

高等学校化学学报 ›› 2014, Vol. 35 ›› Issue (7): 1433.doi: 10.7503/cjcu20131233
收稿日期:2013-12-16
出版日期:2014-07-10
发布日期:2014-06-10
作者简介:联系人简介: 安忠维, 男, 博士, 教授, 博士生导师, 主要从事液晶材料研究. E-mail: 基金资助:
CHEN Ran1, AN Zhongwei1,2,*( ), CHEN Xinbing1, CHEN Pei1
), CHEN Xinbing1, CHEN Pei1
Received:2013-12-16
Online:2014-07-10
Published:2014-06-10
Contact:
AN Zhongwei
E-mail:gmecazw@163.com
Supported by:摘要:
以4-烷基反式环己基甲酸为原料, 经格氏、 黄鸣龙、 碘代及“一锅法”Sonogashira偶联等反应, 制备了10个含乙撑桥键二苯炔四环液晶化合物, 其结构经IR, NMR和MS确认. 利用差示扫描量热仪(DSC)、 偏光显微镜(POM)和阿贝折光仪对目标化合物进行了性能测试. 结果表明, 目标化合物具有高清亮点(220~240 ℃)、 宽向列相区间(59.6~99.6 ℃)和大光学各向异性(Δn>0.3), 其性能可满足液晶光学的应用要求.
中图分类号:
TrendMD:
陈然, 安忠维, 陈新兵, 陈沛. 含乙撑桥键二苯炔四环液晶的合成及性能. 高等学校化学学报, 2014, 35(7): 1433.
CHEN Ran, AN Zhongwei, CHEN Xinbing, CHEN Pei. Syntheses and Properties of Four-ring Liquid Crystals with Ethylene and Internal Alkyne Bridge†. Chem. J. Chinese Universities, 2014, 35(7): 1433.
| Compd. | m. p./℃ | Yield(%) | Appearance | MS, m/z(RI, %) |
|---|---|---|---|---|
| 2-F | 82.7—83.3 | 65 | Light yellow solid | 298(M+, 9), 283(100), 155(28), 115(11), 55(8) |
| 3-F | 87.1—88.2 | 70 | Light yellow solid | 312(M+, 13), 297(100), 159(15) |
| 4-F | 81.4—82.7 | 68 | Light yellow solid | 326(M+, 10), 311(100), 159(24), 155(43), 115(15) |
| 5-F | 79.3—79.8 | 72 | Light yellow solid | 340(M+, 11), 325(100), 155(40), 115(12), 55(7) |
| 2-2 | 159.3—161.5 | 45 | White solid | 454(M+, 100), 329(66), 204(21) |
| 3-2 | 150.1—152.3 | 43 | White solid | 468(M+, 100), 343(27), 329(32), 204(17) |
| 4-2 | 133.8—136.1 | 45 | White solid | 482(M+, 100), 357(19), 329(29), 204(16) |
| 5-2 | 127.5—130.0 | 36 | White solid | 496(M+, 100), 371(9), 357(9), 343(13), 329(16), 204(14) |
| 3-3 | 139.1—141.7 | 40 | White solid | 482(M+, 65), 371(30), 343(100), 204(41) |
| 4-3 | 150.8—153.4 | 40 | White solid | 496(M+, 100), 357(21), 343(29), 204(14) |
| 5-3 | 139.5—142.1 | 40 | White solid | 510(M+, 100), 371(14), 343(27), 204(13) |
| 4-4 | 154.6—156.8 | 40 | White solid | 510(M+, 100), 357(50), 204(24) |
| 5-4 | 143.7—146.1 | 38 | White solid | 524(M+, 100), 371(23), 357(22), 204(17) |
| 5-5 | 143.2—145.6 | 43 | White solid | 538(M+, 100), 371(29), 204(11) |
Table 1 Physical data and MS results of new compounds
| Compd. | m. p./℃ | Yield(%) | Appearance | MS, m/z(RI, %) |
|---|---|---|---|---|
| 2-F | 82.7—83.3 | 65 | Light yellow solid | 298(M+, 9), 283(100), 155(28), 115(11), 55(8) |
| 3-F | 87.1—88.2 | 70 | Light yellow solid | 312(M+, 13), 297(100), 159(15) |
| 4-F | 81.4—82.7 | 68 | Light yellow solid | 326(M+, 10), 311(100), 159(24), 155(43), 115(15) |
| 5-F | 79.3—79.8 | 72 | Light yellow solid | 340(M+, 11), 325(100), 155(40), 115(12), 55(7) |
| 2-2 | 159.3—161.5 | 45 | White solid | 454(M+, 100), 329(66), 204(21) |
