

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (9): 1645.doi: 10.7503/cjcu20170054
• Physical Chemistry • Previous Articles Next Articles
YIN Jinchao, CHEN Yukai, JIANG Jianzhong*( ), CUI Zhenggang
), CUI Zhenggang
Received:2017-01-20
Online:2017-09-10
Published:2017-08-17
Contact:
JIANG Jianzhong
E-mail:jzjiang@jiangnan.edu.cn
Supported by:CLC Number:
TrendMD:
YIN Jinchao, CHEN Yukai, JIANG Jianzhong, CUI Zhenggang. Synthesis and Properties of pH and Redox Dual-switchable Surfactant†[J]. Chem. J. Chinese Universities, 2017, 38(9): 1645.
| Surfactant | cmc/(mol·L-1) | γcmc/(mN·m-1) | A/nm2 |
|---|---|---|---|
| C11-N-Fe | 1.25×10-3 | 47.93 | 0.6139 |
| C11-N-Fe-Ox | 2.5×10-3 | 54.62 | 0.7488 |
Table 1 Surface activity parameter of C11-N-Fe surfactant before and after oxidation in the air/water interface(25 ℃)
| Surfactant | cmc/(mol·L-1) | γcmc/(mN·m-1) | A/nm2 |
|---|---|---|---|
| C11-N-Fe | 1.25×10-3 | 47.93 | 0.6139 |
| C11-N-Fe-Ox | 2.5×10-3 | 54.62 | 0.7488 |
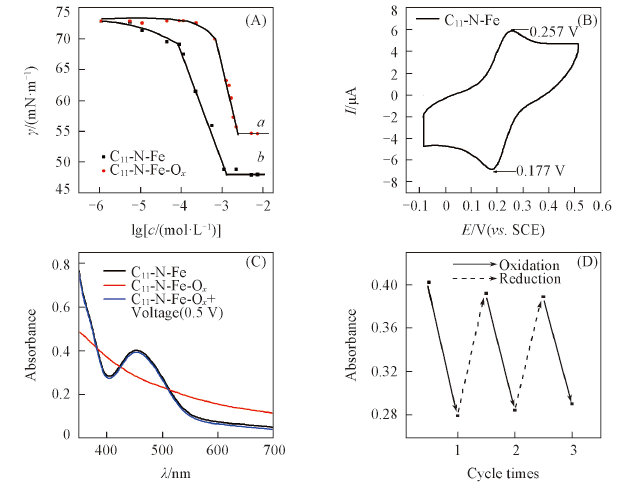
Fig.2 C11-N-Fe hydrochloride γ-lgc graph(25 ℃) before(a) and after oxidation(b)(A), cyclic voltammograms(100 mV/s)(B), UV-Vis spectra of C11-N-Fe ethanol solution(1×10-3 mol/L) before and after oxidation and reduction(C) and the change in absorbance at 455 nm while C11-N-Fe(1×10-3 mol/L) before and after oxidized by H2O2 and reduced by electrification

Fig.4 Foam volume of C11-N-Fe hydrochloride aqueous solution(7×10-3 mol/L)(A) alternate adding NaOH(B), HCl(C), oxidized by adding H2O2(D) and reduced by electrification(E)
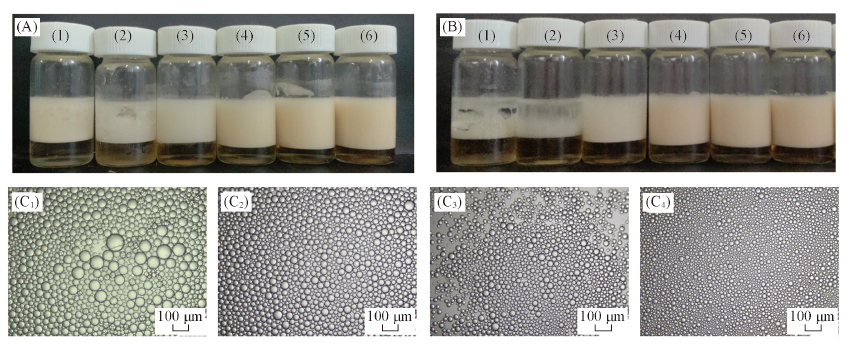
Fig.5 Photographs(A, B) and selected optical micrographs(C) of n-decane-in-water emulsions(volume ratio, 1∶1) stabilized by C11-N-Fe hydrochloride surfactant alone(A) Taken one day; (B, C) one month after preparation. The concentration of C11-N-Fe hydrochloride in the aqueous phase from(Ⅰ) to (Ⅵ) is 1×10-3, 1.3×10-3, 1.6×10-3, 2×10-3, 3×10-3, 4×10-3 mol/L respectively; (C) Concentration of C11-N-Fe hydrochloride in the aqueous/(mol·L-1): (C1) 1×10-3; (C2) 1.6×10-3; (C3) 2×10-3; (C4) 4×10-3.

