

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (11): 1977.doi: 10.7503/cjcu20160434
• Organic Chemistry • Previous Articles Next Articles
QIN Yaoguo1, ZHANG Jingpeng1, SONG Dunlun2, DUAN Hongxia1, LING Yun1, JIANG Biaobiao1, WANG Di1, YANG Xinling1,*( )
)
Received:2016-06-15
Online:2016-11-10
Published:2016-10-18
Contact:
YANG Xinling
E-mail:yangxl@cau.edu.cn
Supported by:CLC Number:
TrendMD:
QIN Yaoguo, ZHANG Jingpeng, SONG Dunlun, DUAN Hongxia, LING Yun, JIANG Biaobiao, WANG Di, YANG Xinling. Design, Synthesis and Biological Activity of Novel Aphid Alarm Pheromone Analogues Containing Isonicotinic Acid†[J]. Chem. J. Chinese Universities, 2016, 37(11): 1977.
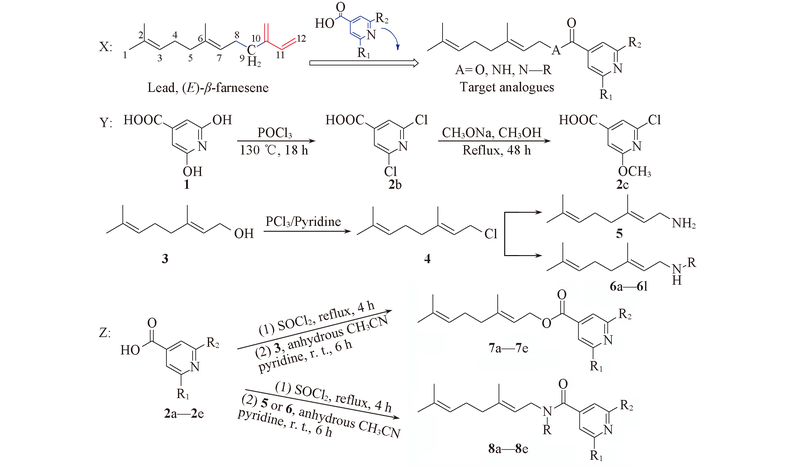
Scheme 1 Design strategy(X), general synthetic route of intermediates(Y) and target compounds(Z)2a, 7a: R1=H, R2=H; 2b, 7b: R1=Cl, R2=Cl; 2c, 7c: R1=Cl, R2=OCH3; 2d, 7d: R1=Cl, R2=H; 2e, 7e: R1=OCH3, R2=H; 6a: R=CH3; 6b: R=CH2CH3; 6c: R=(CH2)2CH3; 6d: R =(CH2)3CH3; 6e: R=(CH2)4CH3; 6f: R=(CH2)5CH3; 6g: R=CH(CH3)2; 6h: R=CH2CH(CH3)2; 6i: R=C(CH3)3; 6j: R=A; 6k: R=B; 6l: R=C; 8a: R1=H, R2=H, R=H; 8b: R1=Cl, R2=Cl, R=H; 8c: R1=Cl, R2=OCH3, R=H; 8d: R1=Cl, R2=Cl, R=CH3; 8e: R1=Cl, R2=Cl, R=CH2CH3; 8f: R1=Cl, R2=Cl, R=(CH2)2CH3; 8g: R1=Cl, R2=Cl, R=(CH2)3CH3; 8h: R1=Cl, R2=Cl, R=(CH2)4CH3; 8i: R1=Cl, R2=Cl, R=(CH2)5CH3; 8j: R1=Cl, R2=Cl, R=CH(CH3)2; 8k: R1=Cl, R2=Cl, R=CH2CH(CH3)2; 8l: R1=Cl, R2=Cl, R=C(CH3)3; 8m: R1=Cl, R2=Cl, R=A; 8n: R1=Cl, R2=Cl, R=B; 8o: R1=Cl, R2=Cl, R=C. A= ; B=; C=.
| Compd. | Chemical formula | MS(calcd.), m/z | Compd. | Chemical formula | MS(calcd.), m/z |
|---|---|---|---|---|---|
| 4 | C11H20O | 169.1(168.1) | 6f | C16H31N | 238.2(237.2) |
| 5 | C10H19N | 154.2(153.2) | 6g | C13H25N | 196.2(195.2) |
| 6a | C11H21N | 168.2(167.2) | 6h | C14H27N | 210.2(209.2) |
| 6b | C12H23N | 182.2(181.2) | 6i | C14H27N | 210.2(209.2) |
| 6c | C13H25N | 196.2(195.2) | 6j | C16H29N | 236.2(235.2) |
| 6d | C14H27N | 210.2(209.2) | 6k | C16H23N | 230.2(229.2) |
| 6e | C15H29N | 224.2(223.2) | 6l | C17H25N | 244.2(243.2) |
Table 1 MS data of intermediates 4—6*
| Compd. | Chemical formula | MS(calcd.), m/z | Compd. | Chemical formula | MS(calcd.), m/z |
|---|---|---|---|---|---|
| 4 | C11H20O | 169.1(168.1) | 6f | C16H31N | 238.2(237.2) |
| 5 | C10H19N | 154.2(153.2) | 6g | C13H25N | 196.2(195.2) |
| 6a | C11H21N | 168.2(167.2) | 6h | C14H27N | 210.2(209.2) |
| 6b | C12H23N | 182.2(181.2) | 6i | C14H27N | 210.2(209.2) |
| 6c | C13H25N | 196.2(195.2) | 6j | C16H29N | 236.2(235.2) |
| 6d | C14H27N | 210.2(209.2) | 6k | C16H23N | 230.2(229.2) |
| 6e | C15H29N | 224.2(223.2) | 6l | C17H25N | 244.2(243.2) |
| Compd. | Appearance | Yield(%) | Chemical formula | ESI-HRMSb(calcd.), m/z | IR(KBr), | |
|---|---|---|---|---|---|---|
| 7a | Yellow liquid | 85.9 | C16H21NO2 | 260.1646(260.1645) | 3031, 2967, 2918, 2857, 1729, 1408, 1278, 1119, 930, 850 | |
| 7b | Yellow liquid | 85.3 | C16H19Cl2NO2 | 328.0867(328.0866) | 3088, 2967, 2926, 2856, 1732, 1582, 1548, 1442, 1358, 1277, 1155, 933, 816 | |
| 7c | Yellow liquid | 70.3 | C17H22ClNO3 | 324.1364(324.1361) | 3091, 2951, 2925, 2856, 1732, 1604, 1555, 1463, 1384, 1357, 1317, 1265, 1153, 1042, 938, 887 | |
| 7d | Yellow liquid | 74.5 | C16H20ClNO2 | 294.1259(294.1255) | 3064, 2968, 2918, 2857, 1731, 1591, 1552, 1462, 1368, 1291, 1259, 1145, 1107, 931, 860 | |
| 7e | Yellow liquid | 63.2 | C17H23NO3 | 290.1750(290.1751) | 2968, 2925, 2857, 1731, 1674, 1611, 1564, 1553, 1449, 1386, 1292, 1259, 1215, 1039, 935, 840 | |
| 8a | Yellow liquid | 63.8 | C16H22N2O | 259.1806(259.1805) | 3304, 3045, 2965, 2924, 2855, 1645, 1600, 1542, 1490, 1449, 1297, 1066, 846 | |
| 8b | White solid | 85.5 | C16H20Cl2N2O | 327.1026(327.1025) | 3273, 3129, 3083, 2964, 2924, 2854, 1644, 1447, 1358, 1294, 1158, 820 | |
| 8c | White solid | 69.5 | C17H23ClN2O2 | 323.1519(323.1521) | 3273, 2924, 2856, 1645, 1603, 1538, 1463, 1379, 1162, 1047, 873 | |
| 8d | Yellow liquid | 68.5 | C17H22Cl2N2O | 363.1000(363.1001) | 3270, 3068, 2971, 2930, 2856, 1644, 1583, 1533, 1455, 1381, 1309, 1286, 1264, 1164, 873, 822 | |
| 8e | Yellow liquid | 83.2 | C18H24Cl2N2O | 355.1339(355.1338) | 3270, 3068, 2971, 2930, 2856, 1644, 1583, 1533, 1455, 1361, 1164, 873, 822 | |
| 8f | Yellow liquid | 91.9 | C19H26Cl2N2O | 369.1496(369.1495) | 3269, 3068, 2966, 2930, 2876, 1643, 1583, 1534, 1437, 1406, 1361, 1261, 1164, 872, 822 | |