| 3-2 | 150.1—152.3 | 43 | White solid | 468(M+, 100), 343(27), 329(32), 204(17) |
| 4-2 | 133.8—136.1 | 45 | White solid | 482(M+, 100), 357(19), 329(29), 204(16) |
| 5-2 | 127.5—130.0 | 36 | White solid | 496(M+, 100), 371(9), 357(9), 343(13), 329(16), 204(14) |
| 3-3 | 139.1—141.7 | 40 | White solid | 482(M+, 65), 371(30), 343(100), 204(41) |
| 4-3 | 150.8—153.4 | 40 | White solid | 496(M+, 100), 357(21), 343(29), 204(14) |
| 5-3 | 139.5—142.1 | 40 | White solid | 510(M+, 100), 371(14), 343(27), 204(13) |
| 4-4 | 154.6—156.8 | 40 | White solid | 510(M+, 100), 357(50), 204(24) |
| 5-4 | 143.7—146.1 | 38 | White solid | 524(M+, 100), 371(23), 357(22), 204(17) |
| 5-5 | 143.2—145.6 | 43 | White solid | 538(M+, 100), 371(29), 204(11) |
| Compd. | 1H NMR(CDCl3, 300 MHz), δ | 13C NMR(CDCl3, 75 MHz), δ |
|---|---|---|
| 2-F | 0.81—0.93(m, 7H), 1.11—1.27(m, 4H), 1.43—1.50(m, 2H), 1.60(s, 6H), 1.70—1.80(m, 4H), 2.51—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 11.5, 30.0, 31.6, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.0, 39.6, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 3-F | 0.84—0.98(m, 7H), 1.10—1.18(m, 4H), 1.25—1.34(m, 2H), 1.42—1.48(m, 2H), 1.60(s, 6H), 1.69—1.78(m, 4H), 2.54—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.0 Hz, 2H) | 14.4, 20.0, 31.6, 33.3, 33.3, 33.3, 37.5, 37.5, 39.1, 39.8, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 4-F | 0.80—0.96(m, 7H), 1.09—1.19(m, 4H), 1.23—1.32(m, 4H), 1.42—1.50(m, 2H), 1.60(s, 6H), 1.69—1.80(m, 4H), 2.53—2.62(m, 2H), 7.07(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 14.2, 23.2, 29.3, 31.6, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 65.6, 82.3, 93.3, 119.9, 128.3, 131.6, 143.6 |
| 5-F | 0.82—0.99(m, 7H), 1.07—1.20(m, 4H), 1.21—1.32(m, 6H), 1.45—1.54(m, 2H), 1.61(s, 6H), 1.69—1.79(m, 4H), 2.53—2.65(m, 2H), 7.10(d, J=8.0 Hz, 2H), 7.32(d, J=8.0 Hz, 2H) | 14.1, 22.7, 26.7, 31.6, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.8, 39.0, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 2-2 | 0.74—0.85(m, 14H), 1.07—1.17(m, 8H), 1.38—1.44(m, 4H), 1.65—1.73(m, 8H), 2.51—2.57(m, 4H), 7.05(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 11.5, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-2 | 0.76—0.93(m, 14H), 1.03—1.10(m, 4H), 1.12—1.21(m, 4H), 1.21—1.28(m, 2H), 1.38—1.46(m, 4H), 1.62—1.75(m, 8H), 2.49—2.60(m, 4H), 7.06(d, J=7.9 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 11.5, 14.4, 20.0, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.6, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-2 | 0.84—0.99(m, 14H), 1.12—1.21(m, 6H), 1.21—1.24(m, 2H), 1.25—1.31(m, 4H), 1.45—1.53(m, 4H), 1.69—1.83(m, 8H), 