

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (9): 2080.doi: 10.7503/cjcu20180019
• Polymer Chemistry • Previous Articles Next Articles
LIU Yina1, YANG Rongjie1,*( ), LI Dinghua1, LIANG Jiaxiang1, HU Weiguo2
), LI Dinghua1, LIANG Jiaxiang1, HU Weiguo2
Received:2018-01-08
Online:2018-09-07
Published:2018-08-03
Contact:
YANG Rongjie
E-mail:yrj@bit.edu.cn
Supported by:CLC Number:
TrendMD:
LIU Yina,YANG Rongjie,LI Dinghua,LIANG Jiaxiang,HU Weiguo. Characterization of High Polymerization Degree Ammonium Polyphosphate and Its Chain Structure by 31P NMR†[J]. Chem. J. Chinese Universities, 2018, 39(9): 2080.
| Preparation conditon | Dissolving temperature/℃ | Testing temperature/℃ | Pfree | Pend | Pmid | n | |||
|---|---|---|---|---|---|---|---|---|---|
| Sfree | Ratio(%) | Send | Ratio(%) | Smid | Ratio(%) | ||||
| NaCl auxiliary | 26 | 26 | 6.69 | 0.64 | 2.62 | 0.25 | 1034.62 | 99.11 | 792 |
| dissolving | 36 | 26 | 6.77 | 0.61 | 2.46 | 0.22 | 1099.07 | 99.17 | 893 |
| 36 | 36 | 6.92 | 0.66 | 2.71 | 0.26 | 1040.72 | 99.08 | 770 | |
| 100 ℃ High temperature dissolving | 100 | 26 | 7.21 | 7.20 | 3.83 | 3.82 | 989.34 | 98.89 | 519 |
Table 1 Characteristic phosphorous and polymerization degree(n) of APP1# at different testing conditions
| Preparation conditon | Dissolving temperature/℃ | Testing temperature/℃ | Pfree | Pend | Pmid | n | |||
|---|---|---|---|---|---|---|---|---|---|
| Sfree | Ratio(%) | Send | Ratio(%) | Smid | Ratio(%) | ||||
| NaCl auxiliary | 26 | 26 | 6.69 | 0.64 | 2.62 | 0.25 | 1034.62 | 99.11 | 792 |
| dissolving | 36 | 26 | 6.77 | 0.61 | 2.46 | 0.22 | 1099.07 | 99.17 | 893 |
| 36 | 36 | 6.92 | 0.66 | 2.71 | 0.26 | 1040.72 | 99.08 | 770 | |
| 100 ℃ High temperature dissolving | 100 | 26 | 7.21 | 7.20 | 3.83 | 3.82 | 989.34 | 98.89 | 519 |
| Preservation temperature/℃ | Polymerization degree, n | |||
|---|---|---|---|---|
| 2 h | 5 h | 8 h | 11 h | |
| 26 | 792 | 782 | 764 | 754 |
| 36 | 770 | 773 | 513 | 460 |
Table 2 Polymerization degree of APP1# solution(10 mg NaCl) experiencing preservation for different time
| Preservation temperature/℃ | Polymerization degree, n | |||
|---|---|---|---|---|
| 2 h | 5 h | 8 h | 11 h | |
| 26 | 792 | 782 | 764 | 754 |
| 36 | 770 | 773 | 513 | 460 |
| Testing number | n | Testing number | n |
|---|---|---|---|
| 1 | 893 | 6 | 838 |
