

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (12): 2475.doi: 10.7503/cjcu20150444
• Physical Chemistry • Previous Articles Next Articles
MO Wenlong, MA Fengyun*( ), LIU Yue’e, LIU Jingmei, ZHONG Mei,
), LIU Yue’e, LIU Jingmei, ZHONG Mei,
Received:2015-06-05
Online:2015-12-10
Published:2015-11-17
Contact:
MA Fengyun
E-mail:ma_fy@126.com
Supported by:CLC Number:
TrendMD:
MO Wenlong, MA Fengyun, LIU Yue’e, LIU Jingmei, ZHONG Mei, . Preparation of Mesoporous Al2O3 with High Specific Surface Area by Evaporation-induced Self-assembly Stategy and Its Application as Ni-Al2O3 Catalysts for CO2-CH4 Reforming†[J]. Chem. J. Chinese Universities, 2015, 36(12): 2475.
| Catalyst | Elemental content(molar fraction, %) | ||
|---|---|---|---|
| Ni | Al | O | |
| PAC0 | 4.13 | 37.73 | 58.14 |
| PAC0.01 | 4.10 | 37.75 | 58.15 |
| PAC0.02 | 4.08 | 37.76 | 58.16 |
| PAC0.03 | 4.07 | 37.74 | 58.19 |
| PAC0.05 | 4.09 | 37.75 | 58.16 |
Table 1 ICP-AES data of the catalysts
| Catalyst | Elemental content(molar fraction, %) | ||
|---|---|---|---|
| Ni | Al | O | |
| PAC0 | 4.13 | 37.73 | 58.14 |
| PAC0.01 | 4.10 | 37.75 | 58.15 |
| PAC0.02 | 4.08 | 37.76 | 58.16 |
| PAC0.03 | 4.07 | 37.74 | 58.19 |
| PAC0.05 | 4.09 | 37.75 | 58.16 |
| Support | SBET/(m2·g-1) | VBJH/(cm3·g-1) | DBJH/nm | Catalyst | SBET/(m2·g-1) | VBJH/(cm3·g-1) | DBJH/nm |
|---|---|---|---|---|---|---|---|
| PA0 | 197.25 | 0.41 | 3.71 | PAC0 | 190.83 | 0.45 | 2.40 |
| PA0.01 | 265.43 | 0.41 | 3.40 | PAC0.01 | 222.21 | 0.43 | 3.12 |
| PA0.02 | 320.12 | 0.39 | 3.32 | PAC0.02 | 280.15 | 0.46 | 3.24 |
| PA0.03 | 281.45 | 0.40 | 4.61 | PAC0.03 | 250.26 | 0.51 | 3.52 |
| PA0.05 | 254.26 | 0.39 | 5.30 | PAC0.05 | 210.44 | 0.50 | 3.87 |
Table 2 Structural parameters of the supports and catalysts
| Support | SBET/(m2·g-1) | VBJH/(cm3·g-1) | DBJH/nm | Catalyst | SBET/(m2·g-1) | VBJH/(cm3·g-1) | DBJH/nm |
|---|---|---|---|---|---|---|---|
| PA0 | 197.25 | 0.41 | 3.71 | PAC0 | 190.83 | 0.45 | 2.40 |
| PA0.01 | 265.43 | 0.41 | 3.40 | PAC0.01 | 222.21 | 0.43 | 3.12 |
| PA0.02 | 320.12 | 0.39 | 3.32 | PAC0.02 | 280.15 | 0.46 | 3.24 |
| PA0.03 | 281.45 | 0.40 | 4.61 | PAC0.03 | 250.26 | 0.51 | 3.52 |
| PA0.05 | 254.26 | 0.39 | 5.30 | PAC0.05 | 210.44 | 0.50 | 3.87 |
| Catalyst | α-NiO(%) | β-NiO(%) |
|---|---|---|
| PAC0 | 66 | 34 |
| PAC0.01 | 45 | 55 |
| PAC0.02 | 18 | 82 |
| PAC0.03 | 35 | 65 |
| PAC0.05 | 61 | 39 |
Table 3 Reduction peak area proportions of different NiO species
| Catalyst | α-NiO(%) | β-NiO(%) |
|---|---|---|
| PAC0 | 66 | 34 |
| PAC0.01 | 45 | 55 |
| PAC0.02 | 18 | 82 |
| PAC0.03 | 35 | 65 |
| PAC0.05 | 61 | 39 |
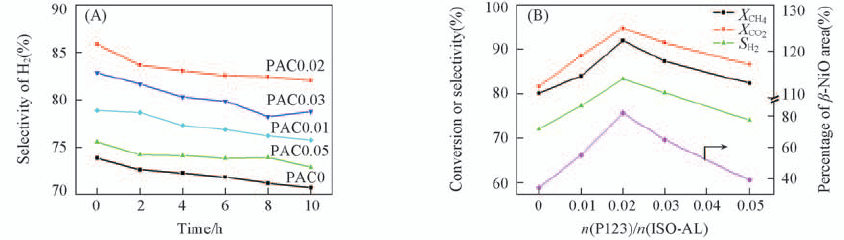
Fig.12 Selectivity of H2(A) and the change of CH4 and CO2 conversions and β-NiO percentage with the increasing of PA/ISO-AL(B) T=800 ℃, P=1.01325×105 Pa, GHSV=18000 h-1, V(CH4)/V(CO2)=1.