Fig.6 Photographs(A) and corresponding optical micrographs(B—D) of n-decane-in-water emulsions stabilised by 1.6×10-3 mol/L C11-N-Fe hydrochloride alternate adding NaOH/HCl undergoing switching off/on cycle(B) Stable emulsion; (C) after adding NaOH; (D) afte adding HCl followed by homogenization for 2 min.

Fig.7 Photographs(A) and corresponding optical micrographs(B—D) of n-decane-in-water emulsions stabilized by 1.6×10-3 mol/L C11-N-Fe hydrochloride oxidation by H2O2 and reduction by electrification undergoing switching off/on cycle(B) Stable emulsion; (C) after oxidation by H2O2; (D) after reduction by electrified followed by homogenization for 2 min.
| [1] | Liu Y. X., Jessop P. G., Cunningham M., Eckert C. A., Liotta C. L., Science,2006, 313(5789), 958—960 |
| [2] | Brown P., Butts C. P., Eastoe J., Soft Matter, 2013, 9(8), 2365—2374 |
| [3] | Fameau A. L., Carl A., Saint-Jalmes A., Klitzing R., ChemPhysChem,2015, 16(1), 66—75 |
| [4] | Rahmani N. H. G., Dabros T., Masliyah J. H., Chem. Eng. Sci., 2004, 59(3), 685—697 |
| [5] | Zhang Y. M., Kong W. W., Wang C., An P. Y., Fang Y., Feng Y. J., Qin Z. R., Liu X. F., Soft Matter, 2015, 11(38), 7469—7473 |
| [6] | Ying G. G., Environ Int., 2006, 32(3), 417—431 |
| [7] | Li T., Zhang L., Chen Y., Guo Y. J., Du X. Y., Chem. J. Chinese Universities, 2015, 36(4), 772—780 |
| (李涛, 张龙, 陈颖, 郭亚军, 杜雪岩.高等学校化学学报,2015, 36(4), 772—780) | |
| [8] | Gao Y. J., Zhang L., Hou J. J., Ma Y. B., Qiu H., Zhang W. J., Du X. Y., Chem. J. Chinese Universities, 2016, 37(6), 1202—1207 |
| (郭亚军, 张龙, 后洁琼, 马泳波, 秋虎, 张文娟, 杜雪岩.高等学校化学学报,2016, 37(6), 1202—1207) | |
| [9] | Jiang J. Z., Zhu Y., Cui Z. G., Binks B. P., Angew. Chem. Int. Ed., 2013, 52, 12373—12376 |
| [10] | Johnsson M., Wagenaar A., Engberts J. B., J. Am. Chem. Soc., 2003, 125(3), 757—760 |
| [11] | Klijn J. E., Stuart M. C. A., Scarzello M., Wagenaar A., Engberts J. B. F. N., J. Phys. Chem. B, 2006, 110(43), 21694—21700 |
| [12] | Fukuda H., Goto A., Yoshioka H., Goto R., Morigaki K., Walde P., Langmuir,2001, 17(14), 4223—4231 |
| [13] | Liu K. H., Lin Q., Cui Z. G., Pei X. M., Jiang J. Z., Chem. J. Chinese Universities, 2017, 38(1), 85—93 |
| (刘凯鸿, 林琪, 崔正刚, 裴晓梅, 蒋建中.高等学校化学学报,2017, 38(1), 85—93) | |
| [14] | Eastoe J., Vesperinas A., Soft Matter, 2005, 1(5), 338—347 |
| [15] | Aathimanikandan S. V., Savariar E. N., Thayumanavan S., J. Am. Chem. Soc., 2005, 127(42), 14922—14929 |
| [16] | Raghavan S. R., Edlund H., Kaler E. W., Langmuir,2002, 18(4), 1056—1064 |
| [17] | Tsuchiya K., Orihara Y., Kondo Y., Yoshino N., Ohkubo T., Sakai H., Abe M., J. Am. Chem. Soc., 2004, 126(39), 12282—12283 |
