

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (8): 1553.doi: 10.7503/cjcu20150221
• Physical Chemistry • Previous Articles Next Articles
SHI Dingfu, TAN Chen, WANG Peng, LI Yongqi, CHEN Shengjie, LIU Ye*( )
)
Received:2015-03-23
Online:2015-08-10
Published:2015-06-30
Contact:
LIU Ye
E-mail:yliu@chem.ecnu.edu.cn
Supported by:CLC Number:
TrendMD:
SHI Dingfu, TAN Chen, WANG Peng, LI Yongqi, CHEN Shengjie, LIU Ye. Biphasic Hydroformylation of 1-Octene Catalyzed by Ionic Rh(Ⅲ)-complex in the Presence of Tertiary Amine Functionalized Ionic Liquid†[J]. Chem. J. Chinese Universities, 2015, 36(8): 1553.
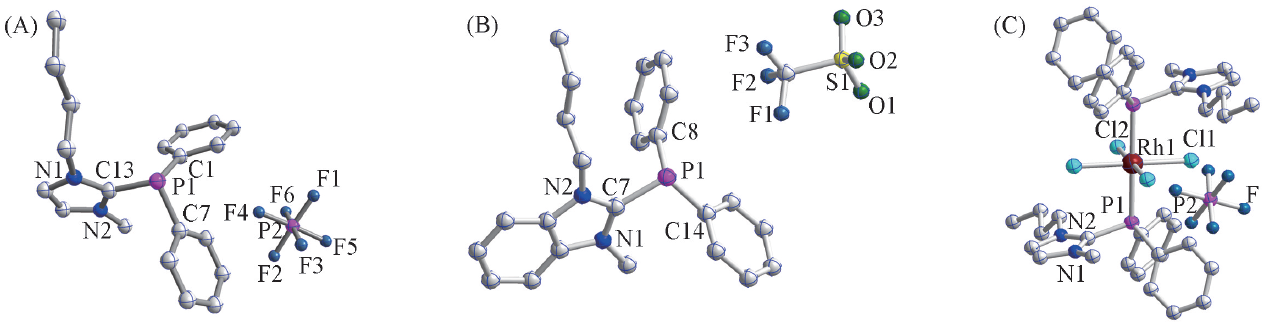
Fig.1 Single crystal structures of the phosphine-functionalized ionic liquids 1(A), 2(B) and the ionic Rh(Ⅲ)-complex 1a(C)Selected bond distance/nm: compound 1: P1—C13 0.1829(4), P1—C1 0.1820(4), P1—C7 0.1840(4); compound 2: P1—C7 0.1842(2), P1—C14 0.1820(3), P1—C8 0.1829(3); compound 1a: Rh—P 0.2378(15), 0.2378(15); Rh—Cl 0.2341(14), 0.2341(14), 0.2345(13), 0.2345(13).
