

Chem. J. Chinese Universities ›› 2022, Vol. 43 ›› Issue (3): 20210701.doi: 10.7503/cjcu20210701
• Articles: Inorganic Chemistry • Previous Articles Next Articles
LI Hua1,2, YANG Ke1, HUANG Junfeng1, CHEN Fengjuan1( )
)
Received:2021-10-04
Online:2022-03-10
Published:2021-12-28
Contact:
CHEN Fengjuan
E-mail:chenfj@lzu.edu.cn
Supported by:CLC Number:
TrendMD:
LI Hua, YANG Ke, HUANG Junfeng, CHEN Fengjuan. Design and Construction of UiO-66-NH2/wood Composite for Efficient Removal of Trace Heavy Metal Ions from Water[J]. Chem. J. Chinese Universities, 2022, 43(3): 20210701.
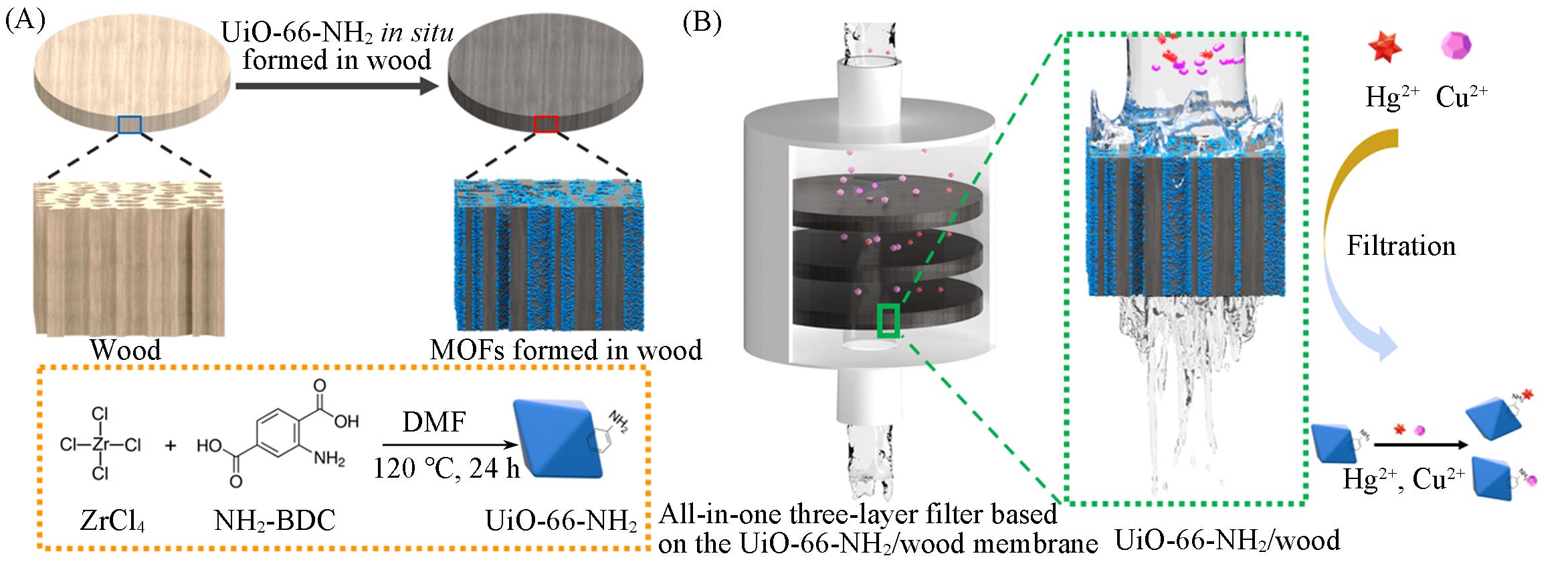
Scheme 1 Schematic illustration of in situ synthesis UiO?66?NH2 MOFs in 3D wood lumens and the all?in?one device for efficient trace heavy metal ions removal

Fig.1 Characterizations of natural wood and UiO?66?NH2/wood membrane(A) Low magnified SEM image of UiO?66?NH2/wood membrane filter; (B) high magnified SEM image of UiO?66/wood membrane; (C) SEM image of the UiO?66 MOF nanoparticles anchored in the wood microchannels; (D) high magnified SEM image of the UiO?66 MOF nanoparticles anchored in the wood microchannels.
