

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (4): 685.doi: 10.7503/cjcu20180741
• Analytical Chemistry • Previous Articles Next Articles
WU Xuemei1, WEI Jie1, XU Hongping2, ZUO Lingxia2, SHAN Xiaoyan2, YAO Huiqin1,2,*( ), ZHENG Zhixiang3
), ZHENG Zhixiang3
Received:2018-11-01
Online:2019-04-03
Published:2019-01-09
Contact:
YAO Huiqin
E-mail:huiqin_yao@163.com
Supported by:TrendMD:
WU Xuemei,WEI Jie,XU Hongping,ZUO Lingxia,SHAN Xiaoyan,YAO Huiqin,ZHENG Zhixiang. Switchable Electrochemical Behavior of Matrine and Sophoridine at Multi-stimuli Responsive Hydrogel Film Electrode and Construction of Logic Gate†[J]. Chem. J. Chinese Universities, 2019, 40(4): 685.
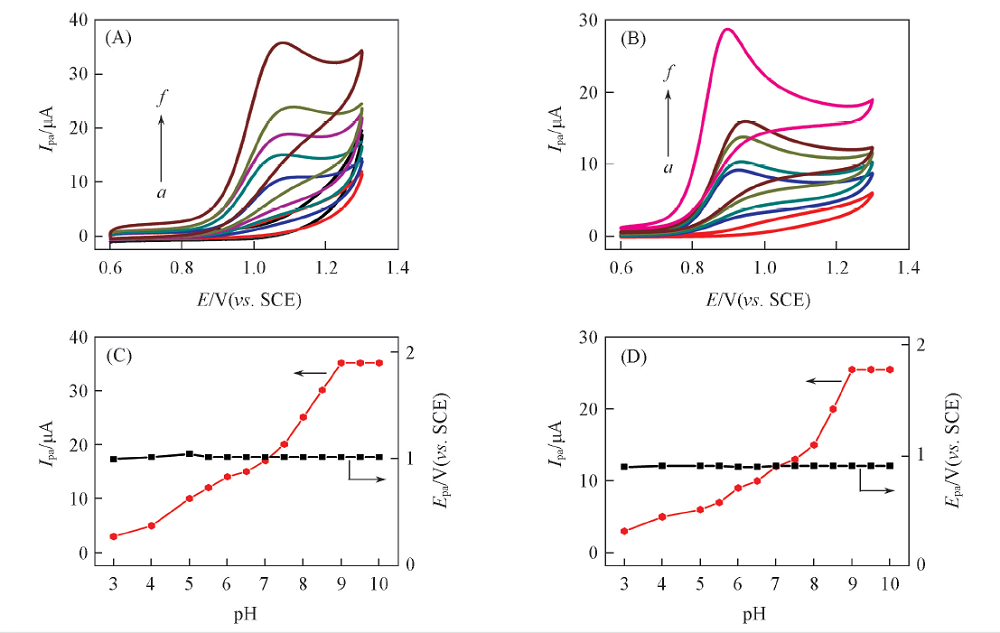
Fig.1 CVs of 0.01 mol/L MT(A) and SR(B) at different pH values and effects of pH value on the Ipa and oxidation peak potential(Epa) of MT(C) and SR(D)pH: a. 4.0; b. 5.0; c. 6.0; d. 7.0; e. 8.0; f. 9.0.
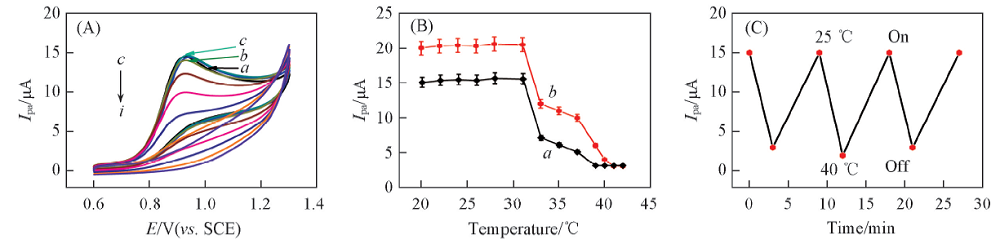
Fig.3 CVs curves of 0.01 mol/L SR at 0.05 V/s in pH=9.0 NaCl solution without Na2SO4 for PNIPAM films at different temperatures(A), influence of solution temperature on Ipa of SR(a) and MT(b) in pH=9.0 NaCl solution(B) and variation of Ipa of 0.01 mol/L SR at 0.05 V/s in pH=9.0 NaCl solution with immersion time at temperature switched between 25 ℃ and 40 ℃ for the same PNIPAM films(C)(A) Temperature/℃: a. 25; b. 27; c. 29; d. 31; e. 33; f. 35; g. 37; h. 39; i. 40.