2.55—2.68(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.41(d, J=8.0 Hz, 4H) | 11.5, 14.1, 23.0, 29.3, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-2 | 0.83—0.96(m, 14H), 1.12—1.22(m, 8H), 1.23—1.30(m, 6H), 1.46—1.54(m, 4H), 1.70—1.83(m, 8H), 2.57—2.72(m, 4H), 7.13(d, J=7.4 Hz, 4H), 7.42(d, J=7.5 Hz, 4H) | 11.5, 14.1, 22.7, 26.7, 30.0, 32.3, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-3 | 0.70—0.92(m, 14H), 1.01—1.16(m, 8H), 1.18—1.30(m, 4H), 1.36—1.47(m, 4H), 1.55—1.79(m, 8H), 2.44—2.65(m, 4H), 7.05(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.4, 20.0, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-3 | 0.72—0.93(m, 14H), 0.98—1.14(m, 8H), 1.16—1.29(m, 6H), 1.36—1.47(m, 4H), 1.59—1.83(m, 8H), 2.43—2.64(m, 4H), 7.06(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.1, 14.4, 20.0, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-3 | 0.77—0.94(m, 14H), 1.05—1.11(m, 8H), 1.16—1.23(m, 8H), 1.38—1.44(m, 4H), 1.63—1.72(m, 8H), 2.50—2.57(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 14.1, 14.4, 20.0, 22.7, 26.7, 32.3, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-4 | 0.84—0.95(m, 14H), 0.98—1.13(m, 8H), 1.15—1.28(m, 8H), 1.37—1.46(m, 4H), 1.58—1.79(m, 8H), 2.36—2.69(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.8 Hz, 4H) | 14.1, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-4 | 0.74—0.93(m, 14H), 1.02—1.12(m, 8H), 1.15—1.26(m, 10H), 1.35—1.47(m, 6H), 1.62—1.73(m, 8H), 2.48—2.60(m, 4H), 7.05(d, J=7.2 Hz, 4H), 7.34(d, J=7.0 Hz, 4H) | 14.1, 22.7, 23.0, 26.7, 29.3, 32.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-5 | 0.82—1.00(m, 14H), 1.10—1.19(m, 8H), 1.20—1.33(m, 12H), 1.45—1.53(m, 4H), 1.68—1.84(m, 8H), 2.52—2.71(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.42(d, J=8.0 Hz, 4H) | 14.1, 22.7, 26.7, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
Table 2 1H NMR and 13C NMR data of new compounds
| Compd. | 1H NMR(CDCl3, 300 MHz), δ | 13C NMR(CDCl3, 75 MHz), δ |
|---|---|---|
| 2-F | 0.81—0.93(m, 7H), 1.11—1.27(m, 4H), 1.43—1.50(m, 2H), 1.60(s, 6H), 1.70—1.80(m, 4H), 2.51—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 11.5, 30.0, 31.6, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.0, 39.6, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 3-F | 0.84—0.98(m, 7H), 1.10—1.18(m, 4H), 1.25—1.34(m, 2H), 1.42—1.48(m, 2H), 1.60(s, 6H), 1.69—1.78(m, 4H), 2.54—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.0 Hz, 2H) | 14.4, 20.0, 31.6, 33.3, 33.3, 33.3, 37.5, 37.5, 39.1, 39.8, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 4-F | 0.80—0.96(m, 7H), 1.09—1.19(m, 4H), 1.23—1.32(m, 4H), 1.42—1.50(m, 2H), 1.60(s, 6H), 1.69—1.80(m, 4H), 2.53—2.62(m, 2H), 7.07(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 14.2, 23.2, 29.3, 31.6, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 65.6, 82.3, 93.3, 119.9, 128.3, 131.6, 143.6 |