| Empirical formula | C24H26N2P·CF3SO3 | Z | 2 |
|---|---|---|---|
| Formula weight | 522.5 | Dcalcd/(g·cm-3) | 1.388 |
| Crystal system | Triclinic | μ(Mo Kα)/mm-1 | 0.246 |
| Space group | P-1 | T/K | 296(2) |
| a/nm | 0.98502(9) | λ/nm | 0.071073 |
| b/nm | 1.13275(11) | Total reflections | 14651 |
| c/nm | 1.23674(11) | Unique reflections, Rint | 4381(0.0232) |
| α/(°) | 97.419(3) | R1 [I>2σ(I)] | 0.0515 |
| β/(°) | 107.282(3) | wR2(all data) | 0.1537 |
| γ/(°) | 103.712(3) | F(000) | 544 |
| V/nm3 | 1.2501(2) | Goodness-of-fit on F2 | 1.028 |
Table 1 Crystal data and structure refinement for the functionalized ionic liquid 2
| Empirical formula | C24H26N2P·CF3SO3 | Z | 2 |
|---|---|---|---|
| Formula weight | 522.5 | Dcalcd/(g·cm-3) | 1.388 |
| Crystal system | Triclinic | μ(Mo Kα)/mm-1 | 0.246 |
| Space group | P-1 | T/K | 296(2) |
| a/nm | 0.98502(9) | λ/nm | 0.071073 |
| b/nm | 1.13275(11) | Total reflections | 14651 |
| c/nm | 1.23674(11) | Unique reflections, Rint | 4381(0.0232) |
| α/(°) | 97.419(3) | R1 [I>2σ(I)] | 0.0515 |
| β/(°) | 107.282(3) | wR2(all data) | 0.1537 |
| γ/(°) | 103.712(3) | F(000) | 544 |
| V/nm3 | 1.2501(2) | Goodness-of-fit on F2 | 1.028 |
| Entry | Rh-complex | Additive | Conv.b(%) | Sel.b(%) | L/Bc | TO | |
|---|---|---|---|---|---|---|---|
| Ald(n+iso) | iso-Octenes | ||||||
| 1 | 1a | 47 | 77 | 23 | 2.1 | 360 | |
| 2 | 1a | Et3N | 93 | 88 | 12 | 2.6 | 820 |
| 3 | 1a | 1-Methylpiperidine | 96 | 88 | 12 | 2.8 | 845 |
| 4e | 1a | [PEmim]BF4 | 88 | 94 | 6 | 2.7 | 830 |
| 5 | 2a | 44 | 72 | 28 | 2.7 | 320 | |
| 6 | 2a | Et3N | 91 | 87 | 13 | 2.6 | 790 |
| 7 | 2a | 1-Methylpiperidine | 93 | 86 | 14 | 2.7 | 800 |
| 8e | 2a | [PEmim]BF4 | 93 | 93 | 7 | 2.5 | 865 |
Table 2 Homogeneous hydroformation of 1-octene catalyzed by the ionic Rh(Ⅲ)-complexes in the presence of auxiliary tertiary aminesa
| Entry | Rh-complex | Additive | Conv.b(%) | Sel.b(%) | L/Bc | TO | |
|---|---|---|---|---|---|---|---|
| Ald(n+iso) | iso-Octenes | ||||||
| 1 | 1a | 47 | 77 | 23 | 2.1 | 360 | |
| 2 | 1a | Et3N | 93 | 88 | 12 | 2.6 | 820 |
| 3 | 1a | 1-Methylpiperidine | 96 | 88 | 12 | 2.8 | 845 |
| 4e | 1a | [PEmim]BF4 | 88 | 94 | 6 | 2.7 | 830 |
| 5 | 2a | 44 | 72 | 28 | 2.7 | 320 | |
| 6 | 2a | Et3N | 91 | 87 | 13 | 2.6 | 790 |
| 7 | 2a | 1-Methylpiperidine | 93 | 86 | 14 | 2.7 | 800 |
| 8e | 2a | [PEmim]BF4 | 93 | 93 | 7 | 2.5 | 865 |
| Run | 1a-[PEmim]BF4 | 2a-[PEmim]BF4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Conv.b(%) | Sel.b(%) | L/Bc | TOFd/h-1 | Conv.b(%) | Sel.b(%) | L/Bc | TOFd/h-1 | ||||
| Nonanal | iso-Octenes | Nonanal | iso-Octenes | ||||||||
| 1 | 90 | 94 | 7 | 1.2 | 280 | 98 | 90 | 10 | 2.1 | 290 | |