Fig.4 SEM top-view images of PNIPAM hydrogel films on GC surface after treated with pH=9.0 NaCl solutions(A) At 25 ℃; (B) at 40 ℃; (C) containing 0.45 mol/L Na2SO4 at 25 ℃; (D) containing 20%(volume fraction) methanol at 25 ℃.
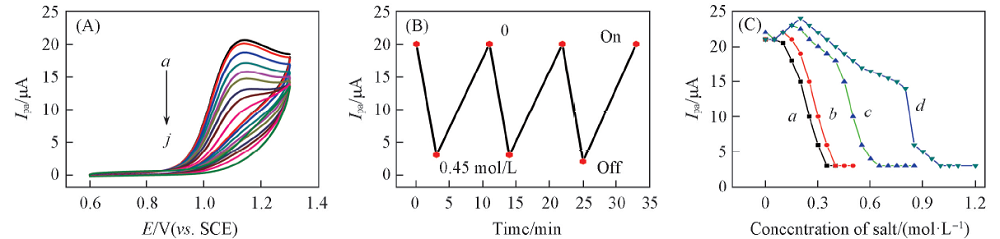
Fig.5 CVs curves of 0.01 mol/L MT at 0.05 V/s in pH=9.0 NaCl solutions containing 0(a), 0.05(b), 0.10(c), 0.15(d), 0.20(e), 0.25(f), 0.30(g), 0.35(h), 0.40(i) and 0.45 mol/L(j) Na2SO4 for PNIPAM films at 25 ℃(A), variation of Ipa of 0.01 mol/L MT with immersion time containing 0 and 0.45 mol/L Na2SO4(B) and dependence of Ipa of 0.01 mol/L MT at 0.05 V/s in pH=9.0 NaCl solutions at 25 ℃ on Na2SO4(a), MgSO4(b), NaCl(c) and NaNO3(d) for PNIPAM films(C)
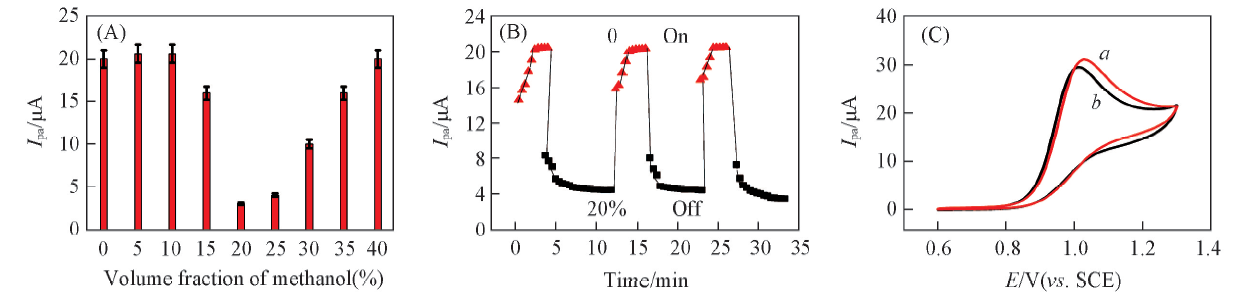
Fig.6 Influence of methanol volume fraction(0—40%) in water/methanol mixture solvent on Ipa of MT in pH=9.0 solutions for PNIPAM films at 25 ℃(A), variation of Ipa with immersion time in solutions containing 0() and 20%() methanol(B) and CVs of 0.01 mol/L MT containing 0(a) and 20%(b) methanol at bare GC electrodes(scan rate: 0.05 V/s)(C)
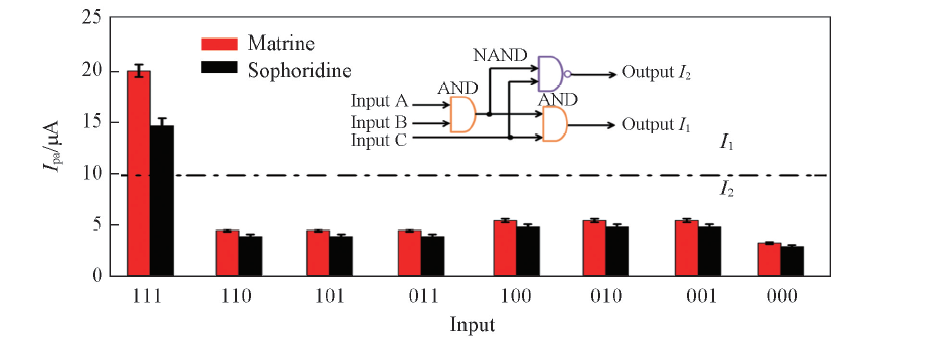
Fig.7 Ipa(CVs) obtained after application of eight combinations of the three input signalsThe dashed lines show the threshold values separating output signals. Inset: logic circuit for the 3-input/2-output logic gate system(input A=temperature, input B=Na2SO4, input C=methanol).
| Input A(Temperature) | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 0 |
|---|---|---|---|---|---|---|---|---|
| Input B(S | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 |
| Input C(Methanol) | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 |
| Output(Ipa) | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
Table 1 Truth values of 3-input/2-output logic gate system
| Input A(Temperature) | 1 | 1 | 1 | 0 | 1 | 0 | 0 | 0 |
|---|---|---|---|---|---|---|---|---|
| Input B(S | 1 | 1 | 0 | 1 | 0 | 1 | 0 | 0 |
| Input C(Methanol) | 1 | 0 | 1 | 1 | 0 | 0 | 1 | 0 |
| Output(Ipa) | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| [1] | Katz E., Poghossian A., Schöning M. J., Anal. Bioanal.Chem.,2017, 409(1), 81—94 |
| [2] | Erbas-Cakmak S., Kolemen S., Sedgwick A. C., Gunnlaugsson T., James T. D., Yoon J., Akkaya E. U., Chem. Soc.Rev.,2018, 47(7), 2228—2248 |
| [3] | Fu T., Lyu Y. F., Liu H., Peng R. Z., Zhang X. B., Ye M., Tan W. H., Trends. Biochem.Sci.,2018, 43(7), 547—560 |