| 5-F | 0.82—0.99(m, 7H), 1.07—1.20(m, 4H), 1.21—1.32(m, 6H), 1.45—1.54(m, 2H), 1.61(s, 6H), 1.69—1.79(m, 4H), 2.53—2.65(m, 2H), 7.10(d, J=8.0 Hz, 2H), 7.32(d, J=8.0 Hz, 2H) | 14.1, 22.7, 26.7, 31.6, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.8, 39.0, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 2-2 | 0.74—0.85(m, 14H), 1.07—1.17(m, 8H), 1.38—1.44(m, 4H), 1.65—1.73(m, 8H), 2.51—2.57(m, 4H), 7.05(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 11.5, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-2 | 0.76—0.93(m, 14H), 1.03—1.10(m, 4H), 1.12—1.21(m, 4H), 1.21—1.28(m, 2H), 1.38—1.46(m, 4H), 1.62—1.75(m, 8H), 2.49—2.60(m, 4H), 7.06(d, J=7.9 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 11.5, 14.4, 20.0, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.6, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-2 | 0.84—0.99(m, 14H), 1.12—1.21(m, 6H), 1.21—1.24(m, 2H), 1.25—1.31(m, 4H), 1.45—1.53(m, 4H), 1.69—1.83(m, 8H), 2.55—2.68(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.41(d, J=8.0 Hz, 4H) | 11.5, 14.1, 23.0, 29.3, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-2 | 0.83—0.96(m, 14H), 1.12—1.22(m, 8H), 1.23—1.30(m, 6H), 1.46—1.54(m, 4H), 1.70—1.83(m, 8H), 2.57—2.72(m, 4H), 7.13(d, J=7.4 Hz, 4H), 7.42(d, J=7.5 Hz, 4H) | 11.5, 14.1, 22.7, 26.7, 30.0, 32.3, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-3 | 0.70—0.92(m, 14H), 1.01—1.16(m, 8H), 1.18—1.30(m, 4H), 1.36—1.47(m, 4H), 1.55—1.79(m, 8H), 2.44—2.65(m, 4H), 7.05(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.4, 20.0, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-3 | 0.72—0.93(m, 14H), 0.98—1.14(m, 8H), 1.16—1.29(m, 6H), 1.36—1.47(m, 4H), 1.59—1.83(m, 8H), 2.43—2.64(m, 4H), 7.06(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.1, 14.4, 20.0, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-3 | 0.77—0.94(m, 14H), 1.05—1.11(m, 8H), 1.16—1.23(m, 8H), 1.38—1.44(m, 4H), 1.63—1.72(m, 8H), 2.50—2.57(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 14.1, 14.4, 20.0, 22.7, 26.7, 32.3, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-4 | 0.84—0.95(m, 14H), 0.98—1.13(m, 8H), 1.15—1.28(m, 8H), 1.37—1.46(m, 4H), 1.58—1.79(m, 8H), 2.36—2.69(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.8 Hz, 4H) | 14.1, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-4 | 0.74—0.93(m, 14H), 1.02—1.12(m, 8H), 1.15—1.26(m, 10H), 1.35—1.47(m, 6H), 1.62—1.73(m, 8H), 2.48—2.60(m, 4H), 7.05(d, J=7.2 Hz, 4H), 7.34(d, J=7.0 Hz, 4H) | 14.1, 22.7, 23.0, 26.7, 29.3, 32.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-5 | 0.82—1.00(m, 14H), 1.10—1.19(m, 8H), 1.20—1.33(m, 12H), 1.45—1.53(m, 4H), 1.68—1.84(m, 8H), 2.52—2.71(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.42(d, J=8.0 Hz, 4H) | 14.1, 22.7, 26.7, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| n | m | Transition temperature /℃[Enthalpy change/(kJ·mol-1)] | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Heating processa | Cooling processb | ||||||||
