

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (6): 1126.doi: 10.7503/cjcu20150130
• Physical Chemistry • Previous Articles Next Articles
CHENG Shangzeng1,2, GUO Qianqian1,2, HUANG Zhanggen1,*( ), SHEN Wenzhong1, HAN Xiaojin1
), SHEN Wenzhong1, HAN Xiaojin1
Received:2015-02-05
Online:2015-06-10
Published:2015-04-28
Contact:
HUANG Zhanggen
E-mail:zghuang@sxicc.ac.cn
Supported by:CLC Number:
TrendMD:
CHENG Shangzeng, GUO Qianqian, HUANG Zhanggen, SHEN Wenzhong, HAN Xiaojin. Cellulose Generated Carbon Materials with Nitrogen Doping for the Desulfurization of Flue Gas†[J]. Chem. J. Chinese Universities, 2015, 36(6): 1126.
| Sample | BET surface area, SBET/(m2·g-1) | Total pore volume, Vtotal/(cm3·g-1) | Micropore volume, Vmicro/(cm3·g-1) | Micropore surface area, Smicro/(m2·g-1) |
|---|---|---|---|---|
| AC-800-3-2 | 1289 | 0.58 | 0.52 | 1024 |
| AC-700-3-2 | 858 | 0.36 | 0.30 | 748 |
| AC-600-3-2 | 518 | 0.25 | 0.21 | 463 |
| AC-600-5-2 | 753 | 0.36 | 0.31 | 684 |
| AC-600-7-2 | 801 | 0.43 | 0.36 | 752 |
| AC-600-3-1 | 454 | 0.19 | 0.17 | 407 |
| AC-600-0-2* | 371 | 0.22 | 0.15 | 275 |
Table 1 Structural parameter of samples
| Sample | BET surface area, SBET/(m2·g-1) | Total pore volume, Vtotal/(cm3·g-1) | Micropore volume, Vmicro/(cm3·g-1) | Micropore surface area, Smicro/(m2·g-1) |
|---|---|---|---|---|
| AC-800-3-2 | 1289 | 0.58 | 0.52 | 1024 |
| AC-700-3-2 | 858 | 0.36 | 0.30 | 748 |
| AC-600-3-2 | 518 | 0.25 | 0.21 | 463 |
| AC-600-5-2 | 753 | 0.36 | 0.31 | 684 |
| AC-600-7-2 | 801 | 0.43 | 0.36 | 752 |
| AC-600-3-1 | 454 | 0.19 | 0.17 | 407 |
| AC-600-0-2* | 371 | 0.22 | 0.15 | 275 |
| Sample | Ca(%) | Na(%) | Oa,b(%) | Ha(%) |
|---|---|---|---|---|
| AC-800-3-2 | 84.80 | 4.45 | 9.66 | 0.31 |
| AC-700-3-2 | 80.45 | 5.31 | 11.03 | 0.61 |
| AC-600-3-2 | 78.30 | 5.97 | 13.85 | 0.61 |
| AC-600-5-2 | 75.02 | 8.13 | 14.80 | 0.82 |
| AC-600-7-2 | 66.54 | 15.07 | 16.11 | 0.57 |
| AC-600-3-1 | 77.40 | 5.56 | 15.32 | 0.88 |
| AC-600-0-2 | 89.59 | 0.35 | 9.61 | 0.70 |
Table 2 Elemental composition of samples
| Sample | Ca(%) | Na(%) | Oa,b(%) | Ha(%) |
|---|---|---|---|---|
| AC-800-3-2 | 84.80 | 4.45 | 9.66 | 0.31 |
| AC-700-3-2 | 80.45 | 5.31 | 11.03 | 0.61 |
| AC-600-3-2 | 78.30 | 5.97 | 13.85 | 0.61 |
| AC-600-5-2 | 75.02 | 8.13 | 14.80 | 0.82 |
| AC-600-7-2 | 66.54 | 15.07 | 16.11 | 0.57 |
| AC-600-3-1 | 77.40 | 5.56 | 15.32 | 0.88 |
| AC-600-0-2 | 89.59 | 0.35 | 9.61 | 0.70 |
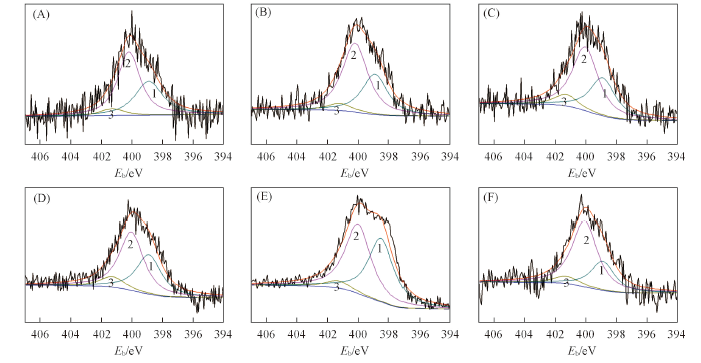
Fig.2 XPS N1s spectra of samples(A) AC-800-3-2; (B) AC-700-3-2; (C) AC-600-3-2; (D) AC-600-5-2; (E) AC-600-7-2; (F) AC-600-3-1.Peak 1: N-6; peak 2: N-5; peak 3: N-Q. 1, 2 and 3 are deconvoluted peaks.