| 2 | 883 | 7 | 857 |
| 3 | 878 | 8 | 865 |
| 4 | 866 | Average | 865 |
| 5 | 840 |
Table 3 Repeatability of polymerization degree(n) of APP1# in 31P NMR tests
| Testing number | n | Testing number | n |
|---|---|---|---|
| 1 | 893 | 6 | 838 |
| 2 | 883 | 7 | 857 |
| 3 | 878 | 8 | 865 |
| 4 | 866 | Average | 865 |
| 5 | 840 |
| APP sample | Sfree(%) | Send(%) | Smid(%) | n |
|---|---|---|---|---|
| 5125# | 0.52 | 0.17 | 98.64 | 1162 |
| 61231# | 0.10 | 0.06 | 95.74 | 3193 |
| 714# | 0.16 | 0.02 | 94.17 | 9419 |
| 715# | 0.08 | — | 102.6 | — |
Table 4 Characteristic phosphorus and polymerization degree of different APP samples with 10 mg NaCl auxiliary dissolving at 36 ℃
| APP sample | Sfree(%) | Send(%) | Smid(%) | n |
|---|---|---|---|---|
| 5125# | 0.52 | 0.17 | 98.64 | 1162 |
| 61231# | 0.10 | 0.06 | 95.74 | 3193 |
| 714# | 0.16 | 0.02 | 94.17 | 9419 |
| 715# | 0.08 | — | 102.6 | — |
| D1/s | Send | Smid | n |
|---|---|---|---|
| 1 | 1.68 | 957.85 | 1142 |
| 10 | 2.48 | 1058.56 | 855 |
| 20 | 2.54 | 1058.78 | 835 |
| 30 | 2.54 | 1058.77 | 835 |
Table 5 Characteristic phosphorus and polymerization degree of APP1# with 10 mg NaCl auxiliary dissolving at 36 ℃ at different relaxation time parameters
| D1/s | Send | Smid | n |
|---|---|---|---|
| 1 | 1.68 | 957.85 | 1142 |
| 10 | 2.48 | 1058.56 | 855 |
| 20 | 2.54 | 1058.78 | 835 |
| 30 | 2.54 | 1058.77 | 835 |
| Testing temperature/℃ | T1/s | ||
|---|---|---|---|
| Pfree | Pend | Pmid | |
| 26 | 8.32 | 2.99 | 0.52 |
| 31 | 8.52 | 4.54 | 0.55 |
| 36 | 8.89 | 5.60 | 0.59 |
| 41 | 10.70 | 6.81 | 0.63 |
| 46 | 10.68 | 7.99 | 0.66 |
| 51 | 10.21 | 8.97 | 0.69 |
| 56 | 11.20 | 10.10 | 0.73 |
Table 6 Relaxation time(T1) of phosphorus at different testing temperatures in 31P NMR for APP1# with 10 mg NaCl auxiliary dissolving at 36 ℃
| Testing temperature/℃ | T1/s | ||
|---|---|---|---|
| Pfree | Pend | Pmid | |
| 26 | 8.32 | 2.99 | 0.52 |
| 31 | 8.52 | 4.54 | 0.55 |
| 36 | 8.89 | 5.60 | 0.59 |
| 41 | 10.70 | 6.81 | 0.63 |
| 46 | 10.68 | 7.99 | 0.66 |
| 51 | 10.21 | 8.97 | 0.69 |
| 56 | 11.20 | 10.10 | 0.73 |
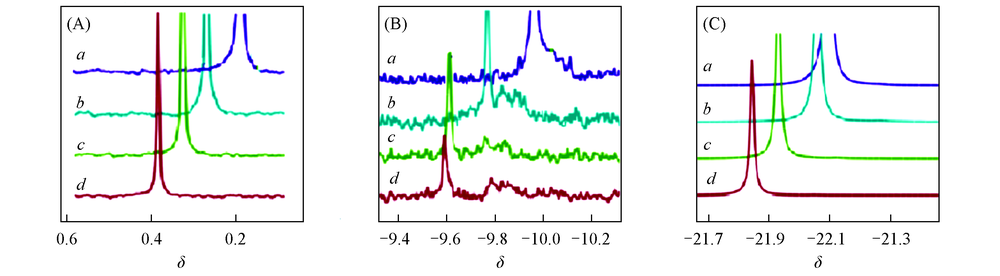
Fig.4 Phosphorous resonance peaks of APP1# with 10 mg NaCl auxiliary dissolving at 36 ℃ at different testing temperatures in 31P NMR (A) Pfree; (B) Pend; (C) Pmid. Temperature/℃: a. 26; b. 36; c. 46; d. 56.
