 | 70 | 90 |
| 3e | 239 | 337 | 240 | 334 | 50 | 70 | 90 | |
| 3f | 239 | 339 | 239 | 338 | 37 | 60 | 80 | |
| 3g | 238 | 336 | 239 | 333 | 29 | 60 | 70 | |
| 3h | 239 | 338 | 238 | 333 | 16 | 50 | 70 | |
Table 3 UV spectrum of target compounds and the maximum time of isomerization
| Entry | Compd. | λmax/nm | f(cis)t(%) | Time of isomerization/min | ||||
|---|---|---|---|---|---|---|---|---|
| Trans isomer | Cis isomer | |||||||
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | Solution | Mesophase | |||
| 1 | 3a | 233 | 347 | 235 | 354 | 11 | 40 | 50 |
| 3b | 223 | 330 | 223 | 327 | 55 | 40 | 70 | |
| 3c | 230 | 334 | 229 | 335 | 38 | 50 | 80 | |
| 2 | 3d | 238 | 340 | 238 | 334 | 37 | 70 | 90 |
| 3e | 239 | 337 | 240 | 334 | 50 | 70 | 90 | |
| 3f | 239 | 339 | 239 | 338 | 37 | 60 | 80 | |
| 3g | 238 | 336 | 239 | 333 | 29 | 60 | 70 | |
| 3h | 239 | 338 | 238 | 333 | 16 | 50 | 70 | |
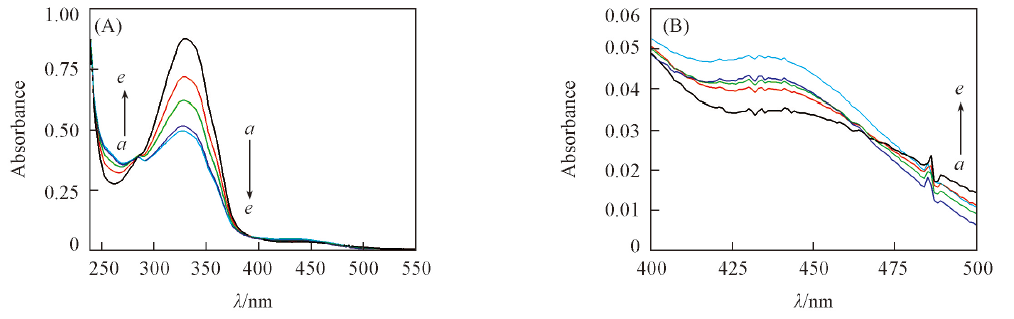
Fig.1 UV-Vis absorption spectra of compound 3b in methanol during trans-to-cis isomerization Fig.1(B) illustrates the small increase of absorption in 430 nm. Time/min: a. 0; b. 10; c. 20; d. 30; e. 40.
| Entry | Compd. | Eox/eV | Φp/eV | Ered/eV | Φn/eV | EHOMO/eV | ELUMO/eV | Eg/eV | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | |||||||
| 1 | 3a | 0.69 | 0.66 | 0.66 | -0.49 | -0.62 | -0.49 | -5.06 | -3.91 | 1.15 |
| 3b | 0.76 | 0.76 | -0.42 | -0.60 | -0.42 | -5.16 | -3.98 | 1.18 | ||
| 3c | 0.76 | 0.63 | 0.63 | -0.53 | -0.64 | -0.53 | -5.03 | -3.87 | 1.16 | |
| 2 | 3d | 0.82 | 0.65 | 0.65 | -0.47 | -0.56 | -0.47 | -5.05 | -3.93 | 1.12 |
| 3e | 0.88 | 0.88 | -0.51 | -0.65 | -0.51 | -5.28 | -3.89 | 1.39 | ||
| 3f | 0.82 | 0.82 | -0.45 | -0.54 | -0.45 | -5.22 | -3.95 | 1.27 | ||
| 3g | 0.83 | 0.83 | -0.54 | -0.64 | -0.54 | -5.23 | -3.86 | 1.37 | ||
| 3h | 0.81 | 0.58 | 0.58 | -0.50 | -0.62 | -0.50 | -4.98 | -3.90 | 1.08 | |