| 2 | 91 | 93 | 7 | 2.4 | 280 | 83 | 66 | 34 | 1.6 | 180 | |
| 3 | 94 | 93 | 7 | 2.0 | 290 | 63 | 65 | 35 | 2.2 | 140 | |
| 4 | 89 | 86 | 14 | 1.3 | 255 | 58 | 46 | 54 | 1.6 | 90 | |
| 5 | 87 | 67 | 33 | 2.2 | 195 | 55 | 32 | 68 | 2.3 | 60 | |
Table 3 Biphasic hydroformylation of 1-octene catalyzed by compounds 1a and 2a in piperidyl-functionalized ionic liquid of [PEmim]BF4 respectivelya
| Run | 1a-[PEmim]BF4 | 2a-[PEmim]BF4 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Conv.b(%) | Sel.b(%) | L/Bc | TOFd/h-1 | Conv.b(%) | Sel.b(%) | L/Bc | TOFd/h-1 | ||||
| Nonanal | iso-Octenes | Nonanal | iso-Octenes | ||||||||
| 1 | 90 | 94 | 7 | 1.2 | 280 | 98 | 90 | 10 | 2.1 | 290 | |
| 2 | 91 | 93 | 7 | 2.4 | 280 | 83 | 66 | 34 | 1.6 | 180 | |
| 3 | 94 | 93 | 7 | 2.0 | 290 | 63 | 65 | 35 | 2.2 | 140 | |
| 4 | 89 | 86 | 14 | 1.3 | 255 | 58 | 46 | 54 | 1.6 | 90 | |
| 5 | 87 | 67 | 33 | 2.2 | 195 | 55 | 32 | 68 | 2.3 | 60 | |
| [1] | Eilbracht P., Bärfacker L., Buss C., Hollmann C., Kitsos-Rzychon B. E., Kranemann C. L., Rische T., Roggenbuck R., Schmidt A., Chem. Rev., 1999, 99(11), 3329—3365 |
| [2] | Roelen O., Production of Oxygenated Carbon Compounds, US 2327066, 1943-08-17 |
| [3] | Kuntz E. G., Chem. Tech., 1987, 17(9), 570—575 |
| [4] | Bartik T., Ding H., Bartik B., Hanson B. E., Bartik T., J. Mol. Catal. A: Chem., 1995, 98(3), 117—122 |
| [5] | Sandee A. J., van der Veen L. A., Reek J. N. H., Kamer P. C. J., Lutz M., Spek A. L., van Leeuwen P. W. N. M., Angew. Chem. Int. Ed., 1999, 38(21), 3231—3235 |
| [6] | Kohlpaintner C. W., Frohning C. D., Applied Homogeneous Catalysis with Organometallic Compounds, 1996, 1, 29—104 |
| [7] | Tolman C.A., Faller J. W., Homogeneous Catalysis with Metal Phosphine Complexes, Plenum Press, New York, 1983, 13—19 |
| [8] | Franke R., Selent D., Börner A., Chem. Rev., 2012, 112(11), 5675—5732 |
| [9] | Yu S. C., Chi Y. M., Guan Z. H., Zou Y. P., Li W., Zhang X. M., Org. Lett., 2009, 11(1), 241—244 |
| [10] | Ali B. E., Catal. Commun., 2003, 4(12), 621—626 |
| [11] | Ali B. E., Tijani J., FettouhiM., J. Mol. Catal. A: Chem., 2005, 230(1/2), 9—16 |
| [12] | Burgess K., van der Donk W. A., Westcott S. A., Marder T. B., Baker R. T., Calabrese J. C., J. Am. Chem. Soc., 1992, 114(24), 9350—9359 |
| [13] | Osborn J.A., Jardine F. H., Young J. F., Wilkinson G.,J. Chem. Soc. A, 1966, (12), 1711—1732 |
| [14] | Bennett M. J., Donaldson P. B., Inorg.Chem., 1977, 16(3), 655—660 |
| [15] | Brink A., Roodt A., Steyl G., Visser H. G., Dalton Trans., 2010, 39(23), 5572—5578 |
| [16] | Tang S., Baker G. A., Zhao H., Chem. Soc. Rev., 2012, 41(10), 4030—4066 |
