

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (7): 1321.doi: 10.7503/cjcu20141107
• Organic Chemistry • Previous Articles Next Articles
WANG Xinxin1, LÜ Xia3, GE Guangbo3, FENG Lei3, XIN Hong3, LI Yaoguang3, CAO Yunfeng3, HAN Guanying2, GUO Bin2,*( )
)
Received:2014-12-18
Online:2015-07-10
Published:2015-05-29
Contact:
GUO Bin
E-mail:jyguobin@126.com
Supported by:CLC Number:
TrendMD:
WANG Xinxin, LÜ Xia, GE Guangbo, FENG Lei, XIN Hong, LI Yaoguang, CAO Yunfeng, HAN Guanying, GUO Bin. Highly Efficient Preparation of Resorufin-β-D-glucuronide†[J]. Chem. J. Chinese Universities, 2015, 36(7): 1321.
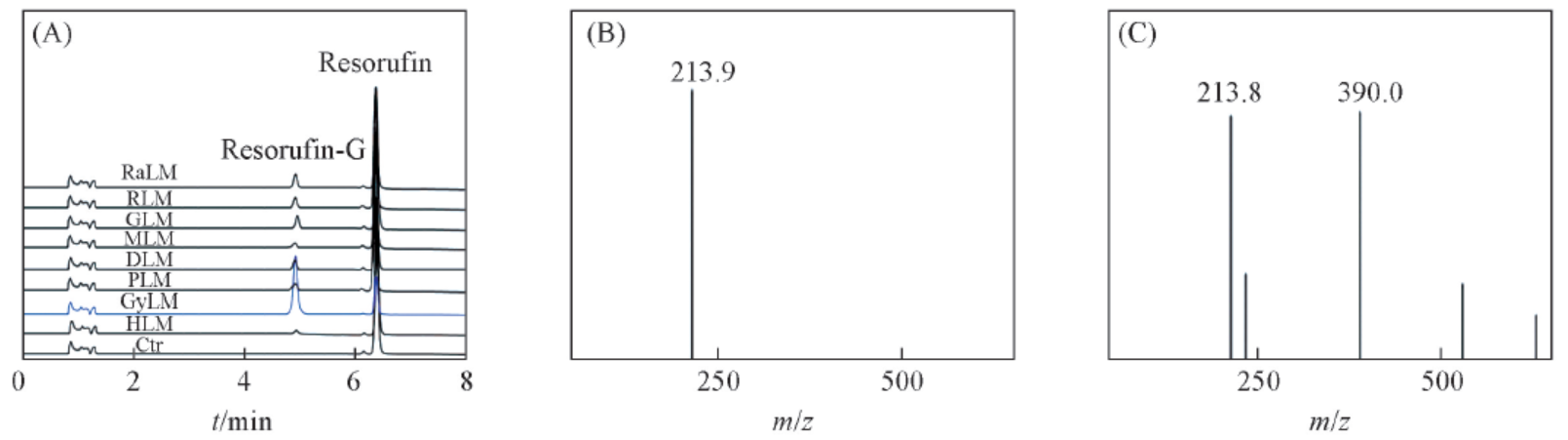
Fig.1 HPLC-UV and MS spectra of resorufin and resorufin β-D-glucuronide(A) HPLC-UV profiles of resorufin and its metabolite in liver microsomes from different species; (B) MS spectrum of resorufin;(C) MS spectrum of resorufin β-D-glucuronide.