| [18] | Ghosh S., Irvin K., Thayumanavan S., Langmuir,2007, 23(15), 7916—7919 |
| [19] | Gallardo B. S., Metcalfe K. L., Abbott N. L., Langmuir,1996, 12(17), 4116—4124 |
| [20] | Arthur T., Harjani J. R., Phan L., Jessop P. G., Hodson P. V., Green Chem., 2012, 14(2), 357—362 |
| [21] | Zhang Y. M., Feng Y. J., Wang Y. J., Li X. L., Langmuir,2013, 29(13), 4187—4192 |
| [22] | Wang D., Dong R. H., Long P. F., Hao J. C., Soft Matter, 2011, 7(22), 10713—10719 |
| [23] | Guo Z. R., Feng Y. J., Zhu D. W., He S., Liu H. B., Shi X. R., Sun J., Qu M. Z., Adv. Funct. Mater., 2013, 23(40), 5010—5018 |
| [24] | Jiang J. Z., He Y. N., Wan L. P., Cui Z. G., Jessop P. G., Chem. Commun., 2013, 49(19), 1912—1914 |
| [25] | Zhu Y., Jiang J. Z., Liu K. H., Cui Z. G., Binks B. P., Langmuir,2015, 31(11), 3301—3307 |
| [26] | Cui Z. K., Phoeung T., Rousseau P. A., Rydzek G., Zhang Q., Bazuin C. G., Lafleur M., Langmuir,2014, 30(36), 10818—10825 |
| [27] | Saji T., Hoshino K., Ishii Y., Goto M., J. Am. Chem. Soc., 1991, 113(2), 450—456 |
| [28] | Chang X. Y., Cheng Z. Y., Ren B. Y., Dong R. F., Peng J., Fu S. Y., Tong Z., Soft Matter, 2015, 11(38), 7494—7501 |
| [29] | Dupin D., Howse J. R., Armes S. P., Randall D. P., J. Mater. Chem., 2008, 18(5), 545—552 |
| [30] | Zhu Y., Jiang J. Z., Cui Z. G., Binks B. P., Soft Matter, 2014, 10(48), 9739—9745 |
| [31] | Bergsma M., Fielden M. L., Engberts J. B. F. N., Journal of Colloid and Interface Science, 2001, 243(2), 491—495 |
| [32] | Jiang Z. Q., Guo X. F., Jia L. H., China Surfactant Detergent & Cosmetics,2013, 43(5), 330—335 |
| (姜志强, 郭祥峰, 贾丽华.日用化学工业,2013, 43(5), 330—335) | |
| [33] | Cui Z.G., Fundamentals of Surfactants Colloids and Interface Chemistry,Chemical Industry Press, Beijing, 2013, 274—277 |
| (崔正刚. 表面活性剂、 胶体与界面化学,北京: 化学工业出版社, 2013, 274—277) |
| [1] | WANG Hong, SAN Khin Nyein Ei, FANG Yun, ZHANG Xinyu, FAN Ye. Pickering Emulsion Stabilization and Interfacial Catalytic Oxidation by Janus Nano-Au [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220105. |
| [2] | TANG Gang, SUN Junjie, ZHANG Dongxin, WU Qiang, ZHANG Hexin, SHEN Haifeng, TAO Yi, LIU Xiuyu. Synthesis of Phosphaphenanthrene-modified Cardanol-based Polyol and Its Performance as Flame Retardant in Rigid Polyurethane Foams [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210847. |
| [3] | YANG Junge, GAO Chengqian, LI Boxin, YIN Dezhong. Preparation of High Thermal Conductivity Phase Change Monolithic Materials Based on Pickering Emulsion Stabilized by Surface Modified Graphene Oxide [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210593. |