| 8g | Yellow liquid | 68.6 | C20H28Cl2N2O | 405.1472(405.1471) | 3068, 2963, 2928, 2872, 1643, 1583, 1534, 1436, 1361, 1290, 1266, 1164, 872, 822 | |
| 8h | Yellow liquid | 59.3 | C21H30Cl2N2O | 419.1628(419.1627) | 3068, 2960, 2930, 2859, 1642, 1583, 1534, 1438, 1360, 1263, 1163, 872, 822 | |
| 8i | Yellow liquid | 91.8 | C22H32Cl2N2O | 411.1967(411.1964) | 3068, 2958, 2929, 2858, 1642, 1583, 1534, 1438, 1360, 1264, 1163, 872, 822 | |
| 8j | Yellow liquid | 83.7 | C19H26Cl2N2O | 369.1496(369.1495) | 3066, 2973, 2929, 1638, 1582, 1534, 1449, 1406, 1360, 1164, 872, 823 | |
| 8k | Yellow liquid | 90.7 | C20H28Cl2N2O | 383.1655(383.1651) | 3068, 2962, 2930, 2873, 1642, 1583, 1534, 1438, 1383, 1360, 1164, 872, 822 | |
| 8l | Yellow liquid | 80.0 | C20H28Cl2N2O | 383.1649(383.1651) | 3068, 2967, 2925, 1645, 1580, 1534, 1421, 1361, 1261,1164, 871, 823 | |
| 8m | Yellow liquid | 60.1 | C22H30Cl2N2O | 409.1808(409.1808) | 3068, 2931, 2856, 1714, 1634, 1582, 1533, 1453, 1405, 1361, 1253, 1164, 1102, 871, 822 | |
| 8n | Yellow liquid | 67.3 | C22H24Cl2N2O | 403.1336(403.1338) | 3066, 2967, 2924, 2855, 1654, 1594, 1582, 1536, 1494, 1420, 1384, 1359, 1293, 1164, 874, 819 | |
| 8o | Yellow liquid | 91.8 | C23H26Cl2N2O | 417.1493(417.1495) | 3066, 3030, 2967, 2924, 2855, 1643, 1583, 1535, 1496, 1451, 1360, 1263, 1164, 872, 822 | |
Table 2 Appearance, yields, HRMS and IR data of target compounds 7 and 8a
| Compd. | Appearance | Yield(%) | Chemical formula | ESI-HRMSb(calcd.), m/z | IR(KBr), | |
|---|---|---|---|---|---|---|
| 7a | Yellow liquid | 85.9 | C16H21NO2 | 260.1646(260.1645) | 3031, 2967, 2918, 2857, 1729, 1408, 1278, 1119, 930, 850 | |
| 7b | Yellow liquid | 85.3 | C16H19Cl2NO2 | 328.0867(328.0866) | 3088, 2967, 2926, 2856, 1732, 1582, 1548, 1442, 1358, 1277, 1155, 933, 816 | |
| 7c | Yellow liquid | 70.3 | C17H22ClNO3 | 324.1364(324.1361) | 3091, 2951, 2925, 2856, 1732, 1604, 1555, 1463, 1384, 1357, 1317, 1265, 1153, 1042, 938, 887 | |
| 7d | Yellow liquid | 74.5 | C16H20ClNO2 | 294.1259(294.1255) | 3064, 2968, 2918, 2857, 1731, 1591, 1552, 1462, 1368, 1291, 1259, 1145, 1107, 931, 860 | |
| 7e | Yellow liquid | 63.2 | C17H23NO3 | 290.1750(290.1751) | 2968, 2925, 2857, 1731, 1674, 1611, 1564, 1553, 1449, 1386, 1292, 1259, 1215, 1039, 935, 840 | |
| 8a | Yellow liquid | 63.8 | C16H22N2O | 259.1806(259.1805) | 3304, 3045, 2965, 2924, 2855, 1645, 1600, 1542, 1490, 1449, 1297, 1066, 846 | |
| 8b | White solid | 85.5 | C16H20Cl2N2O | 327.1026(327.1025) | 3273, 3129, 3083, 2964, 2924, 2854, 1644, 1447, 1358, 1294, 1158, 820 | |
| 8c | White solid | 69.5 | C17H23ClN2O2 | 323.1519(323.1521) | 3273, 2924, 2856, 1645, 1603, 1538, 1463, 1379, 1162, 1047, 873 | |
| 8d | Yellow liquid | 68.5 | C17H22Cl2N2O | 363.1000(363.1001) | 3270, 3068, 2971, 2930, 2856, 1644, 1583, 1533, 1455, 1381, 1309, 1286, 1264, 1164, 873, 822 | |
| 8e | Yellow liquid | 83.2 | C18H24Cl2N2O | 355.1339(355.1338) | 3270, 3068, 2971, 2930, 2856, 1644, 1583, 1533, 1455, 1361, 1164, 873, 822 | |
| 8f | Yellow liquid | 91.9 | C19H26Cl2N2O | 369.1496(369.1495) | 3269, 3068, 2966, 2930, 2876, 1643, 1583, 1534, 1437, 1406, 1361, 1261, 1164, 872, 822 | |
| 8g | Yellow liquid | 68.6 | C20H28Cl2N2O | 405.1472(405.1471) | 3068, 2963, 2928, 2872, 1643, 1583, 1534, 1436, 1361, 1290, 1266, 1164, 872, 822 | |
| 8h | Yellow liquid | 59.3 | C21H30Cl2N2O | 419.1628(419.1627) | 3068, 2960, 2930, 2859, 1642, 1583, 1534, 1438, 1360, 1263, 1163, 872, 822 | |
| 8i | Yellow liquid | 91.8 | C22H32Cl2N2O | 411.1967(411.1964) | 3068, 2958, 2929, 2858, 1642, 1583, 1534, 1438, 1360, 1264, 1163, 872, 822 | |
| 8j | Yellow liquid | 83.7 | C19H26Cl2N2O | 369.1496(369.1495) | 3066, 2973, 2929, 1638, 1582, 1534, 1449, 1406, 1360, 1164, 872, 823 | |
| 8k | Yellow liquid | 90.7 | C20H28Cl2N2O | 383.1655(383.1651) | 3068, 2962, 2930, 2873, 1642, 1583, 1534, 1438, 1383, 1360, 1164, 872, 822 | |
| 8l | Yellow liquid | 80.0 | C20H28Cl2N2O | 383.1649(383.1651) | 3068, 2967, 2925, 1645, 1580, 1534, 1421, 1361, 1261,1164, 871, 823 | |
| 8m | Yellow liquid | 60.1 | C22H30Cl2N2O | 409.1808(409.1808) | 3068, 2931, 2856, 1714, 1634, 1582, 1533, 1453, 1405, 1361, 1253, 1164, 1102, 871, 822 | |
| 8n | Yellow liquid | 67.3 | C22H24Cl2N2O | 403.1336(403.1338) | 3066, 2967, 2924, 2855, 1654, 1594, 1582, 1536, 1494, 1420, 1384, 1359, 1293, 1164, 874, 819 | |
| 8o | Yellow liquid | 91.8 | C23H26Cl2N2O | 417.1493(417.1495) | 3066, 3030, 2967, 2924, 2855, 1643, 1583, 1535, 1496, 1451, 1360, 1263, 1164, 872, 822 | |