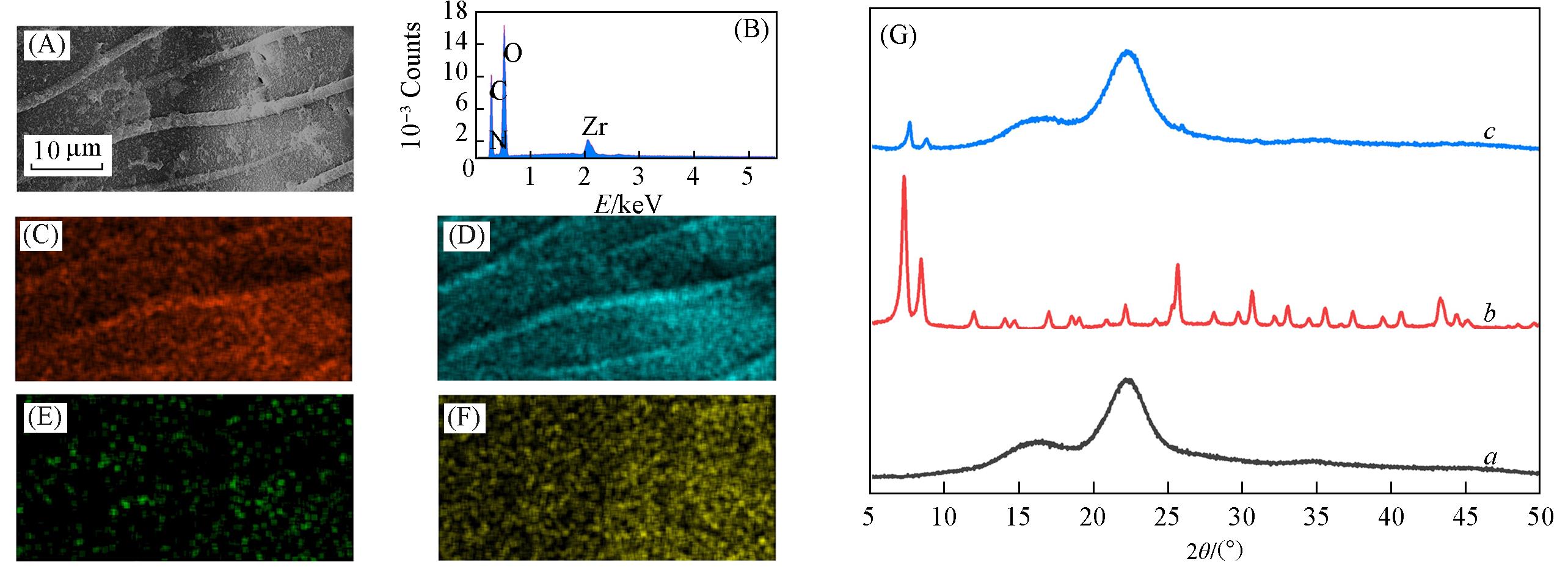
Fig.2 SEM image(A), EDS spectrum(B) and corresponding elemental mapping images of C(C),O(D), N(E) and Zr(F) of UiO?66?NHz/wood membrane and XRD patterns of the wood(a), UiO?66?NH2(b) and UiO?66?NH2/wood membrane(c)(G)
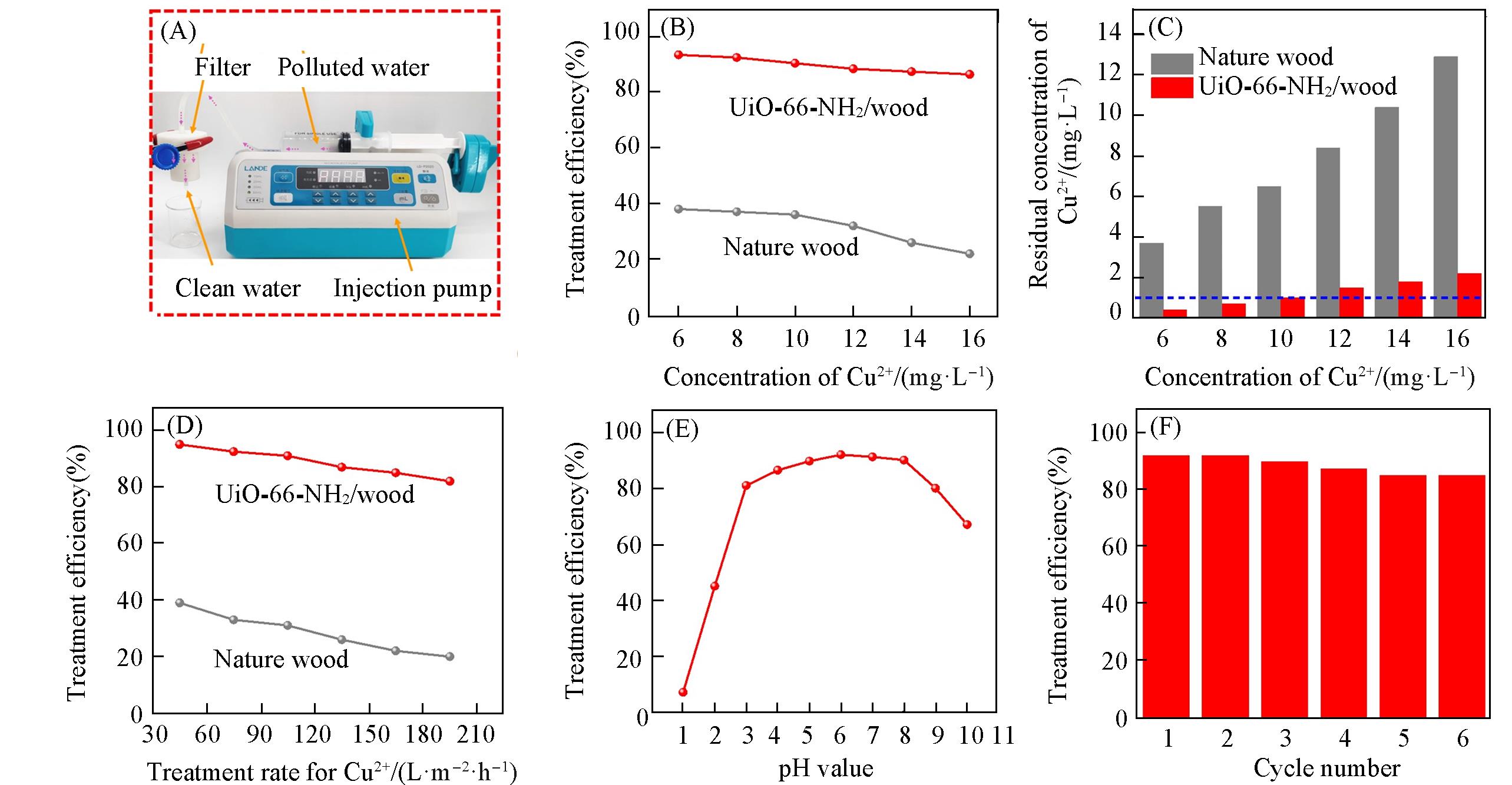
Fig.3 Removal performance for Cu2+ of the all?in?one three?layer filter based on the UiO?66?NH2/wood membrane(A) Photographs of the experimental setup for trace heavy metal Ions removal from water by using the all?in?one three?layer filter; treatment efficiency of natural wood and UiO?66?NH2/wood membrane filter for different concentrations of Cu2+ aqueous solution(flow rate of Cu2+ aqueous solution: 1.1×102 L?m-2?h-1, B) and comparison of the residual Cu2+ concentration in the filtered water with the national maximum allowable concentration(C); (D) treatment efficiency of natural wood and UiO?66?NH2/wood membrane at different flow rates(the initial concentration of Cu2+ aqueous solution: 10 mg/L); (E) treatment efficiency of the UiO?66?NH2/wood membrane at different pH; (F) treatment efficiency of UiO?66?NH2/wood membrane verse different cycles of regeneration.