| [4] | Wang J., Katz E., Anal. Bioanal.Chem.,2010, 398(4), 1591—1603 |
| [5] | Willner I., Shlyahovsky B., Zayats M., Bilha W.,Chem. Soc. Rev., 2008, (37), 1153—1165 |
| [6] | Miyamoto T., Razavi S., Derose R., Inoue T.,ACS Synth.Biol.,2013, 2(2), 72—82 |
| [7] | Bakshi S., Zavalov O., Halámek J., Privman V., Katz E., J. Phys. Chem.B,2013, 117(34), 9857—9865 |
| [8] | Zavalov O., Bocharova V., Privman V., Katz E., J. Phys. Chem.B,2012, 116(32), 9683—9689 |
| [9] | Halámek J., Bocharova V., Arugula M. A., Strack G., Privman V., Katz E., J. Phys. Chem.B,2011, 115(32), 9838—9845 |
| [10] | Zhou J., Arugula M. A., Halámek J., Pita M., Katz E., J. Phys. Chem.B,2009, 113(49), 16065—16070 |
| [11] | Li F. Y., Shi M., Huang C. H., Jin L. P., J. Mater.Chem.,2005, 15(29), 3015—3020 |
| [12] | Meng T. T., Xue L. X., Wang H., Haga M., J. Mater. Chem.C,2017, 5(13), 3390—3396 |
| [13] | Chu L. Y., Yamaguchi T., Nakao S., Advanced Materials,2002, 14(5), 386—389 |
| [14] | Laocharoensuk R., Bulbarello A., Hocevar S. B., Mannino S., Ogorevc B., Wang J., J. Am. Chem.Soc.,2007, 129(25), 7774—7775 |
| [15] | Wang J., Musameh M., Laocharoensuk R., Gonzalez-Garcia O., Oni J., Gervasio D., Electrochem.Commun.,2006, 8(7), 1106—1110 |
| [16] | Urban M.W., Stimuli-Responsive Polymeric Films and Coatings, American Chemical Society, Washington DC, 2005 |
| [17] | Liang Y., Song S. L., Yao H. Q., Hu N. F., Electrochim.Acta,2011, 56(14), 5166—5173 |
| [18] | Song S. L., Hu N. F., J. Phys. Chem.B,2010, 114(35), 11689—11695 |
| [19] | Liang J. Y., Yu X., Yang T. G., Li M. L., Shen L., Jin Y., Liu H. Y., Phys. Chem. Chem. Phys.,2017, 19(33), 22472—22481 |
| [20] | Lounasvuori M. M., Rosillo-Lopez M., Salzmann C. G., Caruana D. J., Holt K. B., Faraday Discuss.,2014, 172, 293—310 |
| [21] | Zhang L., Gao X., Yang L. F., Yu P., Mao L. Q., ACS Appl. Mater.Interfaces,2013, 5(16), 8120—8124 |
| [22] | Estefanía N. B., Agustín M. C. B., Agustín C. G., Fernández-Abedul. M. T., Biosens.Bioelectron.,2017, 91, 824—832 |
| [23] | Liu Z., He D., Zhang X., Li Y., Zhu C., Dong L., Zhang X., Xing Y., Wang C., Qiao H., Chen L., Brain Res.Bull.,2012, 88(4), 379—384 |
| [24] | Panayiotou M., Freitag R., Polymer,2005, 46(18), 6777—6785 |
| [25] | Ding X. L., Fries D., Jun B., Polymer,2006, 47(13), 4718—4725 |