| [17] | Liu Y., Wang S., Liu W., Wan Q., Wu H., Gao G., Curr. Org. Chem., 2009, 13(13), 1322—1346 |
| [18] | Luo S., Zhang L., Cheng L., Chem. Asian J., 2009, 4(8), 1184—1195 |
| [19] | Ohno H., Chem. Soc. Japan, 2006, 79(11), 1665—1680 |
| [20] | Lee S.,Chem. Commun., 2006, (10), 1049—1063 |
| [21] | Li X., Zhao D., Fei Z., Wang L., Sci. China Ser. B: Chem., 2006, 49(5), 385—401 |
| [22] | Kunene T. E., Webb P. B., Cole-Hamilton D. J., Green Chem., 2011, 13(6), 1476—1481 |
| [23] | Kolbeck C., Paape N., Cremer T., Schulz P. S., Maier F., Steinrueck H., Wasserscheid P., Chem.Eur.J., 2010, 16(40), 12083—12087 |
| [24] | Wasserscheid P., Waffenschmidt H., Machnitzki P., Kottsieper K.W., Stelzer O.,Chem. Commun., 2001, (5), 451—452 |
| [25] | Chen S., Li Y., Wang Y., Zhao X., Liu Y., J. Mol. Catal.A: Chem., 2015, 396, 68—76 |
| [26] | Wang X., Wang Y., Zhang J., Zhao X., Liu Y., J. Organomet. Chem., 2014, 762, 40—47 |
| [27] | Zhang J., Wang Y., Zhao X., Liu Y.,Eur. J. Inorg. Chem., 2014, (6), 975—985 |
| [28] | Chen S., Wang Y., Yao W., Zhao X., VO-Thanh G., Liu Y., J. Mol. Catal. A: Chem., 2013, 378, 293—298 |
| [29] | You H., Wang Y., Zhao X., Chen S., Liu Y., Organometallics, 2013, 32(9), 2698—2704 |
| [30] | You H. X., Wang Y. Y., Wang X. Z., Liu Y., Progress in Chemistry, 2013, 25(10), 1656—1666 |
| (尤洪星, 王永勇, 王雪珠, 刘晔. 化学进展, 2013, 25(10), 1656—1666) | |
| [31] | Azouri M., Andrieu J., Picquet M., Cattey H., Inorg. Chem., 2009, 48(3), 1236—1242 |
| [32] | Maaliki C., Lepetit C., Duhayon C., Canac Y., Chauvin R., Chem. Eur. J., 2012, 18(50), 16153—16160 |
| [33] | Canac Y., Debono N., Vendier L., Chauvin R., Inorg. Chem., 2009, 48(12), 5562—5568 |
| [34] | Abdellah I., Lepetit C., Canac Y., Chauvin R., Chem. Eur. J., 2010, 16(44), 13095—13108 |
| [1] | DONG Xu, FENG Boxu, CHEN Li, LI Xin. Asymmetric [3+2] Annulations of 1,4-Di-thiane-2,5-diol and N-Boc Aldimines Catalyzed by Tertiary-amine Thiourea† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1707. |
| [2] | MENG Jiafeng, NI Xufeng, ZHENG Hao, SHEN Zhiquan. Copolymerization of Norbornene and 1-Octene Catalyzed by Bis(phenoxy-imine) Titanium Complex† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1853. |
| [3] | ZHANG Cui,WANG Lijuan,LÜ Zhongwen,XU Sheng,MI Puke. Synthesis of Binuclear Complexes Containing Phenoxyimine Ligands and Application for Ethylene Polymerization and Copolymerization† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2811. |
| [4] | YANG Yongsheng, YANG Xin, TIAN Zhiyue, XUE Ying. Theoretical Studies on the Mechanisms of Transfer Hydroformylation Between Hydrocinnamaldehyde and Norbornadiene Catalyzed by Rh(Xantphos)L Complexes† [J]. Chem. J. Chinese Universities, 2016, 37(12): 2199. |