| Cr | SA | N | I | I | N | SA | Cr | ||
| 2 | 2 | 69.9(10.7) | 160.8(23.2) | 220.4(4.0) | 219.9(-3.8) | 159.4(-20.5) | |||
| 3 | 2 | 50.7(1.9) | 151.4(20.1) | 233.5(6.2) | 232.4(-5.1) | 149.0(-15.9) | |||
| 4 | 2 | 73.1(2.6) | | 135.2(16.1) | 229.0(5.6) | 227.4(-5.3) | 132.3(-15.6) | ||
| 5 | 2 | 78.4(24.7) | 128.3(10.0) | 225.8(4.6) | 225.2(-4.4) | 127.2(-9.4) | |||
| 3 | 3 | 89.4(13.0) | 140.5(10.4) | 240.1(1.7) | 239.6(-2.1) | 138.4(-10.4) | |||
| 4 | 3 | 58.5(14.6) | 152.2(17.1) | 240.4(5.1) | 239.9(-4.6) | | 150.9(-16.5) | ||
| 5 | 3 | 88.0(24.0) | 140.2(12.0) | 239.3(4.4) | 238.8(-4.9) | 138.9(-10.1) | |||
| 4 | 4 | 66.8(17.5) | 155.4(6.6) | 235.7(3.9) | 235.3(-3.8) | 154.3(-16.1) | |||
| 5 | 4 | 79.5(22.1) | 144.9(0.6) | 146.4(7.9) | 234.8(4.5) | 234.3(-4.3) | 146.0(-4.2) | 144.3(-8.9) | 40.9(-18.5) |
| 5 | 5 | 117.6(32.6) | 144.5(8.9) | 156.8(5.1) | 234.0(5.0) | 233.8(-4.9) | 156.5(-4.9) | 141.8(-9.7) | 95.0(-29.5) |
Table 3 Phase transition temperature and enthalpy change of compounds n-m
| n | m | Transition temperature /℃[Enthalpy change/(kJ·mol-1)] | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Heating processa | Cooling processb | ||||||||
| Cr | SA | N | I | I | N | SA | Cr | ||
| 2 | 2 | 69.9(10.7) | 160.8(23.2) | 220.4(4.0) | 219.9(-3.8) | 159.4(-20.5) | |||
| 3 | 2 | 50.7(1.9) | 151.4(20.1) | 233.5(6.2) | 232.4(-5.1) | 149.0(-15.9) | |||
| 4 | 2 | 73.1(2.6) | | 135.2(16.1) | 229.0(5.6) | 227.4(-5.3) | 132.3(-15.6) | ||
| 5 | 2 | 78.4(24.7) | 128.3(10.0) | 225.8(4.6) | 225.2(-4.4) | 127.2(-9.4) | |||
| 3 | 3 | 89.4(13.0) | 140.5(10.4) | 240.1(1.7) | 239.6(-2.1) | 138.4(-10.4) | |||
| 4 | 3 | 58.5(14.6) | 152.2(17.1) | 240.4(5.1) | 239.9(-4.6) | | 150.9(-16.5) | ||
| 5 | 3 | 88.0(24.0) | 140.2(12.0) | 239.3(4.4) | 238.8(-4.9) | 138.9(-10.1) | |||
| 4 | 4 | 66.8(17.5) | 155.4(6.6) | 235.7(3.9) | 235.3(-3.8) | 154.3(-16.1) | |||
| 5 | 4 | 79.5(22.1) | 144.9(0.6) | 146.4(7.9) | 234.8(4.5) | 234.3(-4.3) | 146.0(-4.2) | 144.3(-8.9) | 40.9(-18.5) |
| 5 | 5 | 117.6(32.6) | 144.5(8.9) | 156.8(5.1) | 234.0(5.0) | 233.8(-4.9) | 156.5(-4.9) | 141.8(-9.7) | 95.0(-29.5) |
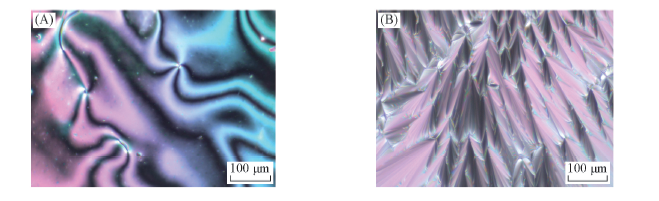
Fig.2 Polarizing optical micrographs of compound 5-5(200×) (A) A schlieren texture of the nematic phase at 226 ℃ on heating; (B) smectic A phase at 156 ℃ on cooling.