| Catalyst | Conversion(%) | Deactivation rate/(point·h-1) | |||||
|---|---|---|---|---|---|---|---|
| 0 h | 10 h | 120 h | 154 h | ||||
| PAC0.02 | 95.02 | 91.89 | 80.02 | 50.44 | 0.313(Ⅰ) | 0.108*(Ⅱ) | 0.870(Ⅲ) |
Table 4 Conversion of CH4 and corresponding deactivation rate
| Catalyst | Conversion(%) | Deactivation rate/(point·h-1) | |||||
|---|---|---|---|---|---|---|---|
| 0 h | 10 h | 120 h | 154 h | ||||
| PAC0.02 | 95.02 | 91.89 | 80.02 | 50.44 | 0.313(Ⅰ) | 0.108*(Ⅱ) | 0.870(Ⅲ) |
| Catalyst | w(%) | Reaction time/h | Carbon deposition rate/(mg·h-1· |
|---|---|---|---|
| PAC0 | 15.85 | 60 | 0.002642 |
| PAC0.02 | 22.10 | 154 | 0.001435 |
Table 5 Carbon deposition rate of PAC0 and PAC0.02
| Catalyst | w(%) | Reaction time/h | Carbon deposition rate/(mg·h-1· |
|---|---|---|---|
| PAC0 | 15.85 | 60 | 0.002642 |
| PAC0.02 | 22.10 | 154 | 0.001435 |
| Catalyst | Ni size*/nm | Icreasing rate(%) | |
|---|---|---|---|
| Before reaction | After reaction | ||
| PAC0 | 7.56 | 13.53 | 78.97 |
| PAC0.02 | 5.72 | 8.04 | 40.56 |
Table 6 Ni size before and after endurance test
| Catalyst | Ni size*/nm | Icreasing rate(%) | |
|---|---|---|---|
| Before reaction | After reaction | ||
| PAC0 | 7.56 | 13.53 | 78.97 |
| PAC0.02 | 5.72 | 8.04 | 40.56 |
| [1] | Chen X., Wang Y., Jiang L., Chem. J. Chinese Universities,2013, 34(2), 249—268 |
| (陈曦, 王耀, 江雷. 高等学校化学学报,2013, 34(2), 249—268) | |
| [2] | Liu Q., Ren J., Qin Z. F., Miao M. Q., Li Z., Chem. J. Chinese Universities,2013, 34(9), 2171—2177 |
| (刘泉, 任军, 秦志峰, 苗茂谦, 李忠. 高等学校化学学报,2013, 34(9), 2171—2177) | |
| [3] | Li Z. X., Na W., Wang H., Gao W. G., Chem. J. Chinese Universities,2014, 35(12), 2616—2623. |
| 李志雄, 纳薇, 王华, 高文桂. 高等学校化学学报, 2014, 35(12), 2616—2623 | |
| [4] | Mo W. L., Ma F. Y., Liu H. X., Hao S. H., Chemical Industry and Engineering Progress,2014, S1, 187—193 |
| (莫文龙, 马凤云, 刘红霞, 郝世豪. 化工进展, 2014, S1, 187—193) | |
| [5] | Xu L. L., Zhao H. H., Song H. L., Chou L. J., Int. J. Hydrogen Energy,2012, 37, 7497—7511 |
| [6] | Seo H. O., Sim J. K., Kim K. D., Kim Y. D., Lim D. C., Kim S. H., Appl. Catal. A: Gen., 2013, 451, 43—49 |
| [7] | Kim D. H., Sim J. K., Lee J., Seo H. O., Jeong M. G., Kim Y. D., Kim S. H., Fuel, 2013, 112, 111—116 |
| [8] | Shi C. K., Zhang P., Appl. Catal. B: Environ., 2012, 115/116, 190—200 |
| [9] | Sang L. X., Sun B., Tan H. Y., Du C. X., Wu Y. T., Ma C. F., Int. J. Hydrogen Energy,2012, 37, 13037—13043 |