| Compd. | 1H NMR(300 MHz, CDCl3), δ | 13C NMR(75 MHz, CDCl3), δ | ||
|---|---|---|---|---|
| 7a | 8.71—8.73(m, 2H, ArH), 7.80—7.82(m, 2H, ArH), 5.37—5.42(m, 1H, C═CH), 5.03—5.05(m, 1H, C═CH), 4.83(d, J=7.1 Hz, 2H, CH2), 2.00—2.12(m, 4H, CH2CH2), 1.74(s, 3H, CH3), 1.63(s, 3H, CH3), 1.56(s, 3H, CH3) | 164.62, 150.06, 142.86, 137.35, 131.42, 123.24, 122.48, 117.36, 62.18, 39.15, 25.84, 25.22, 17.25, 16.13 | ||
| 7b | 7.80—7.81(m, 2H, ArH), 5.41—5.46(m, 1H, C═CH), 5.06—5.09(m, 1H, C═CH), 4.88(d, J=7.3 Hz, 2H, CH2), 2.07—2.16(m, 4H, CH2CH2), 1.68—1.77 [m, 6H, C(CH3)2], 1.61(s, 3H, CH3) | 162.24, 151.00, 143.75, 142.53, 131.51, 123.17, 122.24, 116.80, 62.94, 39.13, 25.83, 25.25, 17.29, 16.19 | ||
| 7c | 7.44(d, J=1.1 Hz, 1H, ArH), 7.23(d, J=1.1 Hz, 1H, ArH), 5.43—5.48(m, 1H, C═CH), 5.07—5.12(m, 1H, C═CH), 4.86(d, J=7.2 Hz, 2H, CH2), 3.97(s, 3H, OCH3), 2.05—2.17(m, 4H, CH2CH2), 1.78(s, 3H, CH3), 1.69(s, 3H, CH3), 1.62(s, 3H, CH3) | 163.99, 163.47, 148.80, 143.14, 142.46, 131.53, 123.24, 117.19, 115.35, 109.30, 62.42, 54.07, 39.15, 25.87, 25.27, 17.30, 16.18 | ||
| 7d | 8.53(dd, J1=5.1 Hz, J2=0.7 Hz, 1H, ArH), 7.89(dd, J1=1.3 Hz, J2=0.8 Hz, 1H, ArH), 7.78(dd, J1=5.1 Hz, J2=1.4 Hz, 1H, ArH), 5.42—5.48(m, 1H, C═CH), 5.06—5.11(m, 1H, C═CH), 4.88(d, J=7.2 Hz, 2H, CH2), 2.07—2.16(m, 4H, CH2CH2), 1.78(s, 3H, CH3), 1.68(s, 3H, CH3), 1.61(s, 3H, CH3) | 163.23, 151.93, 149.98, 143.18, 140.23, 131.35, 123.54, 123.18, 121.19, 117.08, 62.47, 39.08, 25.79, 25.19, 17.22, 16.10 | ||
| 7e | 8.29(dd, J1=5.3 Hz, J2=0.6 Hz, 1H, ArH), 7.43(dd, J1=5.3 Hz, J2=1.4 Hz, 1H, ArH), 7.34(d, J=0.4 Hz, 1H, ArH), 5.44—5.49(m, 1H, C═CH), 5.08—5.13(m, 1H, C═CH), 4.87(d, J=7.1 Hz, 2H, CH2), 3.98(s, 3H, OCH3), 2.06—2.17(m, 4H, CH2CH2), 1.79(s, 3H, CH3), 1.70(s, 3H, CH3), 1.62(s, 3H, CH3) | 164.68, 164.47, 147.24, 142.79, 140.20, 131.53, 123.29, 117.45, 115.41, 110.87, 62.12, 53.42, 39.17, 25.91, 25.29, 17.32, 16.20 | ||
| 8a | 8.71—8.73(m, 2H, ArH), 7.60—7.62(m, 2H, ArH), 6.28(brs, 1H, NH), 5.26—5.31(m, 1H, C═CH), 5.06—5.10(m, 1H, C═CH), 4.04—4.13(m, 2H, CH2), 2.01—2.11(m, 4H, CH2CH2), 1.68—1.72 [m, 6H, C(CH3)2], 1.60(s, 3H, CH3) | 165.09, 149.63, 141.58, 139.27, 131.16, 123.31, 120.87, 119.19, 39.03, 37.76, 25.92, 25.18, 17.19, 15.82 | ||
| 8b | 7.57(s, 2H, ArH), 6.08(brs, 1H, NH), 5.24—5.29(m, 1H, C═CH), 5.06—5.08(m, 1H, C═CH), 4.05(t, J=5.9 Hz, 2H, CH2), 2.03—2.12(m, 4H, CH2CH2), 1.72—1.69 [m, 6H, C(CH3)2], 1.61(s, 3H, CH3) | 162.61, 150.98, 146.81, 140.86, 131.52, 123.26, 120.44, 118.23, 39.11, 38.08, 25.97, 25.29, 17.32, 16.03 | ||
| 8c | 7.21(d, J=1.1 Hz, 1H, ArH), 6.97(d, J=1.1 Hz, 1H, ArH), 6.42(s, 1H, NH), 5.25—5.39(m, 1H, C═CH), 5.07—5.11(m, 1H, C═CH), 4.03(t, J=6.1 Hz, 2H, CH2), 3.96(s, 3H, OCH3) 2.02—2.12(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.70(s, 3H, CH3), 1.62(s, 3H, CH3) | 164.00, 163.89, 148.94, 146.78, 140.61, 131.50, 123.36, 118.63, 113.63, 106.88, 54.09, 39.13, 37.84, 26.00, 25.31, 17.33, 16.01 | ||
| 8d | 7.26 and 7.27(2s, 2H, ArH), 5.07—5.21(m, 2H, C═CH), 4.14 and 3.76(2d, J=7.1, 6.7 Hz, 2H, CH2), 3.04 and 2.85(2s, 3H, N—CH3), 2.03—2.10(m, 4H, CH2CH2), 1.74 and 1.52(2s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3) | 165.66, 165.20, 150.49, 150.43, 149.07, 141.13, 140.59, 131.48, 131.20, 123.37, 123.11, 120.17, 120.12, 117.69, 117.51, 48.65, 44.19, 39.15, 39.00, 35.64, 32.15, 25.86, 25.80, 25.25, 17.27, 15.82, 15.71 | ||
| 8e | 7.25 and 7.27(2s, 2H, ArH), 5.05—5.22(m, 2H, C═CH), 4.14 and 3.75(2d, J=7.0, 5.8 Hz, 2H, CH2), 3.48—3.55 and 3.14—3.21(2m, 2H, N—CH2), 2.05—2.09(m, 4H, CH2CH2), 1.69(s, 3H, CH3), 1.62 [d, J=4.7 Hz, 6H, C(CH3)2], 1.22 and 1.11(2t, J=7.2, 7.1 Hz, 3H, CH3) | 165.37, 150.55, 150.42, 149.45, 149.33, 140.17, 131.52, 131.21, 123.40, 123.15, 120.04, 119.72, 118.41, 118.28, 46.25, 42.28, 41.29, 39.70, 39.13, 39.00, 25.80, 25.26, 17.29, 15.83, 15.71, 13.55, 12.08 | ||
| 8f | 7.22(d, J=2.0 Hz, 2H, ArH), 5.07—5.30(m, 2H, C═CH), 4.13 and 3.74(2d, J=6.9, 6.7 Hz, 2H, CH2), 3.42 and 3.07(2t, J=7.6, 7.5 Hz, 2H, N—CH2), 2.04—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.48(s, 2H, CH2), 0.97 and 0.80(2t, J=7.4 Hz, 3H, CH3) | 165.64, 150.51, 150.40, 149.45, 140.20, 140.12, 131.48, 131.18, 123.40, 123.14, 119.98, 119.91, 118.39, 118.23, 49.23, 46.58, 46.31, 41.62, 39.11, 38.97, 25.79, 25.25, 21.26, 20.10, 17.27, 15.82, 15.67, 10.97, 10.65 | ||