| [26] | Zhang X. Z., Zhuo R. X., Chem. J. Chinese Universities,2000, 21(8), 1309—1311 |
| (张先正, 卓仁禧. 高等学校化学学报, 2000, 21(8), 1309—1311) | |
| [27] | Katsumoto Y., Tanaka T., Ozaki Y., J. Phys. Chem.B,2005, 109(44), 20690—20696 |
| [28] | Tanaka F., Koga T., Kaneda I., Winnik F. M., J. Phys. Condens.Matter.,2011, 23(28), 284105—284112 |
| [29] | Sakota K., Tabata D., Sekiya H., J. Phys. Chem.B,2015, 119(32), 10334—10340 |
| [30] | Meid J., Friedrich T., Tieke B., Lindner P., Richtering W., Phys. Chem. Chem.Phys.,2011, 13(8), 3039—3047 |
| [31] | Zhang Y. J., Furyk S., Bergbreiter D. E., Cremer P. S., J. Am. Chem.Soc.,2005, 127(41), 14505—14510 |
| [32] | Zhang Y. J., Furyk S., Sagle L. B., Cho Y., Bergbreiter D. E., Cremer P. S., J. Phys. Chem.C,2007, 111(25), 8916—8924 |
| [33] | Sakota K., Tabata D., Sekiya H., J. Phys. Chem.B,2015, 119(32), 10334—10340 |
| [34] | Zhang Y., Cremer P., Curr. Opin. Chem.Biol., 2006, 10(6), 658—663 |
| [35] | Liu G. M., Zhang G. Z., Langmuir,2005, 21(5), 2086—2090 |
| [36] | Mukherji D., Wagner M., Watson M. D., Winzen S., Oliveira T. E., Marques C. M., Kremer K., Soft Matter,2016, 12(38), 7983—7995 |
| [37] | Debashish M., Kremer K., Macromolecules,2013, 46(22), 9158—9163 |
| [1] | WU Yushuai, SHANG Yingxu, JIANG Qiao, DING Baoquan. Research Progress of Controllable Self-assembled DNA Origami Structure as Drug Carrier [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220179. |
| [2] | HE Zhechao, XIA Kun, WANG Jing, ZHOU Dan, LU Xinhuan, XIA Qinghua. Controllable Synthesis of SAPO-5 Molecular Sieves and Exploration of the Crystallization Process † [J]. Chem. J. Chinese Universities, 2020, 41(6): 1224. |
| [3] | XIAO Zhourui,HUANG Donghua,BIAN Mengmeng,YUAN Yali,NIE Jinfang. Fluorescence Detection Based on DNA-templated Silver Nanoclusters and the Construction of Multi-level Logic Gate † [J]. Chem. J. Chinese Universities, 2020, 41(1): 102. |
| [4] | LI Dan, WU Zhonghan, ZHOU Dan, JIANG Ding, LU Xinhuan, XIA Qinghua. Controllable Synthesis and Catalytic Applications of Hydrophobic Hybrid Zeolites† [J]. Chem. J. Chinese Universities, 2019, 40(7): 1359. |
| [5] | LEI Xuanxuan, TANG Shaokun, LI Lin, SUN Liwei. Controllable Synthesis of γ-Al2O3 Hexagonal Nanoplates with Ionic Liquids as the Template† [J]. Chem. J. Chinese Universities, 2018, 39(11): 2372. |
| [6] |
WANG Bin, WANG Xiaohong, LIU Zongrui, DUAN Limei, XU Ling, BAI Suozhu.