| [4] | CHU Yao, WANG Shuo, ZHANG Zinuo, WANG Yibo, CAI Yibing. Preparation and Properties of Cu Particles Loaded Foam-based Phase Change Composites [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210619. |
| [5] | QIAO Zhenghua, FAN Qi, HAO Jingcheng. Silicone Surfactant-enhanced Dual Networks and High Temperature Resistance Porous Silicone Elastomers [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220384. |
| [6] | HAN Yixiu, WU Dianguo, LI Hongpu, YIN Hongyao, MEI Yongjun, FENG Yujun, ZHONG Zuqin. Interactions Between Hydrophobic Associating Poly(sodium acrylate) and a Zwitterionic Surfactant in Non-aqueous Media and Low Temperature Environment [J]. Chem. J. Chinese Universities, 2021, 42(6): 2056. |
| [7] | HU Xueyi, HAN Lulu, FANG Yun, XIA Yongmei. Admicelles and Adsolubilization of Extended Surfactants on Alumina [J]. Chem. J. Chinese Universities, 2021, 42(3): 843. |
| [8] | HUANG Yan, ZHANG Shuxin, NULI Yanna, WANG Baofeng, YANG Jun, WANG Jiulin. In⁃situ Growth NiS/nickel Foam as Cathode Current Collector of Magnesium-sulfur Batteries [J]. Chem. J. Chinese Universities, 2021, 42(3): 794. |
| [9] | ZHENG Zirui, LI Zilu, ZHAO Kefei, WU Tianyue, ZHANG Chenhui, GAO Yuxia, DU Fengpei. Interfacial Behaviors of Bio-based Surfactant Escin [J]. Chem. J. Chinese Universities, 2021, 42(10): 3107. |
| [10] | LI Boxin, YANG Junge, YIN Dezhong, GAO Chengqian, ZHANG Qiuyu. Preparation of Large-sized Microencapsulated Phase Change Materials Through Pickering Emulsion Stabilized by Monodisperse Polymer Microspheres [J]. Chem. J. Chinese Universities, 2020, 41(9): 2085. |
| [11] | ZHANG Xuan,ZHANG Tianci,JIANG Ping,GE Jijiang,ZHANG Guicai. Enhancement of CO2 Foam Stability with Modified Silica Nanoparticles in High Salinity Brine † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1076. |
| [12] | REN Wen, ZHANG Guoli, YAN Han, HU Xinghua, LI Kun, WANG Jingfeng, LI Ruiqi. Preparation of Superhydrophobic Polyaniline/Polytetrafluoroethylenethylene Composite Membrane and Its Separation Ability for Oil-Water Emulsion † [J]. Chem. J. Chinese Universities, 2020, 41(4): 846. |
| [13] | LI Ming,CUI Xiaoqian,WANG Xuan,LI Zaijun. Synthesis of Amphiphilic Graphene Quantum Dots and Their Sustained Release Effect on L-Menthol † [J]. Chem. J. Chinese Universities, 2020, 41(2): 324. |
| [14] | ZHANG Xinyu, WANG Hong, FANG Yun, FAN Ye. Stimuli-responsive Fe3O4 Nanoparticle Modified by Conjugated Linoleic Acid [J]. Chem. J. Chinese Universities, 2020, 41(11): 2519. |
| [15] | CHEN Jing,CHEN Jinhua,YIN Dezhong,ZHANG Wei. Phase Inversion of Pickering Emulsion Stabilized by Adipate-functionalized SiO2 Particles † [J]. Chem. J. Chinese Universities, 2020, 41(1): 140. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||