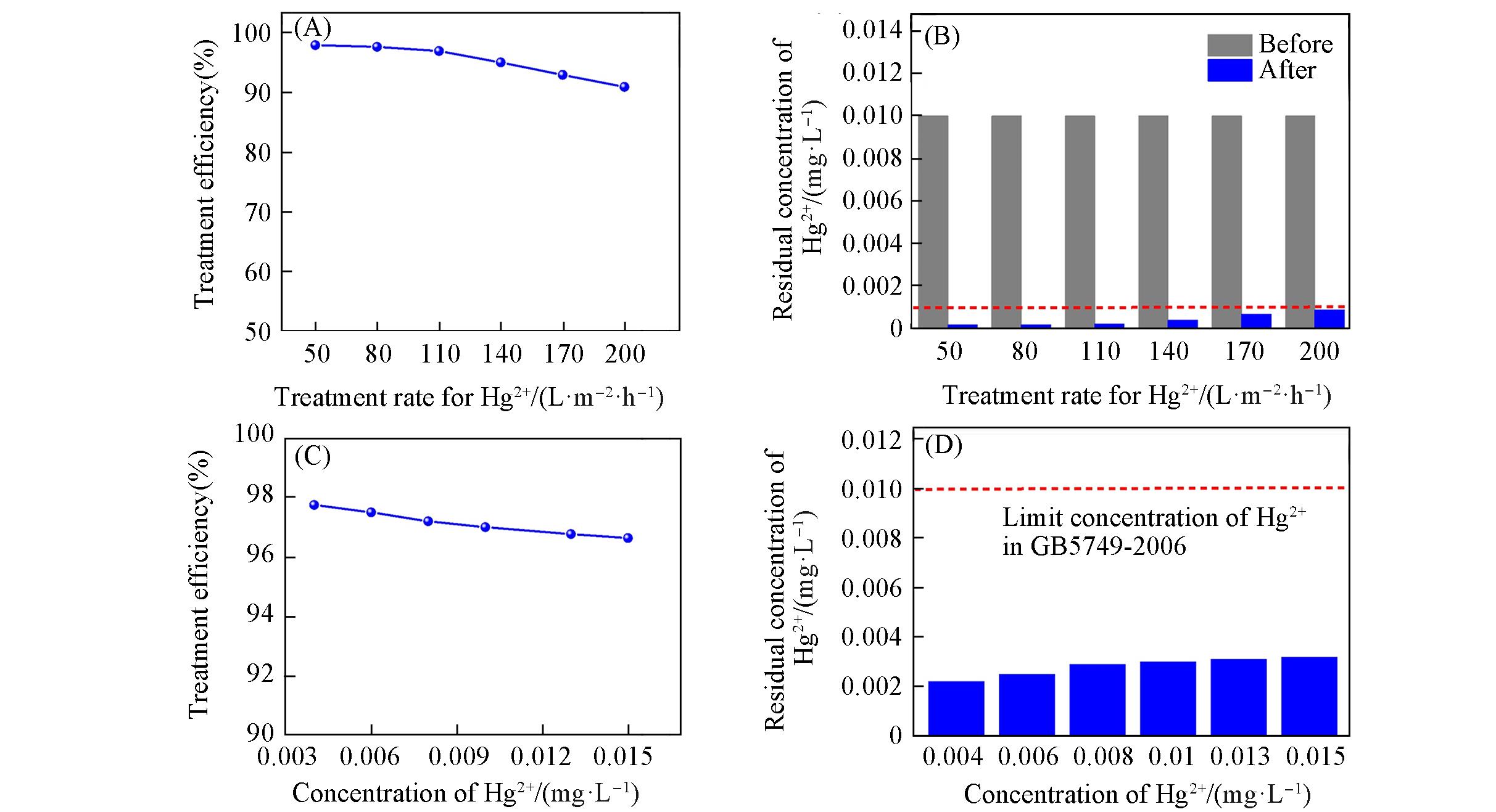
Fig.4 Hg2+ removal performance of the UiO?66?NH2/wood membrane filter(A), (B) Treatment efficiency of natural wood and UiO?66?NH2/wood membrane filter at different flow rates(the initial concentration of Hg2+ aqueous solution: 0.01 mg/L) and comparison of the residual Hg2+ concentration in the filtered water with the national maximum allowable concentration and different treatment rates; (C), (D) treatment efficiency of natural wood and UiO?66?NH2/wood membrane filter for different concentrations of Hg2+ aqueous solution(flow rate of Hg2+ aqueous solution: 110 L?m-2?h-1) and comparison of the residual Hg2+ concentration in the filtered water with the national maximum allowable concentration and different treatment rates.