| Compd. | Structure | Transition temperature/℃ | ||||||
|---|---|---|---|---|---|---|---|---|
| Cr | SX1 | SX2 | SH | SB | N | I | ||
| 1 | 66 | 122 | 150.5 | 174 | 197.5 | 231 | ||
| 2[ | 76 | (63.5) | 148 | 190.5 | 215 | 228 | ||
| 3 | 185 | Above 300 | ||||||
| 4[ | 202 | Above 300 | ||||||
| 5 | 127 | | 155 | 210 | 220 | 325 | ||
| 6[ | 57 | | 62 | 226 | 248 | 312 | ||
| 3-3 | 140.5 | | 240.1 | |||||
| 5-3 | 140.2 | 239.3 | ||||||
Table 4 Phase transition temperatures of compounds 1—6, 3-3 and 5-3*
| Compd. | Structure | Transition temperature/℃ | ||||||
|---|---|---|---|---|---|---|---|---|
| Cr | SX1 | SX2 | SH | SB | N | I | ||
| 1 | 66 | 122 | 150.5 | 174 | 197.5 | 231 | ||
| 2[ | 76 | (63.5) | 148 | 190.5 | 215 | 228 | ||
| 3 | 185 | Above 300 | ||||||
| 4[ | 202 | Above 300 | ||||||
| 5 | 127 | | 155 | 210 | 220 | 325 | ||
| 6[ | 57 | | 62 | 226 | 248 | 312 | ||
| 3-3 | 140.5 | | 240.1 | |||||
| 5-3 | 140.2 | 239.3 | ||||||
| Compd. | 1 | 2-2 | 3-2 | 3-3 | 4-2 | 4-3 | 4-4 | 5-2 | 5-3 | 5-4 | 5-5 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Δn | 0.1998 | 0.3398 | 0.3358 | 0.3278 | 0.3278 | 0.3238 | 0.3198 | 0.3238 | 0.3198 | 0.3158 | 0.3118 |
Table 5 Optical anisotropies of compounds 1 and n-m
| Compd. | 1 | 2-2 | 3-2 | 3-3 | 4-2 | 4-3 | 4-4 | 5-2 | 5-3 | 5-4 | 5-5 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Δn | 0.1998 | 0.3398 | 0.3358 | 0.3278 | 0.3278 | 0.3238 | 0.3198 | 0.3238 | 0.3198 | 0.3158 | 0.3118 |
| [1] | Thomas G., Klaus G., Bernd R., Angew. Chem. Int. Ed., 2013, 52, 2—14 |
| [2] | Seung H. L., Surjya S. B., Heui S. J., Jeong K. U., J. Mater. Chem., 2012, 22(24), 11893—11903 |
| [3] | Kendhale A. M., Schenning A. P. H. J., Debije M. G., J. Mater. Chem. A, 2013, 1, 229—232 |
| [4] | Li L. W., Bryant D., Heugten T. V., Bos P. J., Opt. Express, 2013, 21(7), 8371—8381 |
| [5] | Zhao Y. L., Nan F. Y., Appl. Phys. Lett., 2013, 102(13), 131108-1—131108-5 |
| [6] | Reuter M., Garbat K., Vieweg N., Fischer B. M., Dᶏbrowski R., Koch M., Dziaduszek J., Urban S., J. Mater. Chem.C, 2013, 1(29), 4457—4463 |
| [7] | Buchnev O., Wallauer J., Walther M., Kaczmarek M., Zheludev N. I., Fedotov V. A., Appl. Phys. Lett., 2013, 103(14), 141904-1—141904-4 |
| [8] | Reuter M., Vieweg N., Fischer B. M., Mikulicz M., Koch M., Garbat K., Dᶏbrowski R., APL Mater., 2013, 1(1), 012107-1—012107-7 |