Table 4 Redox potentials, EHOMO, ELUMO and energy gaps of target compounds
| Entry | Compd. | Eox/eV | Φp/eV | Ered/eV | Φn/eV | EHOMO/eV | ELUMO/eV | Eg/eV | ||
|---|---|---|---|---|---|---|---|---|---|---|
| Ⅰ | Ⅱ | Ⅰ | Ⅱ | |||||||
| 1 | 3a | 0.69 | 0.66 | 0.66 | -0.49 | -0.62 | -0.49 | -5.06 | -3.91 | 1.15 |
| 3b | 0.76 | 0.76 | -0.42 | -0.60 | -0.42 | -5.16 | -3.98 | 1.18 | ||
| 3c | 0.76 | 0.63 | 0.63 | -0.53 | -0.64 | -0.53 | -5.03 | -3.87 | 1.16 | |
| 2 | 3d | 0.82 | 0.65 | 0.65 | -0.47 | -0.56 | -0.47 | -5.05 | -3.93 | 1.12 |
| 3e | 0.88 | 0.88 | -0.51 | -0.65 | -0.51 | -5.28 | -3.89 | 1.39 | ||
| 3f | 0.82 | 0.82 | -0.45 | -0.54 | -0.45 | -5.22 | -3.95 | 1.27 | ||
| 3g | 0.83 | 0.83 | -0.54 | -0.64 | -0.54 | -5.23 | -3.86 | 1.37 | ||
| 3h | 0.81 | 0.58 | 0.58 | -0.50 | -0.62 | -0.50 | -4.98 | -3.90 | 1.08 | |
| Entry | Compd. | L/Wa | m.p./℃ | ΔH of m.p./(J·g-1) | d.p.b/℃ |
|---|---|---|---|---|---|
| 1 | 3a | 4.362 | 211.2 | 28.72 | 218—369 |
| 3b | 4.638 | 221.0 | 26.54 | 232—419 | |
| 3c | 4.967 | 208.2 | 23.17 | 226—425 | |
| 2 | 3d | 4.110 | 198.7 | 25.62 | 257—413 |
| 3e | 4.447 | 200.3 | 26.76 | 246—388 | |
| 3f | 4.497 | 199.1 | 21.82 | 230—429 | |
| 3g | 4.549 | 186.8 | 40.39 | 240—393 | |
| 3h | 4.722 | 187.9 | 27.67 | 230—349 |
Table 5 DSC measurement results of target compounds
| Entry | Compd. | L/Wa | m.p./℃ | ΔH of m.p./(J·g-1) | d.p.b/℃ |
|---|---|---|---|---|---|
| 1 | 3a | 4.362 | 211.2 | 28.72 | 218—369 |
| 3b | 4.638 | 221.0 | 26.54 | 232—419 | |
| 3c | 4.967 | 208.2 | 23.17 | 226—425 | |
| 2 | 3d | 4.110 | 198.7 | 25.62 | 257—413 |
| 3e | 4.447 | 200.3 | 26.76 | 246—388 | |
| 3f | 4.497 | 199.1 | 21.82 | 230—429 | |
| 3g | 4.549 | 186.8 | 40.39 | 240—393 | |
| 3h | 4.722 | 187.9 | 27.67 | 230—349 |

Fig.2 POM images of compounds 3a—3h during heating process(A)—(H) Schlieren textures of 3a, 3b, 3c, 3d, 3e, 3f, 3g and 3h taken at 214, 227, 228, 221, 220, 210, 220 and 220 ℃, respectively.