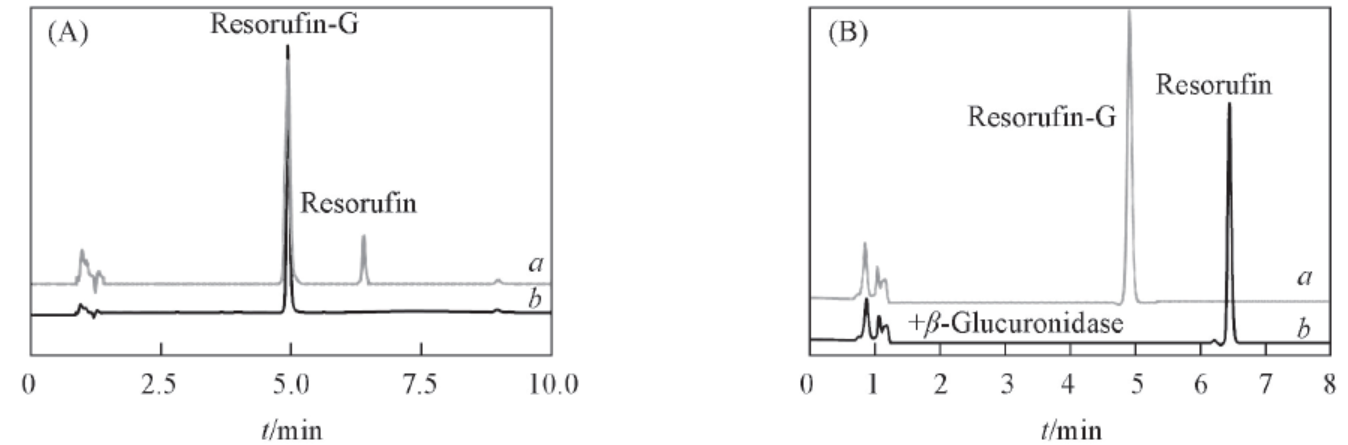
Fig.2 LC-UV chromatography of REG before and after purification(A) and hydrolysis(B)(A) LC-UV chromatography of the reaction mixture(a) and the aim metabolite after isolation and purification by SPE(b); (B) LC-UV profiles of REG(100 μmol/L) before(a) and REG(100 μmol/L) was incubated with β-glucosidase(1030 U/mL, complete hydrolysis) in at 37 ℃ for 15 min(b).
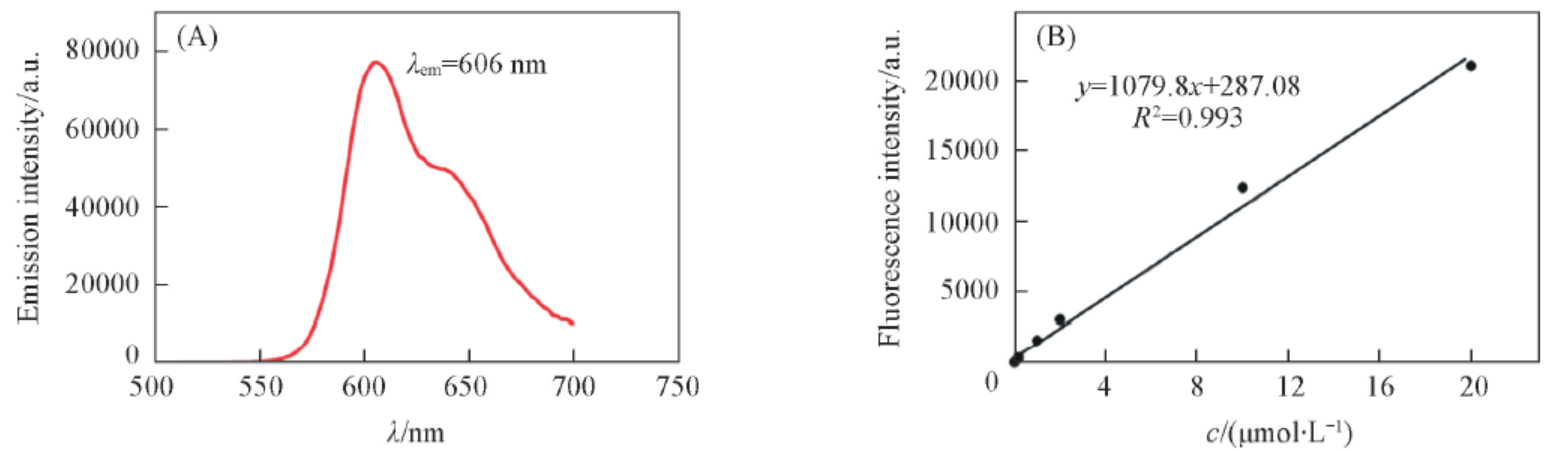
Fig.4 Emission spectrum(A) and calibration curve(B) of resorufin(A) Emission spectrum of REG after hydrolysis with β-glucosidase, the excitation wavelength was set at 500 nm; (B) resorufin calibration curve was obtained with a range of resorufin concentration, (0—20 μmol/L) in an equal volume of acetonitrile and PBS(total volume 400 μL), the excitation and emission wavelength were set at 577 and 620 nm, respectively.