| 1 | United Nations World Water Assessment Programme, Unesco, 2003 |
| 2 | Grimm J., Bessarabov D., Sanderson R., Desalination, 1998, 115(3), 285—294 |
| 3 | Blowes D. W., Ptacek C. J., Jambor J. L., Environ. Sci. Technol., 1997, 31(12), 3348—3357 |
| 4 | Da̧browski A., Hubicki Z., Podkościelny P., Robens E., Chemosphere, 2004, 56(2), 91—106 |
| 5 | Thong Z. W., Han G., Cui Y., Gao J., Chung T. S., Chan S. Y., Wei S., Environ. Sci. Technol., 2014, 48(23), 13880—13887 |
| 6 | Dong Z. H., Wang D., Liu, X., Pei X. F., Chen, L. W., Jin J., Mater. Chem. A, 2014, 2(16), 5034—5040 |
| 7 | Jiang G. Q., Bai J. F., Xing H., Li Y. Z., You X. Z., Cryst. Growth Des., 2006, 6(6), 1264—1266 |
| 8 | Horcajada P., Chalati T., Serre C., Gillet B., Sebrie C., Baati T., Eubank J. F., Heurtaux D., Clayette P., Kreuz C., Chang J. S., Wang Y. K. H., Marsaud V, Bories P. N., Cynober L., Gil S., Férey G., Couvreur P., Gref R., Nat. Mater., 2009, 9(2), 172—178 |
| 9 | Liu X., Demir N. K., Wu Z., Li K., J. Am. Chem. Soc., 2015, 137(22), 6999—7002 |
| 10 | Denny M. S., Cohen S. M., Angew. Chem. Int. Ed., 2015, 54(31), 9029—9032 |
| 11 | Lin K. Y. A., Liu Y. T., Chen S. Y., J. Colloid Interf. Sci., 2016, 461(1), 79—87 |
| 12 | Chen Q., He Q., Lv M., Xu Y., Yang H., Liu X., Wei F., Appl. Surf. Sci., 2015, 327(2), 77—85 |
| 13 | McManamon C., Burke A. M., Holmes J. D., Morris M. A., J. Colloid. Interface Sci., 2012, 369(1), 330—337 |
| 14 | Peterson G. W., DeCoste J. B., Fatollahi⁃Fard F., Britt D. K., Ind. Eng. Chem. Res., 2014, 53(2), 701—707 |
| 15 | Farha O. K., Eryazici I., Jeong N. C., Hauser B. G., Wilmer C. E., Sarjeant A. A., Snurr R. Q., Nguyen S. T., Yazaydin A. O., Hupp J. T., J. Am. Chem. Soc., 2012, 134(36), 15016—15021 |
| 16 | Bu X. H., Zuo J. L., Sci. China: Chem., 2016, 59(8), 927—928 |
| 17 | Alaerts L., Maes M., Giebeler L., Jacobs P. A., Martens J. A., Denayer J. F. M., Kirschhock C. E. A., De Vos D. E., J. Am. Chem. Soc., 2008, 130(43), 14170—14178 |
| 18 | Cai G., Ding M., Wu Q., Jiang H., Natl. Sci. Rev., 2020, 7(1), 37—45 |
| 19 | Lu G., Hupp J. T., J. Am. Chem. Soc., 2010, 132(23), 7832—7833 |
| 20 | Mondloch J. E., Katz M. J., Isley III W. C., Ghosh P., Liao P., Bury W., Wagner G. W., Hall M. G., DeCoste J. B., Peterson G. W., Snurr R. Q., Cramer C. J., Hupp J. T., Farha O. K., Nat. Mater., 2015, 14(5), 512—514 |
| 21 | Horcajada P., Serre C., Maurin G., Ramsahye N. A., Balas F., Vallet⁃Regi M., Sebban M., Taulelle F., Ferey G., J. Am. Chem. Soc., 2008, 130(21), 6774—6780 |
| 22 | Saleem H., Rafique U., Davies R. P., Micropor. Mesopor. Mater., 2016, 221(2), 238—244 |
| 23 | Yu M. H., Space B., Franz D., Zhou W., He C., Li L., Krishna R., Chang Z., Li W., Hu T. L., J. Am. Chem. Soc., 2019, 141(44), 17703—17712 |
| 24 | Shi Y. X., Zhang W. H., Abrahams B. F., Braunstein P., Lang J. P., Angew. Chem. Int. Ed., 2019, 131(28), 9553—9558 |
| 25 | Yee K. K., Reimer N., Liu J., Cheng S.Y.,Yiu S. M., Weber J., Stock N., Xu Z. T., J. Am. Chem. Soc., 2013, 135(21), 7795—7798 |
| 26 | Huang L. J., He M., Chen B. B., Hu B., Chemosphere, 2018, 199, 435—444 |