| [1] | 陈然, 安忠维, 陈新兵, 陈沛. 高等学校化学学报, 2014, 35( 7), 1433- 1438 |
| Chen, R. , An Z., W. , Chen X., B. , Chen, P. , Chem. J. Chinese Universities, 2014, 35( 7), 1433- 1438 | |
| [2] | Ikeda, T. , Tsutsumi, O. , Tsutsumi, O. , Science, 1995, 268( 30), 1873- 1875 |
| [3] | Eich, M. , Wendorff J., H. , Makromol. Chem. Rapid Commun., 1987, 8( 10), 467- 471 |
| [4] | Zhou D., W. , Liang L., Y. , Lu M., G. , Polym. Bull., 2011, 66( 8), 1111- 1123 |
| [5] | Wu, L.F. , Tuo X., L. , Cheng, H. , Chen, Z. , Wang X., G. , Macromolecules, 2001, 34( 23), 8005- 8013 |
| [6] | Krohn, K. , John, M. , Demikhov E., I. , Russian Chemical Bulletin(International Edition ), 2001, 50( 7), 1248- 1254 |
| [7] | Luo Y., N. , Xu X., Z. , Sun F., X. , Yu X., Y. , Zhang, X. , Zhang, T. , Yu L., Y. , Chem. Res. Chinese Universities, 2014, 30( 1), 27- 31 |
| [8] | Lee H., K. , Doi, K. , Harada, H. , Tsutsumi, O. , Kanazawa, A. , Shiono, T. , Ikeda, T. , J. Phys. Chem. B, 2000, 104( 30), 7023- 7028 |
| [9] | Tong, X. , Zhao, Y. , Chem. Mater., 2009, 21( 17), 4047- 4054 |
| [10] | Rau H. , Photochemistry and Photophysics Vol. Ⅱ, CRC Press, Boca Raton, FL, 1990, 119— 141 |
| [11] | Kang X., L. , Zhao J., R. , Li H., Y. , He S., J. , Colloid Polym. Sci., 2013, 291( 9), 2245- 2251 |
| [12] | Yao J., L. , You Y., J. , Liu, H. , Dong L., J. , Xiong C., X. , J. Mater. Sci., 2011, 46( 10), 3343- 3348 |
| [13] | Circu, V. , Mocanu A., S. , Rosu, C. , Manaila-Maximean, D. , Dumitrascu, F. , J. Therm. Anal. Calorim., 2012, 107( 3), 877- 886 |
| [14] | Kaš, par M. , Novotná, V. , Hamplová, V. , Podoliak, N. , Nonnenmacher, D. , Glogarová, F. , Giesselmann, M. , Liq. Cryst., 2011, 38( 5), 309- 315 |
| [15] | 郑敏燕, 魏永生, 安忠维. 有机化学, 2008, 28( 12), 1948- 1953 |
| Zheng M., Y. , Wei Y., S. , An Z., W. , Chinese Journal of Organic Chemistry, 2008, 28( 12), 1948- 1953 | |
| [16] | Luo Y., N. , Xu X., Z. , Zhang, X. , Yu X., Y. , Qu X., S. , Yang Y., Y. , Shen Q., H. , Chem. Res. Chinese Universities, 2013, 29( 6), 1045- 1049 |
| [17] | Kreger, K. , Wolfer, P. , Audorff, H. , Kador, L. , Stingelin-Stutzmann, N. , Smith, P. , Schmidt H., W. , J. Am. Chem. Soc., 2010, 132( 2), 509- 516 |
| [18] | Sevigny, S. , Bouchard, L. , Motallebi, S. , Zhao, Y. , Macromolecules, 2003, 36( 24), 9033- 9041 |
| [19] | Leclair, S. , Mathew, L. , Giguere, M. , Motallebi, S. , Zhao, Y. , Macromolecules, 2003, 36( 24), 9024- 9032 |
| [20] | Hunter C., A. , Sarson L., D. , Tetrahedron Lett., 1996, 37( 5), 699- 702 |
| [21] | Cevasco, G. , Thea, S. , J. Org. Chem., 1999, 64( 15), 5422- 5429 |
| [22] | 樊冬丽, 翟岩, 张妍, 涂伟, 黄耀东. 高等学校化学学报, 2014, 35( 11), 2447- 2454 |