| Sample | Content of nitrogen(%, molar fraction) | Distribution of nitrogen-containing groups(%) | Content of N-6b (%, molar fraction) | |||
|---|---|---|---|---|---|---|
| Bulk | Surface | N-6a | N-5a | N-Qa | ||
| AC-800-3-2 | 3.83 | 3.66 | 37.42 | 57.11 | 5.47 | 1.37 |
| AC-700-3-2 | 4.52 | 4.75 | 34.28 | 59.06 | 6.66 | 1.54 |
| AC-600-3-2 | 5.06 | 5.78 | 30.67 | 60.98 | 8.35 | 1.77 |
| AC-600-5-2 | 6.77 | 7.29 | 37.42 | 56.38 | 6.20 | 3.04 |
| AC-600-7-2 | 13.13 | 12.37 | 45.37 | 49.57 | 5.06 | 5.61 |
| AC-600-3-1 | 4.57 | 4.56 | 27.97 | 67.03 | 5.00 | 1.28 |
Table 3 Distribution of surface nitrogen species obtained by XPS analysis
| Sample | Content of nitrogen(%, molar fraction) | Distribution of nitrogen-containing groups(%) | Content of N-6b (%, molar fraction) | |||
|---|---|---|---|---|---|---|
| Bulk | Surface | N-6a | N-5a | N-Qa | ||
| AC-800-3-2 | 3.83 | 3.66 | 37.42 | 57.11 | 5.47 | 1.37 |
| AC-700-3-2 | 4.52 | 4.75 | 34.28 | 59.06 | 6.66 | 1.54 |
| AC-600-3-2 | 5.06 | 5.78 | 30.67 | 60.98 | 8.35 | 1.77 |
| AC-600-5-2 | 6.77 | 7.29 | 37.42 | 56.38 | 6.20 | 3.04 |
| AC-600-7-2 | 13.13 | 12.37 | 45.37 | 49.57 | 5.06 | 5.61 |
| AC-600-3-1 | 4.57 | 4.56 | 27.97 | 67.03 | 5.00 | 1.28 |
| Sample | Adsorption capacity under different atmosphere/(mg·g-1) | Ratio of adsorption capacity under different atmosphere | |
|---|---|---|---|
| SO2/N2 | SO2+O2/N2 | ||
| AC-600-0-2 | 2.05 | ||
| AC-600-3-2 | 9.38 | 14.44 | 1.54 |
| AC-600-5-2 | 10.64 | 21.87 | 2.06 |
| AC-600-7-2 | 15.42 | 36.08 | 2.34 |
Table 4 SO2 adsorption capacity of samples under different atmosphere
| Sample | Adsorption capacity under different atmosphere/(mg·g-1) | Ratio of adsorption capacity under different atmosphere | |
|---|---|---|---|
| SO2/N2 | SO2+O2/N2 | ||
| AC-600-0-2 | 2.05 | ||
| AC-600-3-2 | 9.38 | 14.44 | 1.54 |
| AC-600-5-2 | 10.64 | 21.87 | 2.06 |
| AC-600-7-2 | 15.42 | 36.08 | 2.34 |
| [1] | Sun F., Gao J. H., Zhu Y. W., Chen G. Q., Wu S. H., Qin Y. K., Adsorpt. J. Int. Adsorpt. Soc., 2013, 19(5), 959-966 |
| [2] | Zhu Y. W., Gao J. H., Li Y., Sun F., Qin Y. K., Korean J. Chem. Eng., 2011, 28(12), 2344-2350 |
| [3] | Sun F., Gao J. H., Zhu Y. W., Qin Y. K., Korean J. Chem. Eng., 2011, 28(11), 2218-2225 |
| [4] | Rubio B., Izquierdo M. T., Waste Management, 2010, 30(7), 1341-1347 |
| [5] | Shen W., Hu T., Wang P., Sun H., Fan W., Chem. Plus. Chem., 2014, 79(2), 284-289 |
| [6] | Mangun C. L., DeBarr J. A., Economy J., Carbon, 2001, 39(11), 1689-1696 |
| [7] | Li K. X., Ling L. C., Liu L., Zhang B. J., Liu Z. Y., J. Environ. Sci., 2001, 21(1), 74-78 |
| (李开喜, 凌立成, 刘朗, 张碧江, 刘振宇.环境科学学报, 2001,21(1), 74-78) | |
| [8] | Boudou J. P., Chehimi M., Broniek E., Siemieniewska T., Bimer J., Carbon, 2003, 41(10), 1999-2007 |