| [9] | Vieweg N., Jansen C., Koch M., Liquid Crystals and Their Applications in the THz Frequency Range, Springer Series in Optical Sciences, Berlin, 2013, 301—326 |
| [10] | Dziaduszek J., Kula P., Dᶏbrowski R., Drzewinski W., Garbat K., Urban S., Gauza S., Liq. Cryst., 2012, 39(2), 239—247 |
| [11] | Jiang Y., An Z. W., Chen P., Chen X. B., Zheng M. Y., Liq. Cryst., 2012, 39(4), 457—465 |
| [12] | Khoo I. C., Webster S., Kubo S., Youngblood W. J., Liou J. D., Mallouk T. E., Lin P., Hagan D. J., van Stryland E. W., J. Mater. Chem., 2009, 19(40), 7525—7531 |
| [13] | Fu Y. H., Chem. J. Chinese Universities, 2012, 33(7), 1490—1492 |
| (傅育红. 高等学校化学学报, 2012, 33(7), 1490—1492) | |
| [14] | Qiao W. Q., Chem. Res. Chinese Universities, 2012, 28(2), 239—241 |
| [15] | Chow H.F., Wan C. W., Low K. H., Yeung Y. Y., J. Org. Chem., 2001, 66, 1910—1913 |
| [16] | Cao X. Y., Zhang W., Zi H., Pei J., Org. Lett., 2004, 6(26), 4845—4848 |
| [17] | Zoltán N., Péter N., András K., Org. Lett., 2004, 6(26), 4917—4920 |
| [18] | Dietrich D., Lothar R., Textures of Liquid Crystal, Verlag Chemie, Weinheim, 1979, 170—212 |
| [19] | Hu M. G., An Z. W., Li J., Chin. J. Liq. Cryst. Displays, 2007, 22(2), 151—155 |
| (胡明刚, 安忠维, 李建. 液晶与显示, 2007, 22(2), 151—155) | |
| [20] | Dᶏbrowski R., Dziaduszek J., Czuprynski K., SPIE-The International Society for Optical Engineering, 1995, 2372, 235—241 |
| [21] | Takatsu H., Sasaki M., Tanaka Y., Sato H., Tolan-type Nematic Liquid Crystalline Compounds, GB 2155465A, 1985-09-25 |
| [22] | Chen X. B., An Z. W., Petrochemical Technology & Application, 2001, 19(6), 393—394 |
| (陈新兵, 安忠维. 石化技术与应用, 2001, 19(6), 393—394) | |
| [23] | Gauza S., Wang H. Y., Wen C. H.,Wu S. T., Alexander J. S., Dᶏbrowski R., Jpn. J. Appl. Phys., 2003, 42(6A), 3463—3466 |
| (Ed.: P, H, V, K) |
| [1] | 黄智宇, 陈宏辉, 马文乐, 黄毅, 朱丹, 陈永胜. 太赫兹隐身及屏蔽材料研究进展[J]. 高等学校化学学报, 2019, 40(6): 1103. |
| [2] | 李娟利, 彭增辉, 李建, 胡明刚, 安忠维, 张璐. 新型二氟乙烯端基异硫氰酸酯化合物的合成及性能[J]. 高等学校化学学报, 2017, 38(10): 1788. |
| [3] | 方虹霞, 张琪, 张慧丽, 秦丹, 洪治, 杜勇. 奥美沙坦与苯甲酸共晶的太赫兹光谱与密度泛函理论分析[J]. 高等学校化学学报, 2015, 36(12): 2504. |
| [4] | 于雷, 华小辉, 翟延君, 杨丽敏, 赵国忠, 孟田华, 翁诗甫, 徐怡庄, 刘克新, 吴瑾光, 陈佳洱. 两种半乳糖醇氯化钆配合物的制备与表征[J]. 高等学校化学学报, 2011, 32(6): 1244. |
| [5] | 杨丽敏,赵国忠,赵夔,石小溪,贾新锋,翁诗甫,徐怡庄,鲁向阳,谢大弢,吴瑾光,陈佳洱 . 胆酸和脱氧胆酸分子的远红外与THz吸收光谱研究[J]. 高等学校化学学报, 2008, 29(6): 1116. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||