| 8g | 7.23(s, 2H, ArH), 5.06—5.23(m, 2H, C═CH), 4.13 and 3.74(2d, J=6.8, 6.4 Hz, 2H, CH2), 3.45 and 3.09(2t, J=7.6 Hz, 2H, N—CH2), 2.04—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.48(s, 2H, CH2), 0.97 and 0.80(2t, J=7.4 Hz, 3H, CH3) | 166.14, 165.95, 150.54, 150.46, 149.64, 140.27, 140.18, 131.55, 131.25, 123.39, 123.13, 120.24, 119.88, 118.25, 188.03, 55.00, 51.34, 46.89, 41.74, 39.13, 38.98, 26.39, 26.23, 25.88, 25.79, 25.29, 19.79, 19.43, 17.32, 15.90, 15.69 | ||
| 8h | 7.22(d, J=2.0 Hz, 2H, ArH), 5.06—5.23(m, 2H, C═CH), 4.13 and 3.74(2d, J=6.8, 6.6 Hz, 2H, CH2), 3.44 and 3.08(2t, J=7.7, 7.6 Hz, 2H, N—CH2), 2.04—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.12—1.58(m, 6H, CH2CH2CH2), 0.83—0.95(m, 3H, CH3) | 165.53, 150.51, 150.40, 149.47, 140.12, 131.48, 131.18, 123.39, 123.14, 119.98, 119.88, 118.40, 118.25, 47.60, 46.53, 44.71, 41.65, 39.12, 38.98, 28.71, 28.22, 27.64, 26.49, 25.80, 25.24, 21.98, 21.68, 17.27, 15.85, 15.69, 13.54, 13.37 | ||
| 8i | 7.23(s, 2H, ArH), 5.05—5.23(m, 2H, C═CH), 4.13 and 3.74(2d, J=7.1, 6.5 Hz, 2H, CH2), 3.44 and 3.08(2t, J=7.7, 7.1 Hz, 2H, N—CH2), 2.06—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.17—1.57(m, 8H, CH2CH2CH2CH2), 0.84—0.91(m, 3H, CH3) | 165.51, 150.50, 150.39, 149.47, 140.17, 140.09, 131.46, 131.16, 123.39, 123.14, 119.98, 119.89, 118.41, 118.26, 47.60, 46.53, 44.73, 41.64, 39.12, 38.97, 31.08, 30.75, 27.92, 26.77, 26.23, 25.80, 25.74, 25.24, 22.12, 21.97, 17.26, 15.84, 15.68, 13.56, 13.44 | ||
| 8j | 7.21(s, 2H, ArH), 5.01—5.24(m, 2H, C═CH), 4.64—4.72 and 4.00—4.02(2m, 2H, CH2), 3.68—3.79(m, 2H, CH2), 2.01—2.11(m, 4H, CH2CH2), 1.73(s, 3H, CH3), 1.68(s, 3H, CH3), 1.61(s, 3H, CH3), 1.34(s, 1H, CH), 1.27 and 1.18[2d, J=6.8, 6.5 Hz, 6H, C(CH3)2] | 165.97, 165.22, 150.56, 150.24, 149.94, 138.24, 137.04, 131.41, 131.00, 123.57, 123.27, 120.78, 119.92, 119.46, 50.13, 46.37, 42.72, 38.88, 38.38, 25.81, 25.24, 20.88, 19.72, 17.28, 15.79, 15.56 | ||
| 8k | 7.19—7.21(m, 2H, ArH), 5.03—5.23(m, 2H, C═CH), 4.14 and 3.75(2d, J=7.0, 6.4 Hz, 2H, CH2), 3.31 and 2.95(2d, J=7.6, 7.5 Hz, 2H, N—CH2), 2.04—2.09(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.45(s, 1H, CH), 0.97 and 0.81[2d, J=6.7 Hz, 6H,(CH3)2] | 165.58, 150.54, 150.43, 149.46, 140.16, 131.53, 131.22, 123.40, 123.14, 119.98, 119.89, 118.38, 118.23, 47.39, 46.54, 44.47, 41.66, 39.14, 38.99, 30.11, 28.94, 25.81, 25.26, 19.82, 19.33, 17.29, 15.88, 15.71, 13.42, 13.15 | ||
| 8l | 7.19(s, 2H, ArH), 5.05—5.08(m, 2H, C═CH), 3.77(d, J=5.6 Hz, 2H, CH2), 2.01—2.11(m, 4H, CH2CH2), 1.69(s, 3H, CH3), 1.62(s, 3H, CH3), 1.52[s, 9H, C(CH3)3], 1.34(s, 3H, CH3) | 167.08, 151.36, 150.28, 137.39, 131.44, 123.35, 121.75, 119.50, 57.71, 45.35, 38.81, 28.06, 28.01, 25.77, 25.30, 17.35, 15.66 | ||
| 8m | 7.21(s, 2H, ArH), 5.02—5.15(m, 2H, C═CH), 4.02 and 3.69(2d, J=5.4, 5.9 Hz, 2H, CH2), 4.34 and 3.23(2t, J=14.1, 12.5 Hz, 2H, N—CH2), 2.00—2.17(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.68(s, 3H, CH3), 1.61(s, 3H, CH3), 1.73—1.85 and 1.05—1.57(2m, 10H, CH2CH2CH2CH2CH2) | 166.07, 165.38, 150.60, 150.22, 149.99, 137.98, 136.96, 131.44, 131.02, 123.58, 123.28, 120.97, 119.93, 119.45, 58.71, 54.37, 53.13, 43.10, 39.49, 38.86, 30.17, 25.86, 25.43, 25.24, 17.30, 15.86, 15.58 | ||
| 8n | 7.24—7.30(m, 4H, ArH), 7.01—7.08(m, 2H, ArH), 7.02(d, J=6.9 Hz, 1H, ArH), 5.28—5.33(m, 1H, C═CH), 5.01—5.03(m, 1H, C═CH), 4.48(d, J=7.3 Hz, 2H, CH2), 1.98—2.07(m, 4H, CH2CH2), 1.68(s, 3H, CH3), 1.57(t, J=2.6 Hz, 3H, CH3), 1.50(s, 3H, CH3) | 164.52, 149.94, 148.83, 141.01, 140.73, 131.22, 129.25, 127.74, 127.67, 123.46, 121.54, 117.63, 47.63, 39.14, 25.91, 25.32, 17.32, 15.75 | ||
| 8o | 7.24—7.41(m, 7H, ArH), 5.06—5.30(m, 2H, C═CH), 4.70 and 4.37(2s, 2H, CH2), 4.11 and 3.67(2d, J=6.8, 6.5 Hz, 2H, CH2), 2.04—2.11(m, 4H, CH2CH2), 1.70(s, 3H, CH3), 1.63(s, 3H, CH3), 1.38(s, 3H, CH3) | 166.07, 150.59, 149.03, 141.27, 140.69, 135.99, 135.30, 131.60, 131.31, 128.66, 128.43, 128.01, 127.67, 127.46, 126.35, 123.52, 123.28, 120.06, 117.98, 117.61, 53.17, 51.03, 47.25, 45.83, 41.93, 39.04, 25.83, 25.37, 17.41, 15.82 | ||
Table 3 1H NMR and 13C NMR data of target compounds 7 and 8*
| Compd. | 1H NMR(300 MHz, CDCl3), δ | 13C NMR(75 MHz, CDCl3), δ | ||
|---|---|---|---|---|
| 7a | 8.71—8.73(m, 2H, ArH), 7.80—7.82(m, 2H, ArH), 5.37—5.42(m, 1H, C═CH), 5.03—5.05(m, 1H, C═CH), 4.83(d, J=7.1 Hz, 2H, CH2), 2.00—2.12(m, 4H, CH2CH2), 1.74(s, 3H, CH3), 1.63(s, 3H, CH3), 1.56(s, 3H, CH3) | 164.62, 150.06, 142.86, 137.35, 131.42, 123.24, 122.48, 117.36, 62.18, 39.15, 25.84, 25.22, 17.25, 16.13 | ||