| Acetic acid content(%) | Pfree ratio(%) | Pend ratio(%) | Short chain Pmid ratio(%) | Long chain Pmid ratio(%) | ||
|---|---|---|---|---|---|---|
| Peak 1 | Peak 2 | Peak 3 | ||||
| 0 | 1.18 | 0.14 | 0.14 | 0.16 | 0.21 | 98.2 |
| 30 | 1.40 | 0.13 | 0.22 | 0.20 | 0.21 | 97.8 |
| 50 | 1.41 | 0.12 | 0.32 | 0.37 | 0.27 | 97.5 |
Table 7 Characteristic phosphorus in APP1# solution with acetic acid added
| Acetic acid content(%) | Pfree ratio(%) | Pend ratio(%) | Short chain Pmid ratio(%) | Long chain Pmid ratio(%) | ||
|---|---|---|---|---|---|---|
| Peak 1 | Peak 2 | Peak 3 | ||||
| 0 | 1.18 | 0.14 | 0.14 | 0.16 | 0.21 | 98.2 |
| 30 | 1.40 | 0.13 | 0.22 | 0.20 | 0.21 | 97.8 |
| 50 | 1.41 | 0.12 | 0.32 | 0.37 | 0.27 | 97.5 |
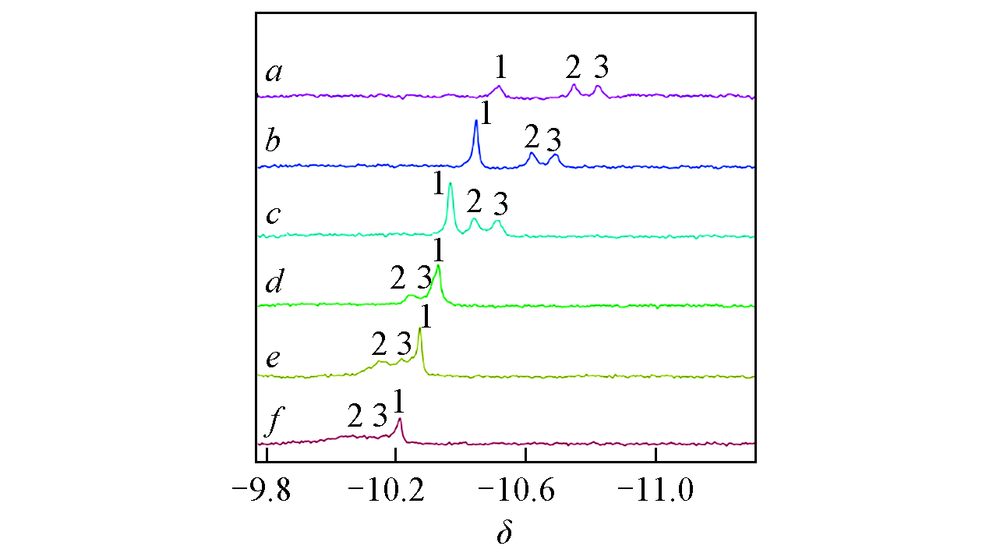
Fig.9 Pend characteristics of of APP1# solutions with different NaCl concentrationsNaCl concentration/(mg·mL-1): a. 0(100 ℃); b. 5; c. 10; d. 15; e. 20; f. 25.