| Fan D., L. , Zhai, Y. , Zhang, Y. , Tu, W. , Huang Y., D. , Chem. J. Chinese Universities, 2014, 35( 11), 2447- 2454 | |
| [23] | Wang W., Y. , Wang M., Z. , Polymer Bulletin, 2007, 59( 4), 537- 544 |
| [24] | Ya, Q. , Dong X., Z. , Chen W., Q. , Duan X., M. , Dyes and Pigments, 2008, 79( 2), 159- 165 |
| [25] | Arias, J. , Bardaj, M. , Espinet, P. , Folcia C., L. , Ortega, J. , Etxebarria, J. Inorg. Chem., 2009, 48( 13), 6205- 6210 |
| [26] | 向能军, 李狄豪, 梁万里, 苏树江, 石建新, 龚孟濂. 化学学报, 2006, 64( 11), 1157- 1162 |
| Xiang N., J. , Lee T., H. , Leung L., M. , So S., K. , Shi J., X. , Gong M., L. , Acta Chimica Sinica, 2006, 64( 11), 1157- 1162 | |
| [27] | Gao, L. , Zhang, J. , He, C. , Zhang, Y. , Sun Q., J. , Li Y., F. , Science China, 2014, 57( 7), 966- 972 |
| [28] | 金长清, 苏树江, 梁万里, 马凤英, 王立军, 刘云, 张春玉. 发光学报, 2004, 25( 5), 541- 545 |
| Jin C., Q. , So S., K. , Leung L., M. , Ma F., Y. , Wang L., J. , Liu, Y. , Zhang C., Y. , Chinese Journal of Luminescence, 2004, 25( 5), 541- 545 | |
| [29] | Karim M., R. , Yahya, R. , Sheikh M. R., K. , Salleh N., M. , Hassan, A. , Ekramul M. H. N., M. , Journal of Polymer Research, 2014, 21( 6), 487- 496 |
| [1] | 谢璠, 陈珊珊, 卓龙海, 陆赵情, 高坤, 代啓阳. 聚对二甲苯纳米纤维阵列的CVD液晶模板法制备及降解性能[J]. 高等学校化学学报, 2021, 42(8): 2643. |
| [2] | 樊晔, 李倩, 方云, 夏咏梅. 用共轭亚油酸构筑层状液晶的药物传递系统[J]. 高等学校化学学报, 2020, 41(4): 750. |
| [3] | 魏福志, 冯微, 夏艳, 李东风, 侯瑞斌. 基于四硫富瓦烯和氰基联苯单元的玻璃态液晶化合物的合成及介晶性能[J]. 高等学校化学学报, 2018, 39(7): 1549. |
| [4] | 王毅, 张苹, 吴生秀, 孙媛媛, 赵彤, 刘世熙. 质谱中反偶电子规则并含双电荷离子的两类联苯化合物的裂解机理[J]. 高等学校化学学报, 2018, 39(3): 435. |
| [5] | 夏萌, 彭雄伟, 高红飞, 严超, 陈慧茹, 程晓红. 巴比妥酸楔形棒状液晶分子的合成、性质及与三嗪衍生物的氢键识别组装体[J]. 高等学校化学学报, 2017, 38(7): 1203. |
| [6] | 郭妙才, 和亚宁, 王晓工. 各向异性光交联作用对液晶在起伏光栅表面的取向影响[J]. 高等学校化学学报, 2015, 36(4): 751. |
| [7] | 杨光, 王传明, 唐萍, 顾松园. 对称柔性-半刚性-柔性三嵌段共聚物自组装行为[J]. 高等学校化学学报, 2014, 35(8): 1799. |
| [8] | 范玉霞, 谭慧, 留玲微, 吴朝阳, 沈国励, 俞汝勤. 新型超支状液晶核酸传感器用于p53突变基因的检测[J]. 高等学校化学学报, 2013, 34(4): 806. |
| [9] | 李广, 陈龙聪, 陈萌梦, 高斌, 熊兴良. 基于金纳米颗粒生长的液晶生物传感器检测酪氨酸[J]. 高等学校化学学报, 2013, 34(11): 2493. |
| [10] | 罗丙红, 李建华, 杨静, 徐宠恩, 田金环, 周长忍. 静电纺丝法制备胆固醇-g-聚乳酸液晶/ 聚乳酸复合纳米纤维膜[J]. 高等学校化学学报, 2012, 33(06): 1360. |
| [11] | 彭海炎, 毕曙光, 廖永贵, 周兴平, 解孝林. KCD/MDEA/TA三元引发剂引发丙烯酸酯/液晶复合体系光聚合动力学[J]. 高等学校化学学报, 2012, 33(03): 640. |
| [12] | 史小靖, 彭海炎, 廖永贵, 解孝林. 丙烯酸酯/液晶复合体系的光聚合动力学[J]. 高等学校化学学报, 2011, 32(6): 1407. |
| [13] | 杨胜园, 谭慧, 刘艳梅, 吴朝阳, 沈国励, 俞汝勤. 非标记液晶型免疫传感器检测赭曲霉素A[J]. 高等学校化学学报, 2011, 32(5): 1065. |
| [14] | 钱鹰, 林国强, 周志强, 崔一平. 吡啶星型分子的双光子上转换荧光特性[J]. 高等学校化学学报, 2011, 32(4): 868. |
| [15] | 唐敏健, 丁珊, 闵翔, 焦延鹏, 周长忍, 李红. 胶原液晶膜的制备及对细胞生长的影响[J]. 高等学校化学学报, 2011, 32(12): 2891. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||