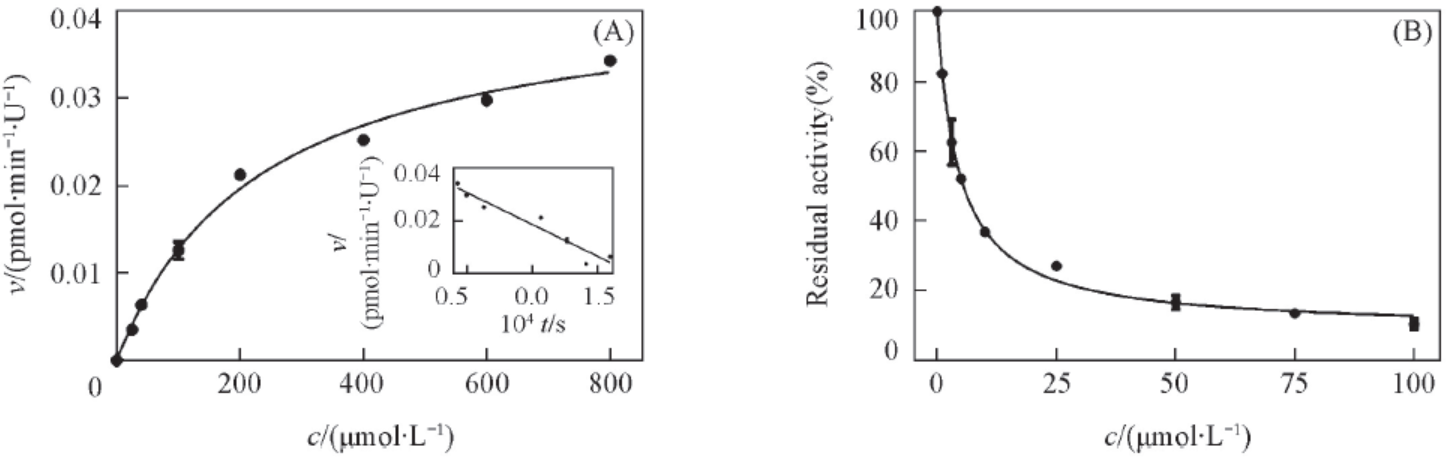
Fig.5 Enzymatic kinetic and inhibition effect of REG hydrolysis in β-glucosidase(A) Enzymatic kinetic of REG hydrolysis in β-glucosidase, the abscissa is the concentration of resorufin glucuronide, the ordinate is the generation rate of resorufin. Eadie-Hofstee plot is shown as inset. Each point represents the average of two independent experiments in duplicate; (B) inhibitory effect of D-saccharic acid 1,4-lactone towards β-glucosidase with resorufin as the substrate IC50 value was obtained by fitting data to IC50 equation of GraphPad Prism 6.0.