| 7b | 7.80—7.81(m, 2H, ArH), 5.41—5.46(m, 1H, C═CH), 5.06—5.09(m, 1H, C═CH), 4.88(d, J=7.3 Hz, 2H, CH2), 2.07—2.16(m, 4H, CH2CH2), 1.68—1.77 [m, 6H, C(CH3)2], 1.61(s, 3H, CH3) | 162.24, 151.00, 143.75, 142.53, 131.51, 123.17, 122.24, 116.80, 62.94, 39.13, 25.83, 25.25, 17.29, 16.19 | ||
| 7c | 7.44(d, J=1.1 Hz, 1H, ArH), 7.23(d, J=1.1 Hz, 1H, ArH), 5.43—5.48(m, 1H, C═CH), 5.07—5.12(m, 1H, C═CH), 4.86(d, J=7.2 Hz, 2H, CH2), 3.97(s, 3H, OCH3), 2.05—2.17(m, 4H, CH2CH2), 1.78(s, 3H, CH3), 1.69(s, 3H, CH3), 1.62(s, 3H, CH3) | 163.99, 163.47, 148.80, 143.14, 142.46, 131.53, 123.24, 117.19, 115.35, 109.30, 62.42, 54.07, 39.15, 25.87, 25.27, 17.30, 16.18 | ||
| 7d | 8.53(dd, J1=5.1 Hz, J2=0.7 Hz, 1H, ArH), 7.89(dd, J1=1.3 Hz, J2=0.8 Hz, 1H, ArH), 7.78(dd, J1=5.1 Hz, J2=1.4 Hz, 1H, ArH), 5.42—5.48(m, 1H, C═CH), 5.06—5.11(m, 1H, C═CH), 4.88(d, J=7.2 Hz, 2H, CH2), 2.07—2.16(m, 4H, CH2CH2), 1.78(s, 3H, CH3), 1.68(s, 3H, CH3), 1.61(s, 3H, CH3) | 163.23, 151.93, 149.98, 143.18, 140.23, 131.35, 123.54, 123.18, 121.19, 117.08, 62.47, 39.08, 25.79, 25.19, 17.22, 16.10 | ||
| 7e | 8.29(dd, J1=5.3 Hz, J2=0.6 Hz, 1H, ArH), 7.43(dd, J1=5.3 Hz, J2=1.4 Hz, 1H, ArH), 7.34(d, J=0.4 Hz, 1H, ArH), 5.44—5.49(m, 1H, C═CH), 5.08—5.13(m, 1H, C═CH), 4.87(d, J=7.1 Hz, 2H, CH2), 3.98(s, 3H, OCH3), 2.06—2.17(m, 4H, CH2CH2), 1.79(s, 3H, CH3), 1.70(s, 3H, CH3), 1.62(s, 3H, CH3) | 164.68, 164.47, 147.24, 142.79, 140.20, 131.53, 123.29, 117.45, 115.41, 110.87, 62.12, 53.42, 39.17, 25.91, 25.29, 17.32, 16.20 | ||
| 8a | 8.71—8.73(m, 2H, ArH), 7.60—7.62(m, 2H, ArH), 6.28(brs, 1H, NH), 5.26—5.31(m, 1H, C═CH), 5.06—5.10(m, 1H, C═CH), 4.04—4.13(m, 2H, CH2), 2.01—2.11(m, 4H, CH2CH2), 1.68—1.72 [m, 6H, C(CH3)2], 1.60(s, 3H, CH3) | 165.09, 149.63, 141.58, 139.27, 131.16, 123.31, 120.87, 119.19, 39.03, 37.76, 25.92, 25.18, 17.19, 15.82 | ||
| 8b | 7.57(s, 2H, ArH), 6.08(brs, 1H, NH), 5.24—5.29(m, 1H, C═CH), 5.06—5.08(m, 1H, C═CH), 4.05(t, J=5.9 Hz, 2H, CH2), 2.03—2.12(m, 4H, CH2CH2), 1.72—1.69 [m, 6H, C(CH3)2], 1.61(s, 3H, CH3) | 162.61, 150.98, 146.81, 140.86, 131.52, 123.26, 120.44, 118.23, 39.11, 38.08, 25.97, 25.29, 17.32, 16.03 | ||
| 8c | 7.21(d, J=1.1 Hz, 1H, ArH), 6.97(d, J=1.1 Hz, 1H, ArH), 6.42(s, 1H, NH), 5.25—5.39(m, 1H, C═CH), 5.07—5.11(m, 1H, C═CH), 4.03(t, J=6.1 Hz, 2H, CH2), 3.96(s, 3H, OCH3) 2.02—2.12(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.70(s, 3H, CH3), 1.62(s, 3H, CH3) | 164.00, 163.89, 148.94, 146.78, 140.61, 131.50, 123.36, 118.63, 113.63, 106.88, 54.09, 39.13, 37.84, 26.00, 25.31, 17.33, 16.01 | ||
| 8d | 7.26 and 7.27(2s, 2H, ArH), 5.07—5.21(m, 2H, C═CH), 4.14 and 3.76(2d, J=7.1, 6.7 Hz, 2H, CH2), 3.04 and 2.85(2s, 3H, N—CH3), 2.03—2.10(m, 4H, CH2CH2), 1.74 and 1.52(2s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3) | 165.66, 165.20, 150.49, 150.43, 149.07, 141.13, 140.59, 131.48, 131.20, 123.37, 123.11, 120.17, 120.12, 117.69, 117.51, 48.65, 44.19, 39.15, 39.00, 35.64, 32.15, 25.86, 25.80, 25.25, 17.27, 15.82, 15.71 | ||
| 8e | 7.25 and 7.27(2s, 2H, ArH), 5.05—5.22(m, 2H, C═CH), 4.14 and 3.75(2d, J=7.0, 5.8 Hz, 2H, CH2), 3.48—3.55 and 3.14—3.21(2m, 2H, N—CH2), 2.05—2.09(m, 4H, CH2CH2), 1.69(s, 3H, CH3), 1.62 [d, J=4.7 Hz, 6H, C(CH3)2], 1.22 and 1.11(2t, J=7.2, 7.1 Hz, 3H, CH3) | 165.37, 150.55, 150.42, 149.45, 149.33, 140.17, 131.52, 131.21, 123.40, 123.15, 120.04, 119.72, 118.41, 118.28, 46.25, 42.28, 41.29, 39.70, 39.13, 39.00, 25.80, 25.26, 17.29, 15.83, 15.71, 13.55, 12.08 | ||
| 8f | 7.22(d, J=2.0 Hz, 2H, ArH), 5.07—5.30(m, 2H, C═CH), 4.13 and 3.74(2d, J=6.9, 6.7 Hz, 2H, CH2), 3.42 and 3.07(2t, J=7.6, 7.5 Hz, 2H, N—CH2), 2.04—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.48(s, 2H, CH2), 0.97 and 0.80(2t, J=7.4 Hz, 3H, CH3) | 165.64, 150.51, 150.40, 149.45, 140.20, 140.12, 131.48, 131.18, 123.40, 123.14, 119.98, 119.91, 118.39, 118.23, 49.23, 46.58, 46.31, 41.62, 39.11, 38.97, 25.79, 25.25, 21.26, 20.10, 17.27, 15.82, 15.67, 10.97, 10.65 | ||
| 8g | 7.23(s, 2H, ArH), 5.06—5.23(m, 2H, C═CH), 4.13 and 3.74(2d, J=6.8, 6.4 Hz, 2H, CH2), 3.45 and 3.09(2t, J=7.6 Hz, 2H, N—CH2), 2.04—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.48(s, 2H, CH2), 0.97 and 0.80(2t, J=7.4 Hz, 3H, CH3) | 166.14, 165.95, 150.54, 150.46, 149.64, 140.27, 140.18, 131.55, 131.25, 123.39, 123.13, 120.24, 119.88, 118.25, 188.03, 55.00, 51.34, 46.89, 41.74, 39.13, 38.98, 26.39, 26.23, 25.88, 25.79, 25.29, 19.79, 19.43, 17.32, 15.90, 15.69 | ||