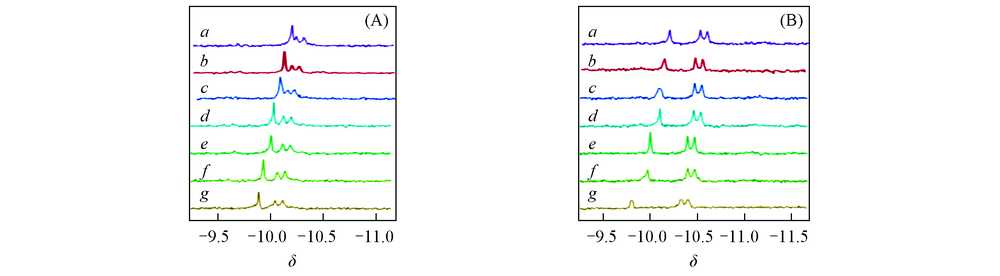
Fig.10 Pend characteristics of APP1# solutions at different testing temperatures (A) NaCl auxiliary dissolving at 36 ℃; (B) high temperature dissolving at 100 ℃. Testing temperature/℃: a. 26; b. 31; c. 36; d. 41; e. 46; f. 51; g. 56.
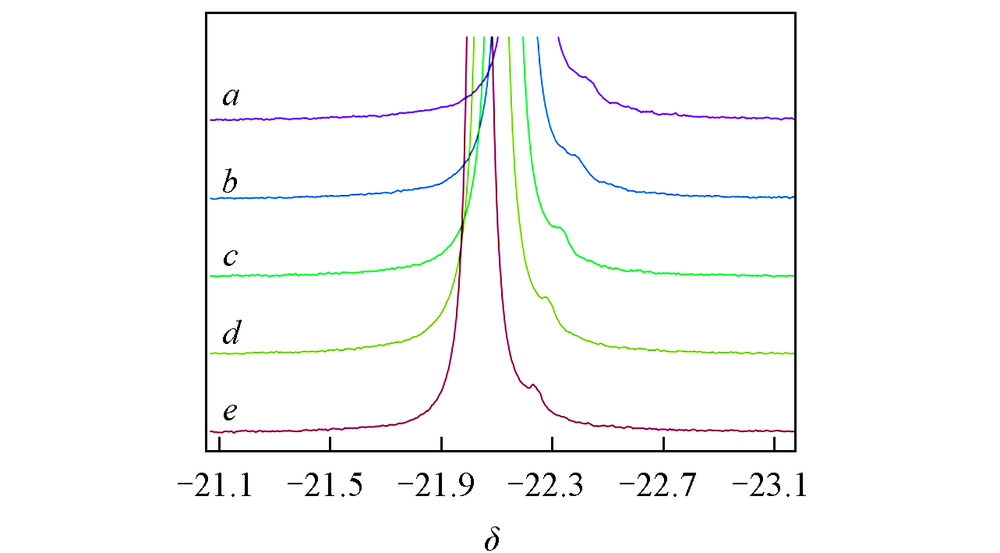
Fig.11 Resonance peaks at different testing temperature of middle phosphorus Pmid in APP1# solution with 10 mg NaCl auxiliary dissolving at 36 ℃Testing temperature/℃: a. 26; b. 31; c. 36; d. 41; e. 46.
