

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (5): 1004.doi: 10.7503/cjcu20141102
• Polymer Chemistry • Previous Articles Next Articles
LI Xianhua1, ZHANG Leigang1, WANG Xuexue1, YU Qingbo2,*( )
)
Received:2014-12-16
Online:2015-05-10
Published:2015-03-31
Contact:
YU Qingbo
E-mail:yuqingbo007@163.com
Supported by:CLC Number:
TrendMD:
LI Xianhua, ZHANG Leigang, WANG Xuexue, YU Qingbo. Fabrication and Multi-antitumor Effect of Novel Dextran-hemin Crosslinked Micelles Triggered by Photo Conditions†[J]. Chem. J. Chinese Universities, 2015, 36(5): 1004.
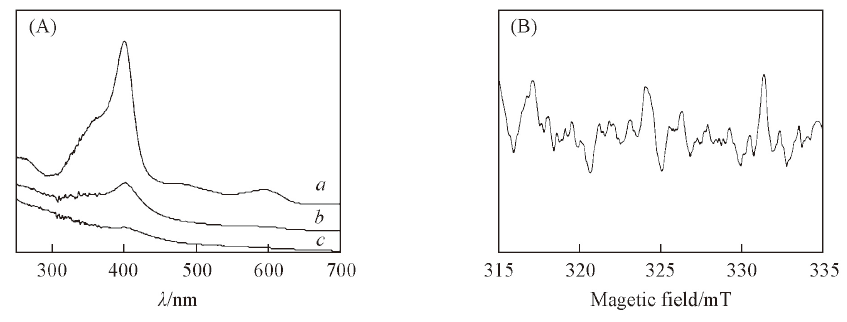
Fig.1 UV-Vis spectra of hemin measured at different irradiation time(A) and ESR spectrum of hemin after laser irradiation(B) Time/min: a. 0; b. 5; c. 10.
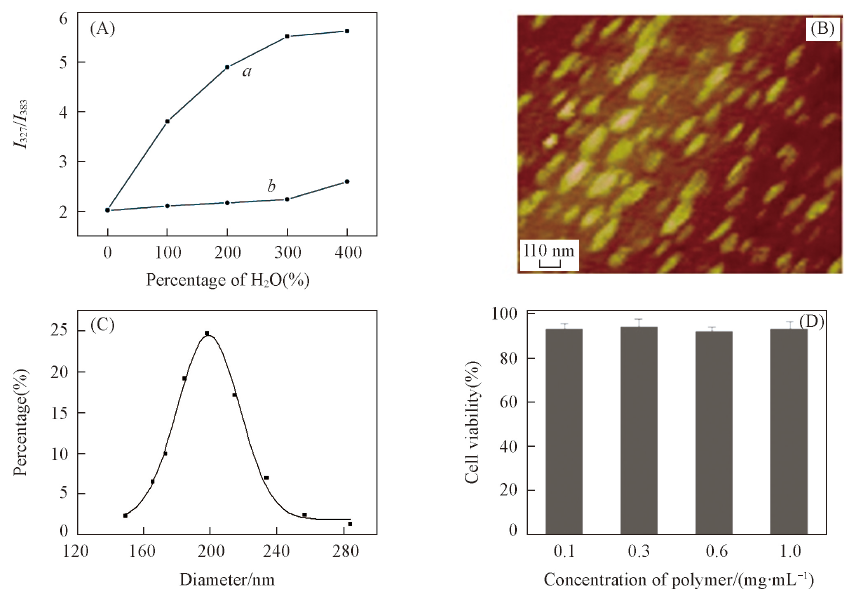
Fig.4 Change of I372/I383 of micelles before(a) and after(b) cross-link upon addition of H2O(A), AFM image(B) and dynamic light scattering(DLS)(C) of crosslinked micelles after laser irradiation(5 min) and in vitro cytotoxicity of crosslinked micelles against HeLa cells at different concentrations of polymer after 24 h incubation(D)
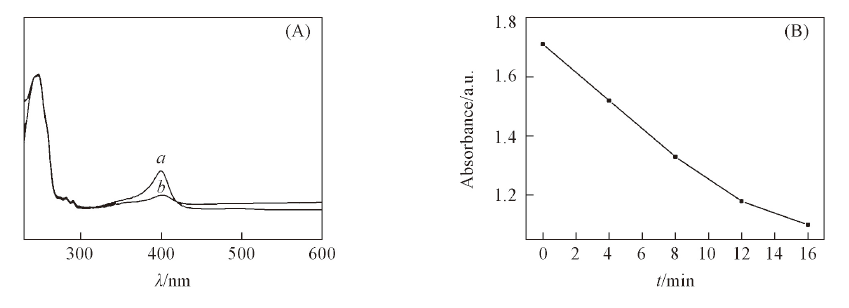
Fig.6 UV-Vis spectra of crosslinked micelles measured(A) and singlet oxygen produced with crosslinked micelles under laser irradiation(B) t/min: a. 0; b. 6.