| 27 | Oveisi M., Asli M. A., Mahmoodi N. M., J. Hazard. Mater., 2018, 347, 123—140 |
| 28 | Chen F. J., Gong A. S., Zhu M., Chen G., Lacey S. D., Jiang F., Li Y., Wang Y., Dai J., Yao Y., Song J., Liu B., Fu K., Das S., Hu L., ACS Nano., 2017, 11(4), 4275—4282 |
| 29 | Dong X. Y., Zhuo X., Wei J., Zhang G., Li Y. F., ACS Appl. Mater. Interfaces, 2017, 9(1), 9070—9078 |
| 30 | Luu C. L., Nguyen T. T. V., Nguyen T., Hoang T. C., Adv. Nat. Sci: Nanosci. Nanotechnol., 2015, 6(2), 025004—025009 |
| 31 | Wang P., Sun D., Deng M. X., Zhang S. N., Bi Q.Y., Zhao W., Huang F. Q., Environ. Sci.: Nano, 2020, 7(4), 1266—1274 |
| 32 | Zhou H., Zhu H. X., Xue F., He H., Wang S. F., Chem. Engin. J., 2020, 385(4), 123879—123889 |
| 33 | Liu Y. L., Xu J., Cao Z., Fu R.Q., Zhou C. C., Wang Z. N., Xu X. H., J. Colloid Interface Sci., 2020, 559(2), 215—225 |
| 34 | Liu L. P., Zhang J. Y., Fang H. B., Chen L. P., Su C. Y., Chem. Asian J., 2016, 11(16i), 2278—2283 |
| 35 | Yang Z., Liu H. W., Li J., Yang K., Zhang Z. Z., Chen F. J., Wang B. D., ACS Appl. Mater. Interfaces, 2020, 12(13), 15002—15011 |
| 36 | Deng S., Zhang G. S., Wang X., Zheng T., Wang P., Chem. Eng. J., 2015, 276(9), 349—357 |
| [1] | CHANG Shuqing, XIN Xu, HUANG Yaqi, ZHANG Xincong, FU Yanghe, ZHU Weidong, ZHANG Fumin, LI Xiaona. Pyroelectrically-induced Catalytic Performance of Zr-based MOF Under Cold-hot Alternation [J]. Chem. J. Chinese Universities, 2021, 42(8): 2558. |
| [2] | LIU Yigang,ZHAO Peng,HAN Yugui,SONG Xin,HAN Zhipeng,XIE Liangbo,LI Zhuang,JIA Xiaoqing,LI Yi. W Element Doped CeO2 as Heterogeneous Electro-Fenton Catalyst for Efficient Treatment of Oily Wastewater † [J]. Chem. J. Chinese Universities, 2020, 41(3): 498. |
| [3] | DING Yao-Ying, WANG Cheng-Zhi, WEN Xian-Fang, ZHANG Xin-Peng, YE Lin, ZHANG Ai-Ying, FENG Zeng-Guo. Preparation of Amidoxime Modified Polyacrylonitrile Nanofibers and Its Application in Metal-ion Containing Waste Water Treatment [J]. Chem. J. Chinese Universities, 2013, 34(7): 1758. |
| [4] | ZHANG Xue-Na, ZHONG Xin-Wen, LIU Ming-Yu, LI Chen, LIN Hai-Bo. Effect of Ion Exchange Membrane on Pentachlorophenol Degradation by Biofilm-electrode Method [J]. Chem. J. Chinese Universities, 2013, 34(12): 2841. |
| [5] | SUN Hong-Liang*, ZHU Li-Zhong, ZHU Jian-Xi. Correlation of Structure and Sorption Behavior of Modified Bentonite [J]. Chem. J. Chinese Universities, 2011, 32(8): 1825. |
| [6] | YANG Shao-Xia, FENG Yu-Jie, WAN Jia-Feng, CAI Wei-Min ZHU Wan-Peng, JIANG Zhan-Peng . Structure and Activity of RuO2/γ-Al2O3 Catalyst Doped with CeO2 in Wet Air Oxidation Degradation of Phenol [J]. Chem. J. Chinese Universities, 2005, 26(5): 897. |
| [7] | QIN Ying, YUE Ying-Hong, GAO Zi . Chemical Liquid Deposition and Hydrophobicity of Zeolites [J]. Chem. J. Chinese Universities, 1998, 19(9): 1479. |
| [8] | Huang Zi-qiang, Chen Li-gen, Yang Yan, Zhu Chuan-zheng . The Effect of Magnetic Field on the Coagulation of Ni-Contained Wastewater [J]. Chem. J. Chinese Universities, 1991, 12(10): 1405. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||