| 8h | 7.22(d, J=2.0 Hz, 2H, ArH), 5.06—5.23(m, 2H, C═CH), 4.13 and 3.74(2d, J=6.8, 6.6 Hz, 2H, CH2), 3.44 and 3.08(2t, J=7.7, 7.6 Hz, 2H, N—CH2), 2.04—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.12—1.58(m, 6H, CH2CH2CH2), 0.83—0.95(m, 3H, CH3) | 165.53, 150.51, 150.40, 149.47, 140.12, 131.48, 131.18, 123.39, 123.14, 119.98, 119.88, 118.40, 118.25, 47.60, 46.53, 44.71, 41.65, 39.12, 38.98, 28.71, 28.22, 27.64, 26.49, 25.80, 25.24, 21.98, 21.68, 17.27, 15.85, 15.69, 13.54, 13.37 | ||
| 8i | 7.23(s, 2H, ArH), 5.05—5.23(m, 2H, C═CH), 4.13 and 3.74(2d, J=7.1, 6.5 Hz, 2H, CH2), 3.44 and 3.08(2t, J=7.7, 7.1 Hz, 2H, N—CH2), 2.06—2.11(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.17—1.57(m, 8H, CH2CH2CH2CH2), 0.84—0.91(m, 3H, CH3) | 165.51, 150.50, 150.39, 149.47, 140.17, 140.09, 131.46, 131.16, 123.39, 123.14, 119.98, 119.89, 118.41, 118.26, 47.60, 46.53, 44.73, 41.64, 39.12, 38.97, 31.08, 30.75, 27.92, 26.77, 26.23, 25.80, 25.74, 25.24, 22.12, 21.97, 17.26, 15.84, 15.68, 13.56, 13.44 | ||
| 8j | 7.21(s, 2H, ArH), 5.01—5.24(m, 2H, C═CH), 4.64—4.72 and 4.00—4.02(2m, 2H, CH2), 3.68—3.79(m, 2H, CH2), 2.01—2.11(m, 4H, CH2CH2), 1.73(s, 3H, CH3), 1.68(s, 3H, CH3), 1.61(s, 3H, CH3), 1.34(s, 1H, CH), 1.27 and 1.18[2d, J=6.8, 6.5 Hz, 6H, C(CH3)2] | 165.97, 165.22, 150.56, 150.24, 149.94, 138.24, 137.04, 131.41, 131.00, 123.57, 123.27, 120.78, 119.92, 119.46, 50.13, 46.37, 42.72, 38.88, 38.38, 25.81, 25.24, 20.88, 19.72, 17.28, 15.79, 15.56 | ||
| 8k | 7.19—7.21(m, 2H, ArH), 5.03—5.23(m, 2H, C═CH), 4.14 and 3.75(2d, J=7.0, 6.4 Hz, 2H, CH2), 3.31 and 2.95(2d, J=7.6, 7.5 Hz, 2H, N—CH2), 2.04—2.09(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.69(s, 3H, CH3), 1.61(s, 3H, CH3), 1.45(s, 1H, CH), 0.97 and 0.81[2d, J=6.7 Hz, 6H,(CH3)2] | 165.58, 150.54, 150.43, 149.46, 140.16, 131.53, 131.22, 123.40, 123.14, 119.98, 119.89, 118.38, 118.23, 47.39, 46.54, 44.47, 41.66, 39.14, 38.99, 30.11, 28.94, 25.81, 25.26, 19.82, 19.33, 17.29, 15.88, 15.71, 13.42, 13.15 | ||
| 8l | 7.19(s, 2H, ArH), 5.05—5.08(m, 2H, C═CH), 3.77(d, J=5.6 Hz, 2H, CH2), 2.01—2.11(m, 4H, CH2CH2), 1.69(s, 3H, CH3), 1.62(s, 3H, CH3), 1.52[s, 9H, C(CH3)3], 1.34(s, 3H, CH3) | 167.08, 151.36, 150.28, 137.39, 131.44, 123.35, 121.75, 119.50, 57.71, 45.35, 38.81, 28.06, 28.01, 25.77, 25.30, 17.35, 15.66 | ||
| 8m | 7.21(s, 2H, ArH), 5.02—5.15(m, 2H, C═CH), 4.02 and 3.69(2d, J=5.4, 5.9 Hz, 2H, CH2), 4.34 and 3.23(2t, J=14.1, 12.5 Hz, 2H, N—CH2), 2.00—2.17(m, 4H, CH2CH2), 1.72(s, 3H, CH3), 1.68(s, 3H, CH3), 1.61(s, 3H, CH3), 1.73—1.85 and 1.05—1.57(2m, 10H, CH2CH2CH2CH2CH2) | 166.07, 165.38, 150.60, 150.22, 149.99, 137.98, 136.96, 131.44, 131.02, 123.58, 123.28, 120.97, 119.93, 119.45, 58.71, 54.37, 53.13, 43.10, 39.49, 38.86, 30.17, 25.86, 25.43, 25.24, 17.30, 15.86, 15.58 | ||
| 8n | 7.24—7.30(m, 4H, ArH), 7.01—7.08(m, 2H, ArH), 7.02(d, J=6.9 Hz, 1H, ArH), 5.28—5.33(m, 1H, C═CH), 5.01—5.03(m, 1H, C═CH), 4.48(d, J=7.3 Hz, 2H, CH2), 1.98—2.07(m, 4H, CH2CH2), 1.68(s, 3H, CH3), 1.57(t, J=2.6 Hz, 3H, CH3), 1.50(s, 3H, CH3) | 164.52, 149.94, 148.83, 141.01, 140.73, 131.22, 129.25, 127.74, 127.67, 123.46, 121.54, 117.63, 47.63, 39.14, 25.91, 25.32, 17.32, 15.75 | ||
| 8o | 7.24—7.41(m, 7H, ArH), 5.06—5.30(m, 2H, C═CH), 4.70 and 4.37(2s, 2H, CH2), 4.11 and 3.67(2d, J=6.8, 6.5 Hz, 2H, CH2), 2.04—2.11(m, 4H, CH2CH2), 1.70(s, 3H, CH3), 1.63(s, 3H, CH3), 1.38(s, 3H, CH3) | 166.07, 150.59, 149.03, 141.27, 140.69, 135.99, 135.30, 131.60, 131.31, 128.66, 128.43, 128.01, 127.67, 127.46, 126.35, 123.52, 123.28, 120.06, 117.98, 117.61, 53.17, 51.03, 47.25, 45.83, 41.93, 39.04, 25.83, 25.37, 17.41, 15.82 | ||
| Compd. | Repellent activity(%) | Compd. | Repellent activity(%) | Compd. | Repellent activity(%) |
|---|---|---|---|---|---|
| 7a | 41.9±1.6 | 8d | 33.1±4.5 | 8l | 36.2±2.6 |
| 7b | 59.0±1.6 | 8e | 47.4±2.4 | 8m | 50.7±3.5 |
| 7c | 51.9±3.2 | 8f | 62.0±1.9 | 8n | 61.0±2.7 |
| 7d | 62.6±2.6 | 8g | 56.2±3.6 | 8o | 56.1±0.9 |
| 7e | 21.0±1.8 | 8h | 40.4±2.2 | EBF | 90.6±1.0 |
| 8a | 31.7±2.7 | 8i | 43.6±3.1 | 2b | 0.0±0.0 |
| 8b | 27.7±0.7 | 8j | 47.8±2.3 | 2d | 0.0±0.0 |
| 8c | 25.5±2.8 | 8k | 44.7±3.2 | 3 | 3.5±3.1 |
Table 4 Repellent activity of the target compounds against Myzus persicae(5 μg)
| Compd. | Repellent activity(%) | Compd. | Repellent activity(%) | Compd. | Repellent activity(%) |
|---|---|---|---|---|---|
| 7a | 41.9±1.6 | 8d | 33.1±4.5 | 8l | 36.2±2.6 |
| 7b | 59.0±1.6 | 8e | 47.4±2.4 | 8m | 50.7±3.5 |
| 7c | 51.9±3.2 | 8f | 62.0±1.9 | 8n | 61.0±2.7 |