| [9] | Zhang X., Zhang Y., Wang S., Zhang J., Zhou W., Sci. China Technol. Sci., 2010, 53(9), 2515-2520 |
| [10] | Yang G., Chen H., Qin H., Feng Y., Appl. Surf. Sci., 2014, 293(0), 299-305 |
| [11] | Zhao W., Fierro V., Fernández-Huerta N., Izquierdo M., Celzard A., Int. J. Hydrogen Energy, 2013, 38(25), 10453-10460 |
| [12] | Raymundo-Piñero E., Cazorla-Amorós D., Linares-Solano A., Find J., Wild U., Schlögl R., Carbon, 2002, 40(4), 597-608 |
| [13] | Shen W., Fan W., J. Mater. Chem. A, 2013, 1(4), 999-1013 |
| [14] | Zhu M., Wang J., Holloway B. C., Outlaw R. A., Zhao X., Hou K., Shutthanandan V., Manos D. M., Carbon, 2007, 45(11), 2229-2234 |
| [15] | Wen Q., Wang S., Yan J., Cong L., Chen Y., Xi H., Bioelectrochemistry, 2014, 95, 23-28 |
| [16] | Shen W., Hu T., Fan W., RSC Advances, 2014, 4(18), 9126-9132 |
| [17] | Seredych M., Bandosz T. J., Energy & Fuels, 2008, 22(2), 850-859 |
| [18] | Biniak S., Szymański G., Siedlewski J.,wiᶏtkowski A., Carbon, 1997, 35(12),1799-1810 |
| [19] | Kapteijn F., Moulijn J. A., Matzner S., Boehm H. P., Carbon, 1999, 37(7), 1143-1150 |
| [20] | Pietrzak R., Wachowska H., Nowicki P., Energy Fuels, 2006, 20(3), 1275-1280 |
| [21] | Li J., Wang X. B., Yang J., Yang X. Y., Wan L., Chem. J. Chinese Universities, 2013, 34(4), 800-805 |
| (李静, 王贤保, 杨佳, 杨旭宇, 万丽. 高等学校化学学报 , 2013, 34(4), 800-805) | |
| [22] | Mao W. K., Lu Z. Z., Tu Q. X., Tang Y. X., Hua J., Yin H. Q., Chinese J. Environ. Eng., 2011, 5(4), 893-897 |
| (毛炜坤, 鲁贞真, 涂秦霞, 汤尧旭, 华坚, 尹华强.环境工程学报, 2011,5(4), 893-897) | |
| [23] | Raymundo-Piñero E., Cazorla-Amorós D., Linares-Solano A., Carbon, 2003, 41(10), 1925-1932 |
| [24] | Raymundo-Piñero E., Cazorla-Amorós D., Linares-Solano A., Carbon, 2001, 39(2), 231-242 |
| [25] | Xu L., Guo J., Jin F., Zeng H., Chemosphere, 2006, 62(5), 823-826 |
| [26] | Strelko V. V., Kuts V. S., Thrower P. A., Carbon, 2000, 38(10), 1499-1503 |
| [27] | Stöhr B., Boehm H. P., Schlögl R., Carbon, 1991, 29(6), 707-720 |
| [1] | YANG Lijun, YU Yang, ZHANG Lei. Construction of Dual-functional 2D/3D Hydrid Co2P-CeO x Heterostructure Integrated Electrode for Electrocatalytic Urea Oxidation Assisted Hydrogen Production [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220082. |
| [2] | MENG Xianglong, YANG Ge, GUO Hailing, LIU Chenguang, CHAI Yongming, WANG Chunzheng, GUO Yongmei. Synthesis of Nano-zeolite and Its Adsorption Performance for Hydrogen Sulfide [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210687. |
| [3] | SUN Jinshi, CHEN Peng, JING Liping, SUN Fuxing, LIU Jia. Synthesis of Hierarchical Porous Aromatic Frameworks for Immobilization of Thiourea Catalyst [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220171. |