Preparation and Electroswitchable Luminescence Properties of Nanocomposite Films of Polyoxometalates and Ru(phen)3C |
| [7] | GUO Xiao, LIN Shaoying, QIN Kai, ZHANG Yahong, TANG Yi. Controllable Detemplation of Nanozeolites and Research of Molecular Diffusing Limitation† [J]. Chem. J. Chinese Universities, 2015, 36(4): 713. |
| [8] | WANG Qingguo, LI Dan, LU Ning, TONG Dingyi, ZHUANG Hejing, LI Fuqiu, WANG Lan, DUAN Haifeng, LIN Yingjie. Anion Probe and Molecular Logic Gate Based on Tryptanthrin Derivative† [J]. Chem. J. Chinese Universities, 2014, 35(10): 2114. |
| [9] | LIU Ming-Yang*, ZHAO Jing-Hong, ZOU Ming-Qiang, LIU Da. Controllable Fabrication of SnO2 Nanomaterials with Three Kinds of Morphologies and Their Application as Cataluminescence Sensor [J]. Chem. J. Chinese Universities, 2011, 32(5): 1112. |
| [10] | HE Li-Qin, GU Hong-Xia, YIN Deng-Ke, ZHANG Yi-Hua*, WANG Xiao-Shan . Synthesis and Anti-cancer Activity of Nitric Oxide Donor-based Matrine Derivatives [J]. Chem. J. Chinese Universities, 2010, 31(8): 1541. |
| [11] | QIN Sheng, CHEN Liu-Sheng, SHI Yi, JIN Xi-Gao . Controllable Assembly of Colloid Particles on the Surface of Polyelectrolyte Multilayer Films [J]. Chem. J. Chinese Universities, 2005, 26(8): 1548. |
| [12] | LIU Xin-Cai, ZHOU Hong-Wei, LI Zhi-Cheng, CHEN Chun-Hai, WEI Zhan-Hai, ZHANG Wan-Jin . Dynamic Mechanical Properties of Controllable Cross-linking Poly(ether ether ketone)s [J]. Chem. J. Chinese Universities, 2005, 26(3): 580. |
| [13] | LIU Xin-Cai, ZHOU Hong-Wei, CHEN Chun-Hai, LI Zhi-Cheng, CAO Hui, YANG Xiao-Qing, BEN Teng, ZHANG Wan-Jin . Dynamic Rheological Behavior for Controllable Cross-linking Poly(ether ether ketone)s [J]. Chem. J. Chinese Universities, 2003, 24(11): 2116. |
| [14] | LIU Xin-Cai, CHEN Chun-Hai, CAO Hui, BEN Teng, GAO Zi-Hong, ZHAO Xiao-Gang, YU You-Hai, REN Dian-Fu, LI Zhi-Cheng, WU Zhong-Wen, ZHANG Wan-Jin. Synthesis and Thermal Properties of Controllable Cross-linking Poly(ether ether ketone)s [J]. Chem. J. Chinese Universities, 2002, 23(9): 1817. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||