| 7d | 62.6±2.6 | 8g | 56.2±3.6 | 8o | 56.1±0.9 |
| 7e | 21.0±1.8 | 8h | 40.4±2.2 | EBF | 90.6±1.0 |
| 8a | 31.7±2.7 | 8i | 43.6±3.1 | 2b | 0.0±0.0 |
| 8b | 27.7±0.7 | 8j | 47.8±2.3 | 2d | 0.0±0.0 |
| 8c | 25.5±2.8 | 8k | 44.7±3.2 | 3 | 3.5±3.1 |
| Compd. | Insecticidal activity(%) | Compd. | Insecticidal activity(%) | Compd. | Insecticidal activity(%) |
|---|---|---|---|---|---|
| 7a | 23.5±3.7 | 8d | 70.2±0.3 | 8l | 42.9±4.1 |
| 7b | 63.2±1.1 | 8e | 57.0±4.2 | 8m | 23.8±3.3 |
| 7c | 19.9±1.2 | 8f | 51.1±1.5 | 8n | 46.9±5.3 |
| 7d | 21.3±2.3 | 8g | 30.8±4.6 | 8o | 36.3±4.0 |
| 7e | 30.5±5.6 | 8h | 19.9±2.5 | EBF | 40.0±1.1 |
| 8a | 73.6±3.3 | 8i | 36.8±4.8 | Pymetrozine | 80.7±3.8 |
| 8b | 81.1±5.2 | 8j | 20.1±5.7 | ||
| 8c | 60.3±1.3 | 8k | 30.2±5.1 |
Table 5 Insecticidal activity of the target compounds against Myzus persicae(300 μg/mL)
| Compd. | Insecticidal activity(%) | Compd. | Insecticidal activity(%) | Compd. | Insecticidal activity(%) |
|---|---|---|---|---|---|
| 7a | 23.5±3.7 | 8d | 70.2±0.3 | 8l | 42.9±4.1 |
| 7b | 63.2±1.1 | 8e | 57.0±4.2 | 8m | 23.8±3.3 |
| 7c | 19.9±1.2 | 8f | 51.1±1.5 | 8n | 46.9±5.3 |
| 7d | 21.3±2.3 | 8g | 30.8±4.6 | 8o | 36.3±4.0 |
| 7e | 30.5±5.6 | 8h | 19.9±2.5 | EBF | 40.0±1.1 |
| 8a | 73.6±3.3 | 8i | 36.8±4.8 | Pymetrozine | 80.7±3.8 |
| 8b | 81.1±5.2 | 8j | 20.1±5.7 | ||
| 8c | 60.3±1.3 | 8k | 30.2±5.1 |
| [1] | Yang, Y. , Liu Y., X. , Song H., J. , Li Y., Q. , Wang Q., M. , Bioorg. Med. Chem., 2016, 24( 3), 391- 402 |
| [2] | 杨新玲, 黄文耀, 凌云, 阚伟, 方宇凌, 张钟宁. 高等学校化学学报, 2004, 25( 9), 1657- 1661 |
| Yang X., L. , Huang W., Y. , Ling, Y. , Kan, W. , Fang Y., L. , Zhang Z., N. , Chem. J. Chinese Universities, 2004, 25( 9), 1657- 1661 ( | |
| [3] | David, P. , Environ. Dev. Sustain., 2005, 7( 2), 229- 252 |
| [4] | 黄文耀, 杨新玲, 张钟宁. 化学通报, 2002, 3, 157- 161 |
| Huang W., Y. , Yang X., L. , Zhang Z., N. , Chemistry Bulletin, 2002, 3, 157- 161 ( | |
| [5] | 张钟宁, 刘珣, 梅雪琴. 动物学集刊, 1993, 10, 1- 3 |
| Zhang Z., N. , Liu, X. , Mei X., Q. , Animal Bulletin, 1993, 10, 1- 3 ( | |
| [6] | 闫凤鸣, 陈巨莲, 汤清波. 植物保护, 2013, 39( 5), 9- 15 |
| Yan F., M. , Chen J., L. , Tang Q., B. , Plant Protection, 2013, 39( 5), 9- 15 ( | |
| [7] | Kislow C., J. , Edward L., J. , Nature, 1972, 235( 5333), 108- 109 |
| [8] | Bowers W., S. , Nault L., R. , Webb R., E. , Dutky S., R. , Science, 1972, 175( 4026), 1121- 1122 |
| [9] | Edwards L., J. , Siddall J., B. , Dunham L., L. , Uden, P. , Kislow C., J. , Nature, 1973, 241( 5385), 126- 127 |
| [10] | Van O. A., M. , Gut, J. , Harrewijn, P. , Piron P. G., M. , Acta Phytopathol. Entomol. Hung., 1990, 25( 1-4), 331- 342 |
| [11] | CuiL., L. , Dong, J. , Francis, F. , Liu Y., J. , Heuskin, S. , Lognay, G. , Chen J., L. , Bragard, C. , Tooker J., F. , Liu, Y. , Crop Protection, 2012, 35, 91- 96 |
| [12] | Nishino, C. , Bowers W., S. , Montgomery M., E. , Nault L., R. , Appl. Entomol. Zool., 1976, 11( 4), 340- 343 |
| [13] | Dawson G., W. , Gibson R., W. , Griffiths D., C. , Pickett J., A. , Rice A., D. , Woodcock C., M. , J. Chem. Ecol., 1982, 8( 11), 1377- 1388 |
| [14] | 李正名, 王天生, 么恩云, 陈学仁, 朱兰蕙, 王素华. 化学学报, 1987, 45, 1124- 1128 |
| Li Z., M. , Wang T., S. , Me E., Y. , Chen X., R. , Zhu L., H. , Wang S., H. , Acta Chemical Sinica, 1987, 45, 1124- 1128 ( | |
| [15] | 张钟宁, 刘珣, Pickett John. 昆虫学报, 1988, 31( 4), 435- 438 |
| Zhang Z., N. , Liu, X. , Pickett, J. , Acta Entomol. Sinica, 1988, 31( 4), 435- 438 ( | |
| [16] | 孙玉凤, 李永强, 凌云, 宇红莲, 杨绍祥, 杨新玲. 有机化学, 2011, 31( 9), 1425- 1432 |
| Sun Y., F. , Li Y., Q. , Ling, Y. , Yu H., L. , Yang S., X. , Yang X., L. , Chin. J. Org. Chem., 2011, 31( 9), 1425- 1432 ( | |
| [17] | 秦耀果, 曲焱焱, 张景朋, 谭晓庆, 宋丽芳, 李文浩, 宋敦伦, 杨新玲. 有机化学, 2015, 35( 2), 455- 461 |
| Qin Y., G. , Qu Y., Y. , Zhang J., P. , Tan X., Q. , Song L., F. , Li W., H. , Song D., L. , Yang X., L. , Chin. J. Org. Chem., 2015, 35( 2), 455- 461 ( | |
| [18] | Song B. A., Jin L. H., New Heterocyclic Pesticide, Chemical Industry Press, Beijing, 2010, 118— 128 |
| ( 宋宝安, 金林红. 新杂环农药, 北京: 化学工业出版社, 2010, 118— 128) | |
| [19] | 张燕, 王宝雷, 詹益周, 张丽媛, 李永红. 李正名. 高等学校化学学报, 2016, 37( 6), 1100- 1107 |
| Zhang, Y. , Wang B., L. , Zhan Y., Z. , Zhang L., Y. , Li Y., H. , Li Z., M. , Chem. J. Chinese Universities, 2016, 37( 6), 1100- 1107 ( | |
| [20] | Gao, Y. , Wang B., W. , Gao, S. , Zhang R., H. , Yang C., Y. , Sun, Z. , Liu Z., H. , Chem. Res. Chinese Universities, 2016, 32( 4), 594- 599 |