| [4] | MA Yukun, JIN Hui, REN Chuanli, LI Zhibo. Ring-opening Polymerization of Cyclic Esters Using Recyclable Polystyrene Supported Urea-Base Binary Catalyst [J]. Chem. J. Chinese Universities, 2021, 42(9): 2968. |
| [5] | ZHANG Renli, WANG Yao, YU Zhiquan, SUN Zhichao, WANG Anjie, LIU Yingya. Molybdenum Peroxide Anchored on Fluoronated UiO-66 as Catalyst in the Oxidation of Sulfur Containing Compounds [J]. Chem. J. Chinese Universities, 2021, 42(6): 1914. |
| [6] | FU Jinzhou, WANG Hanwei, LI Yingying, WANG Chao, LI Caicai, SUN Qingfeng, LI Huiqiao. Micro/Nanocellulose Functional Membranes for Energy and Environment [J]. Chem. J. Chinese Universities, 2021, 42(5): 1407. |
| [7] | ZHAO Ziyi,ZHENG Hongzhi,XU Yan. Multi-color Circularly Polarized Luminescence Properties of Cellulose Nanocrystal † [J]. Chem. J. Chinese Universities, 2020, 41(5): 1120. |
| [8] | HAO Yan, YANG Hua, WANG Xiang, LI Qingyang, ZHAO Pan, TANG Qinghu, SONG Shili, XI Guoxi. Palladium-based Nanocatalysts Supported on Polybenzoxazine for Aromatic Alcohol Oxidation † [J]. Chem. J. Chinese Universities, 2020, 41(4): 757. |
| [9] | KONG Jinfeng, ZHU Yuzhang, JIN Jian. Sulfonated Cellulose Nanofibers Film Supported Nanofiltration Membrane for High-flux and High-rejection Desalination † [J]. Chem. J. Chinese Universities, 2020, 41(4): 690. |
| [10] | WANG Xiao,JIN Biao,WANG Yubin,XU Zhuoyue,ZHANG Lu,ZHANG Xiaoting,YANG Liushuan. Interaction Mechanism of Anions in Hydrothermal Crystallization of Desulfurization Gypsum Whiskers † [J]. Chem. J. Chinese Universities, 2020, 41(3): 473. |
| [11] | ZHANG Li,QIAN Mingchao,LIU Xueke,Gao Shuaitao,YU Jiang,XIE Haishen,WANG Hongbin,SUN Fengjiang,SU Xianghong. Dynamic Study of Oxidative Desulfurization by Iron-based Ionic Liquids/NHD † [J]. Chem. J. Chinese Universities, 2020, 41(2): 317. |
| [12] | WU Rong, DONG Qihui, SUN Yiyi, SU Erzheng. Efficient Enzyme Immobilization by Combining Adsorption and Cellulose Membrane Coating † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1888. |
| [13] | HU Xueyi, CHEN Miaomiao, FANG Yun, FENG Ruiqin, HAN Huihui. Investigation on Pseudo-polyanions of Cationic Cellulose-Sodium Dodecylbenzenesulfonate† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1464. |
| [14] | GONG Minghui,YANG Shanshan,LI Shusheng,KUANG Rui,KONG Xiangzheng. Preparation of Fluorescence Polyurea Microspheres Through Precipitation Polymerization and Their Use for Fe3+ Determination† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1317. |
| [15] | WANG Ying, SUN Chuanyin, WANG Runwei, ZHANG Zhendong, ZHANG Daming, ZHANG Zongtao, QIU Shilun. Preparation of Amphiphilic Ti/ZSM-5 Zeolite and Its Catalytic Performance in Oxidative Desulfurization† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1265. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||