| [1] | Camino G., Luda M. P., Fire Retardancy of Polymers: the Use of Intumescence, The Royal Society of Chemistry, Cambrige, 1988, 48—73 |
| [2] | Bbargava H. N., Varma B. K., Agrawal P. K., Colloid and Polymer Science, 1978, 256, 452—458 |
| [3] | Dong Y., Wang G. J., Su Q., Surface Coatings Technology, 2014, 246, 71—76 |
| [4] | Griffith E. J., J. Am. Chem. Soc., 1957, 79, 509—513 |
| [5] | Liu G. S., Duan X. Z., Advanced Materials Research, 2011, 832—835 |
| [6] | Yang R.J., Yi D. Q., Ammonium Polyphosphate, Science Press, Beijing, 2015 |
| (杨荣杰, 仪德启. 聚磷酸铵, 北京:科学出版社, 2015) | |
| [7] | Greenfield S., Oxford M.C., Analytical Chemistry of the Condensed Phosphates, Pergamon Press, New York, 1975, 18—32 |
| [8] | Van Wazer J. R., Callis C. F., Shoolery J. N., J. Am. Chem. Soc., 1955, 77(18), 4945—4946 |
| [9] | Van Wazer J. R., Phosphorous and Its Compounds, Interscience, New Yourk, 1958, 419—477 |
| [10] | Van Wazer J. R., J. Am. Chem. Soc., 1950, 72, 906—908 |
| [11] | Callis C. F., van Wazer J. R., Shoolery J. N., J. Am. Chem. Soc., 1957, 78, 2719—2726 |
| [12] | Callis C. F., van Wazer J. R., Arvan P. G., Chemical Reviews, 1954, 54, 777—796 |
| [13] | Macdonald J. C., Mazurek M., Journal of Magnetic Rreonance, 1987, 72, 48—60 |
| [14] | Glonek T., Costello J. R., Myers T. C., van Wazer J. R., The Journal of Physical Chemistry, 1975, 79(12), 1214—1218 |
| [15] | Wang Q. C., Yang Y. J., Inorganic Chemicals Industry, 2005, 32(12), 53—56 |
| (王清才, 杨荣杰. 无机盐工业, 2005, 32(12), 53—56) | |
| [16] | Greenwood N. N., Earnshaw A., Chemistry of the Elements, 2nd Ed., Butterworth-Heinermann, Oxford and Boston, 1997, 473—531 |
| [17] | Waerstad K. R., Mcclellan G., Food Chemistry, 1976, 24(2), 412—415 |
| [18] | HG/T 2770-2008, Chemical Industry Standard of the People's Republic of China, Chemical Industry Press, Beijing, 2008 |
| (HG/T 2770-2008, 中华人民共和国行业标准, 北京: 化学工业出版社, 2008) | |
| [19] | Wang Q. C., Studies on Characterization of the Molecular Weight and Crystal Structure of Ammonium Polyphosphate, Beijing Institute of Technology, Beijing, 2006 |
| (王清才. 聚磷酸铵(APP)分子链表征及结晶结构研究, 北京: 北京理工大学, 2006) | |
| [20] | Deng Y. P., Characterization on Polymerization Degree of Long Chain Ammonium Polyphosphate, Beijing Institute of Technology, Beijing, 2015 |
| (邓银萍. 长链聚磷酸铵的聚合度表征研究, 北京: 北京理工大学, 2015) | |
| [21] | Van Wazer J. R., Callis C. F., Shoolery J. N., Jones R. C., J. Am. Chem. Soc., 1956, 78(22), 5715—5726 |
| [22] | Gao H.B., Zhang Z. F., Nuclear Magnetic Resonance Principle and Experimental Method, Wuhan University Press,Wuhan, 2008 |
| (高汉宾, 张振芳. 核磁共振原理与实验方法, 武汉: 武汉大学出版社, 2008) | |
| [23] | Crutchfield M. M., Callis C. V., Irani R. R., Roth G. C., Inorganic Chemistry, 1962, 1(4), 813—817 |
| [1] | LIU Yina, YANG Rongjie, HU Weiguo, LI Dinghua. Analysis of Characteristic Phosphorus in High Polymerization Ammonium Polyphosphate by 31P NMR [J]. Chem. J. Chinese Universities, 2020, 41(12): 2832. |