| [21] | Tian Z., Z. , Zhang, D. , Guo, B. , Tian, G. , Liu X., X. , Yue H., J. , Chem. Res. Chinese Universities, 2015, 31( 2), 249- 252 |
| [22] | 王华森, 怀其勇. 天然产物研究与开发, 2013, 25( 2), 237- 240 |
| Wang H., S. , Huai Q., Y. , Nat. Prod. Res. Dev., 2013, 25( 2), 237- 240 ( | |
| [23] | Wild, N. , Groth, U. , Eur. J. Org. Chem., 2003, 2003( 22), 4445- 4449 |
| [24] | Peter, W. , John P., M. , Org. Lett., 2008, 10( 19), 4383- 4386 |
| [25] | Maciej, A. , Srinivasa R., A. , Rajarathnam E., R. , Tetrahedron, 2002, 58( 34), 6951- 6963 |
| [26] | 孙亮. EBF类似物CAU-1204的合成工艺及结构优化研究. 北京: 中国农业大学, 2013) |
| Sun, L. , Synthesis Process and Structure Optimization of EBF Analogue CAU- 1204, China Agricultural University, Beijing, 2013 ( | |
| [27] | Ravikumar P., C. , Yao L., H. , Fleming F., F. , J. Org. Chem., 2009, 74( 19), 7294- 7299 |
| [1] | WEI Simin,WANG Yinghui,TANG Zhishu,SU Rui,HU Jinhang,GUO Hui,LI Chen,JIANG Jintao,SONG Zhongxing. Novel Biosynthesis Method of Silver Nanoparticle by UV Radiation of Cornus Officinalis Aqueous Extract and Biological Activities † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1391. |
| [2] | GAO Qilong, LIANG Erjun, XING Xianran, CHEN Jun. Negative Thermal Expansion in Prussian Blue Analogues † [J]. Chem. J. Chinese Universities, 2020, 41(3): 388. |
| [3] | LI Yang, LI Zhiwen, ZHU Junfei, LIU Shihui, HE Junlin. Construction of Pyrenyl Pairs in dsDNA: Fluorescent Properties of Multiple Pyrenyl-attached dsDNAs Through 7-Substituted 8-Aza-7-deaza-2'-deoxyadenosine Analogues† [J]. Chem. J. Chinese Universities, 2018, 39(10): 2206. |
| [4] | SHI Yujun, FANG Yuan, LI Yang, CHEN Jia, LI Gang, WANG Qingmin, DAI Hong. Synthesis and Biological Activities of Novel Cyanoacrylate Derivatives Carrying 5-Arylisoxazole Group† [J]. Chem. J. Chinese Universities, 2017, 38(3): 421. |
| [5] | ZHANG Lingzhi, JIANG Minrui, WEI Ping, ZHU Qihua, GONG Guoqing, BIAN Xueguo, XU Yungen. Synthesis, Metabolic Stability and Biological Activity in vivo of Lorcaserin Derivatives† [J]. Chem. J. Chinese Universities, 2017, 38(10): 1778. |
| [6] | BAI Xinfa, MA Xuan, XIE Xiaoxia, SHAO Mingsha, GUO Ningning, YAN Ning, YAO Lei. Synthesis and Anti-tumor Activity of Tubulysins Analogues† [J]. Chem. J. Chinese Universities, 2017, 38(1): 47. |
| [7] | ZHANG Yan, WANG Baolei, ZHAN Yizhou, ZHANG Liyuan, LI Yonghong, LI Zhengming. Synthesis and Biological Activities of Novel 5-(Pyridine-3-yl)-1,2,4-triazole Mannich Bases and Bis-Mannich Bases Containing Piperazine Moiety† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1100. |
| [8] | XU Gaofei, LIU Yanhong, YANG Xinling, WANG Daoquan, YUAN Dekai. Synthesis and Biological Activity of Novel Dicyano-containning Cyclopropane-1-carboxamides† [J]. Chem. J. Chinese Universities, 2016, 37(3): 486. |
| [9] | WANG Zijian, SUN Xiaohong, LIU Yuanfa, CHEN Bang, SHEN Shengqiang, JIN Ruyi, MA Haixia. Synthesis and Biological Activities of 1,2,4-Triazole Schiff Bases Containing Pyrazole Rings† [J]. Chem. J. Chinese Universities, 2015, 36(7): 1315. |
| [10] | LI Xu, JIANG Jianhong, HAN Buxing, GU Huiwen, XIE Zhaofeng, CHEN Lan, XIAO Shengxiong, LI Chuanhua, LI Aitao, LI Xia, YAO Feihong, WANG Qun, LI Qiangguo. Synthesis and Biological Activities of o-Vanillin-histidine Schiff-base and Its Lanthanum Complex† [J]. Chem. J. Chinese Universities, 2015, 36(5): 856. |
| [11] | SHEN Shengqiang, SUN Xiaohong, LIU Yuanfa, CHEN Bang, JIN Ruyi, MA Haixia. Synthesis and Biological Activity of 1,3,4-Oxadiazole Mannich Bases Containing Pyrimidine Rings† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1427. |
| [12] | LIU Shaohua, LING Yun, LI Hongyue, XU Dan, YANG Xinling. Design, Synthesis and Biological Activity of Novel Substituted Phenyl Oxazol Based Compounds† [J]. Chem. J. Chinese Universities, 2014, 35(2): 281. |
| [13] | WANG Xin-Zhong, LIU Jian-Guo, TANG Yu-Feng, WANG Yu, ZHANG Pei-Biao, CHEN Xue-Si. Preparation of Controlled-release PLA Microspheres Loading Methylsulfonylmethane and Its Biological Activity [J]. Chem. J. Chinese Universities, 2013, 34(4): 984. |
| [14] | JI Xu, BIAN Liu-Jiao. Bioactive Change and Structural Change of Bacillus Amyloliquefaciens α-Amylases Induced by Ca2+ [J]. Chem. J. Chinese Universities, 2013, 34(11): 2517. |
| [15] | LI Zuo-Sheng, ZOU Lu-Yi, REN Ai-Min, FENG Ji-Kang. Theoretical Study of the Electronic Spectral Property of Firefly Oxyluciferin Analogues [J]. Chem. J. Chinese Universities, 2012, 33(12): 2757. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||