| [1] | Kowalczyk M. C., Spears E., Narog M., Zoltaszek R., Kowalczyk P., Hanausek M., Yoshimi N., Slaga T. J., Walaszek Z., Oncol. Rep., 2011, 26(3), 551—556 |
| [2] | Beaud D., Tailliez P., Anba-Mondoloni J., Microbiol-Sgm., 2005, 151, 2323—2330 |
| [3] | Matsuura T., Hosoda K., Ichihashi N., Kazuta Y., Yomo T., J. Biol. Chem., 2011, 286(25), 22028—22034 |
| [4] | Hanioka N., Ozawa S., Jinno H., Ando M., Saito Y., Sawada J., Xenobiotica, 2001, 31(10), 687—699 |
| [5] | Stringer A. M., Gibson R. J., Logan R. M., Bowen J. M., Yeoh A. S. J., Keefe D. M. K., Cancer Biol. Ther., 2008, 7(12), 1919—1925 |
| [6] | Ahmad S., Hughes M. A., Lane K. T., Redinbo M. R., Yeh L. A., Scott J. E., Current Chemical Genomics, 2011, 5, 13—20 |
| [7] | Chen X., Yu T., Chen Z., Zhao R., Mao S., Xenobiotica, 2014, 44(9), 785—91 |
| [8] | Schroeder C., Lutterbach R., Stöckigt J., Tetrahedron, 1996, 52(3), 925—934 |
| [9] | Zhao L., Huimin M., Chem. J. Imaging Science and Photochemistry, 2014, 32(1), 60—68 |
| (李照, 马会民. 影像科学与光化学,2014, 32(1), 60—68) | |
| [10] | Motabar O., Shi Z. D., Goldin E., Liu K., Southall N., Sidransky E., Austin C. P., Griffiths G. L., Zheng W., Anal. Biochem., 2009, 390(1), 79—84 |
| [11] | Hofmann J., Sernetz M., Anal. Chim. Acta, 1984, 163, 67—72 |
| [12] | Gao W., Xing B., Tsien R.Y., Rao J., J. Am. Chem. Soc., 2003, 125(37), 11146—11147 |
| [13] | Albers A. E., Rawls K. A., Chang C. J., Chem. Commun., 2007, 44, 4647—4649 |
| [14] | Lavis L. D., Chao T. Y., Raines R. T., Chem. Sci., 2011, 2(3), 521—530 |
| [15] | Hanif M., Khan I., Rama N. H., Noreen S., Choudhary M. I., Jones P. G., Iqbal M., Med. Chem. Res., 2012, 21(11), 3885—3896 |
| [16] | Schroeder C., Lutterbach R., Stöckigt J., Tetrahedron, 1996, 52(3), 925—934 |
| [17] | Wimmer Z., Pechová L., Saman D., Molecules, 2004, 9(11), 902—912 |
| [18] | Reiss P., Burnett D. A., Zaks A., Bioorg. Med. Chem., 1999, 7(10), 2199—2202 |
| [19] | Chen X., Yu T., Chen Z., Zhao R., Mao S., Xenobiotica, 2014, 44(9), 785—91 |
| [20] | Tian M. L., Lee Y. R., Park D. W., Row K. H., Chem. Res. Chinese Universities, 2013, 29(4), 663—666 |
| [21] | Yang G., Zhu L. L., Lü X., Wu D. C., Xia Y. L., Ma X. C., Xin Y., Hou J., Chem. J. Chinese Universities, 2014,35(2), 314—318 |
| (杨刚, 朱亮亮, 吕侠, 吴大畅, 夏杨柳, 马骁驰, 辛毅, 侯洁. 高等学校化学学报,2014, 35(2), 314—318) | |
| [22] | Rao W., Yin Y. L., Long F., Zhang C. H., Chem. J. Chinese Universities, 2015, 36(3), 449—455 |
| (饶维, 尹玉立, 龙芳, 张朝辉. 高等学校化学学报,2015, 36(3), 449—455) | |
| [23] | Liebherr R. B., Renner M., Gorris H. H., J. Am. Chem. Soc., 2014, 136(16), 5949—5955 |
| [24] | Magro G., Bain R. E. S., Woodall C. A., Matthews R. L., Gundry S. W., Davis A. P., Environ. Sci. Technol., 2014, 48(16), 9624—9631 |
| [25] | Zhou M. J., Diwu Z. J., Panchuk Voloshina N., Haugland R. P., Anal. Biochem., 1997, 253(2), 162—168 |
| [26] | Narita M., Nagai E., Hagiwara H., Aburada M., Yokoi T., Kamataki T., Xenobiotica, 1993, 23(1), 5—10 |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||