| [10] | Yu Z. H., Yan Z., Fan H., Li Z., Chem. J. Chinese Universities,2014, 35(10), 2227—2233 |
| (于智慧, 闫泽, 范辉, 李忠. 高等学校化学学报,2014, 35(10), 2227—2233) | |
| [11] | Molina R, Poncelet G, J. Catal., 1998, 173, 257—267 |
| [12] | Yang R. C., Li X. G., Wu J. S., Appl. Catal. A: Gen., 2009, 368, 105—112 |
| [13] | Yang Z., Xia Y., Mokaya R., Adv. Mater., 2004, 16, 727—732 |
| [14] | Xia K., Fergusona D., Djaoued Y., Appl. Catal. A: Gen., 2010, 387, 231—241 |
| [15] | Liu S. G., Li J. P., Zhao N., Wei W., Sun Y. H., Chinese J. Catal., 2007, 28(11), 1019—1023 |
| (刘永刚, 李军平, 赵宁, 魏伟, 孙予罕. 催化学报, 2007, 28(11), 1019—1023) | |
| [16] | Belin T., Epron F., Mater. Sci. Eng. B,2005, 119, 105—118 |
| [17] | Zhang S., Wang J., Liu H., Wang X., Catal. Commun., 2008, 9, 995—1000 |
| [18] | Zou X. J., Wang X. G., Li L., Int. J. Hydrogen Energy,2010, 35, 12191—12200 |
| [19] | Yang J., Wang X. G., Li L., Shen K., Lu X. G., Ding W. Z., Appl. Catal. B: Environ., 2010, 96, 232—237 |
| [20] | Zhang J., Xu H. Y., Jin X. L., Ge Q. J., Li W. Z., Appl. Catal. A: Gen., 2005, 290, 87—96 |
| [21] | Martha Barroso-Qu iroga M, Eduardo Cast ro-Luna A, Int. J. Hydrogen Energy, 2010, 35, 6052—6056 |
| [22] | Guo J., Lou H., Zheng X., Carbon, 2007, 45(6), 1314—1321 |
| [23] | Xu J. K., Zhou W., Wang J. H., Li Z. J., Ma J. X., Chinese J. Catal., 2009, 30(11), 1076—1084 |
| (徐军科, 周伟, 汪吉辉, 李兆静, 马建新. 催化学报, 2009, 30(11), 1076—1084) | |
| [24] | Frusteri F., Spadarol Arena F., Chuvilin A., Carbon, 2002, 40(7), 1063—1070 |
| [25] | Pereniguezez R., Gonzalez-delacruz V. M., Caballero A., Holgado J. P., Appl. Catal. B: Environ., 2012, 123, 324—332 |
| [1] | JI Keming, MENG Fanhui, GAO Yuan, LI Zhong. Effect of Fuel on Structure and Catalytic Performance for Slurry Methanation over Ni-Al2O3 Catalysts Prepared by Combustion Method† [J]. Chem. J. Chinese Universities, 2016, 37(1): 134. |
| [2] | LI Yu-Hui, FENG Li-Juan*, WANG Jing-Gang, ZHOU Xuan, CHENG Bin-Bin, WANG Xiao-Yan, LI Chun-Hu. Catalytic Oxidative Desulfurization of Model Oil by MoO3/Mesoporous Al2O3 [J]. Chem. J. Chinese Universities, 2011, 32(3): 778. |
| [3] | LI Zhi-Ping1, ZHAO Rui-Hong2, GUO Fen1*, CHEN Jian-Feng1, WANG Gang1. Preparation and Characterization of Ordered Mesoporous Alumina with High Specific Surface Area with F127 as Template [J]. Chem. J. Chinese Universities, 2008, 29(1): 13. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||