| [2] | WANG Na,YANG Fei,ZHANG Jing,FANG Qinghong. Inflame-retardant Water-borne Epoxy Resin of APP Microsphere with Carrageenan Cladding† [J]. Chem. J. Chinese Universities, 2019, 40(2): 385. |
| [3] | NIU Qingtao,ZOU Chen,WANG Riguo,LI Lange,HE Aihua. Fractionation and Fraction Characterization of trans-Polyisoprene Rubber Alloys within Reactor† [J]. Chem. J. Chinese Universities, 2018, 39(10): 2312. |
| [4] | LIU Jianchao, XU Miaojun, LI Bin. Hydrophobic Modification of Ammonium Polyphosphate and Its Application in Flame Retardant Polypropylene Composites† [J]. Chem. J. Chinese Universities, 2015, 36(6): 1228. |
| [5] | ZHOU Li-Juan, ZHANG Xiu-Qin, LI Rong-Bo, SU Yun-Lan, ZHAO Ying, XU Duan-Fu. Effect of γ-Ray Irradiation on Chain Structure and Crystallization Behavior of Isotactic Polypropylene [J]. Chem. J. Chinese Universities, 2012, 33(04): 850. |
| [6] | CHEN Wen-Yan, LIU Xin-Chun, LIU Gou-Sheng, YU Jian-Guo. Synthesis and Characterization of Reticulated Structure Ammonium Polyphosphate with Crystal Form V [J]. Chem. J. Chinese Universities, 2010, 31(12): 2494. |
| [7] |
LIN Mei-Qin*, DONG Zhao-Xia, LI Ming-Yuan, WU Zhao-Liang.
27Al NMR Studies on HPAM/AlCit Crosslinking System with Low Concentration Polymer [J]. Chem. J. Chinese Universities, 2007, 28(8): 1573. |
| [8] | LI Yi-Jie1, 2, WEI Dong-Shan1, 2, JIN Xi-Gao1, Han Charles C.1, LIAO Qi1*. Molecular Dynamics Simulation of Confined Polymer Melts [J]. Chem. J. Chinese Universities, 2007, 28(5): 992. |
| [9] | LI Dong-Hua, XU Xiao-Ming, ZHOU Wen-Ting, HAN Guo-Bin. Kinetics of Micellization of Quaternary Ammonium Gemini Surfactants [J]. Chem. J. Chinese Universities, 2006, 27(10): 1927. |
| [10] | KONG Xu-Xin, XU Kun, WANG Pi-Xin, CHEN Qun. Solid State NMR Study of Starch-sodium Acrylate Grafted Copolymer [J]. Chem. J. Chinese Universities, 2005, 26(9): 1735. |
| [11] | GAO Hong-Chang, MAO Shi-Zhen, YUAN Han-Zhen, DU You-Ru . Nuclear Magnetic Resonance Relaxation of Cohesional Entanglement of Nylon 6 Chains in Trifluoroacetic Acid Solution [J]. Chem. J. Chinese Universities, 2004, 25(8): 1555. |
| [12] | ZHOU Xue-Qin, XU Jing, ZHAO Xiao-Dong, ZHANG Zhi-Hui, YAO Kang-De, LIU Dong-Zhi . Spectroscopic Evidence of Azone Effect on the Keratin in Mouse Stratum Corneum [J]. Chem. J. Chinese Universities, 2004, 25(7): 1273. |
| [13] | YU Ruo-Bing, ZHOU Chi-Xing, YU Wei . Relationship Between Rheology Behavior of TLCP/PEI Blend System and Its Multi-scales of Structure [J]. Chem. J. Chinese Universities, 2004, 25(11): 2140. |
| [14] | LI Chuan-Bi, ZHANG Li-Juan, SUN Ying-Hua, LI Yong, WANG Tie-Gang, XU Ji-Qing . Hydrothermal Synthesis and Structure of A Novel [Co(phen)(μ3-md)]n [J]. Chem. J. Chinese Universities, 2003, 24(8): 1366. |
| [15] | ZHANG Qiu Jin, CHEN Qun. Hydrogen-bonding Interaction of Ethylene Vinyl Alcohol Copolymers and Its Relation to the Chain Structures as Studied by VT 1H NMR Spectroscopy [J]. Chem. J. Chinese Universities, 2003, 24(4): 728. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||