| [5] | LIU Jia, YAN Li, JIANG Miao, DING Yunjie. Effect of Metal Particle Size on the Performance of Tethered-phosphine Modified Rh/SiO2 in Hydroformylation† [J]. Chem. J. Chinese Universities, 2016, 37(1): 114. |
| [6] | ZHANG Ding-Lin, FU Hai-Yan, ZHAO Xian-Ying, ZHAO Hua-Wen, CHEN Hua, LIU Yi-Min, LI Xian-Jun. HRhCO(TPPTS)3/HAP Hydroformylate 1-Hexene High Selectivity and Special Isomerization Performance [J]. Chem. J. Chinese Universities, 2012, 33(08): 1835. |
| [7] | WANG Bin, LIU Chen-Jiang, WANG Ji-De, LEI Zhen-Kai, HU Dong-Lin. Synthesis of Functionalized Ionic Liquids Based on Benzimidazolium and Their Properties [J]. Chem. J. Chinese Universities, 2012, 33(01): 76. |
| [8] | LI Jin-Jin, MA Lan, HE De-Hua*, LI Guang-Xing*. Hydroformylation of 1-Octene over Amorphous Co-P-B Catalysts [J]. Chem. J. Chinese Universities, 2011, 32(12): 2844. |
| [9] | LIU Ye, LI Min, LU Yong*, WU Hai-Hong, GAO Guo-Hua. Palladium Complex Catalyzed Heck Coupling Reaction in Synergic Functionalized Ionic Liquid System [J]. Chem. J. Chinese Universities, 2007, 28(4): 723. |
| [10] |
ZHANG Sheng-Mao, LI Jian, WU Zhi-Shen, ZHANG Ping-Yu, ZHANG Zhi-Jun.
Preparation and Structural Characterization of Carboxyl-functional Ionic Liquidsurface-modified TiO2 Nanoparticles [J]. Chem. J. Chinese Universities, 2006, 27(9): 1615. |
| [11] | GONG Sheng-Min, MA Hong-Yang,WAN Xin-Hua,ZHAO Yong-Feng,HE Ji-Yu, ZHOU Qi-Feng. Synthesis and Characterization of New Cyno-group Functionalized Ionic Liquids and Their Rheological Properties [J]. Chem. J. Chinese Universities, 2006, 27(4): 761. |
| [12] | FAN Bao-Min1, XIE Jian-Hua2, ZHOU Zhang-Tao2, ZHANG Qi2, TU Yong-Qiang1, ZHOU Qi-Lin2. Application of Chiral Spiro Monophosphorus Ligands in Rhodium-catalyzed Asymmetric Hydroformylation [J]. Chem. J. Chinese Universities, 2006, 27(10): 1894. |
| [13] | ZHANG Qing-Shan, LIU Ai-Xia, GUO Bing-Nan, WU Feng . Synthesis of Ionic Liquids Based on the N-Methyl-N-allyl Morpholinium Cation [J]. Chem. J. Chinese Universities, 2005, 26(2): 340. |
| [14] | LI Min, XU Bin, CHEN Hua, HE Yu-E, HUANG Xue-Yuan, LI Li-Bo, LI Yao-Zhong, LI Xian-Jun . Hydroformylation of 1-Dodecene Catalyzed by Water Soluble Rhodium-Phos-phine Complexes in Micellar System with Cationic Gemini Surfactant [J]. Chem. J. Chinese Universities, 2004, 25(4): 717. |
| [15] | XU Bin, LI Min, YANG Min, ZHENG Hong-Jie, HE Yu-E, CHEN Hua, LI Xian-Jun . Promotion Effect of Cationic Gemini Surfactants on 1-Dodecene Hydroformylation in Biphasic Catalytic System [J]. Chem. J. Chinese Universities, 2004, 25(11): 2060. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||