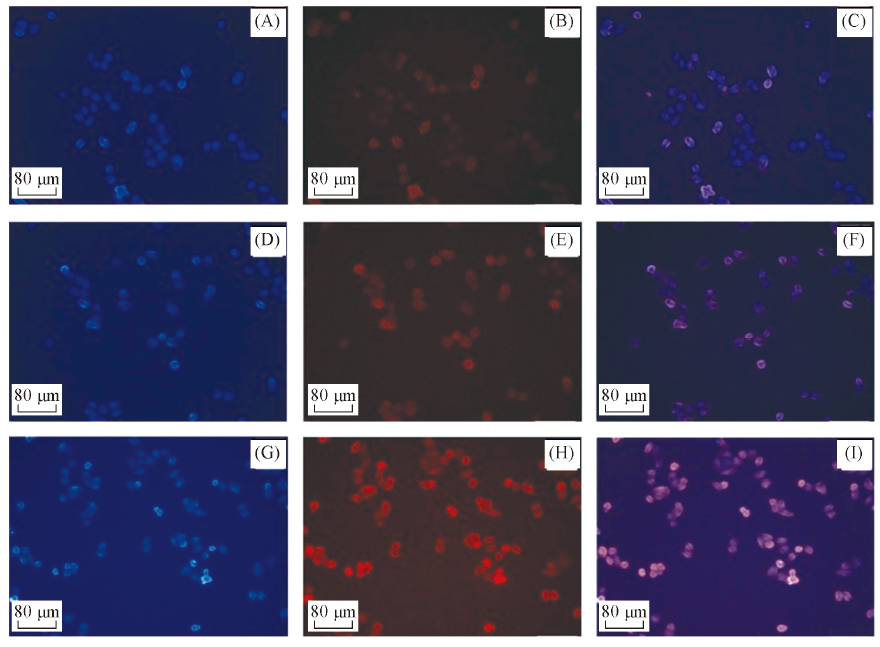
Fig.7 Fluorescence images of HeLa cells incubated with free 5FU(A—C), crosslinked micelles(D—F) and 5FU-loaded crosslinked micelles for 3 h(G—I)(H) and (I) are exposed to laser light source for 1 min after 1.5 h incubation. Each series can be classified to cell nucleus dyed in blue by Hoechst 33342(A, D, G), red by PI(B, E, H) and the merged images of both above(C, F, I), respectively.
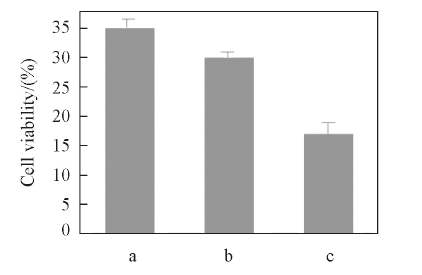
Fig.8 Cell viability with free 5FU(a), crosslinked micelles(b) and 5FU-loaded crosslinked micelles under laser irradiation for 24 h incubation(c)The cell viability profiles determined by MTT.
| [1] | Zhang, L. , Gu F., X. , Chan J., M. , Wang A., Z. , Langer R. S.. Farokhzad O., C. , Clin Pharmacol Ther., 2008, 83, 761- 769 |
| [2] | Luo, H. , Yang, C. , Zhang, X. , Zhao, M. , Jiang, D. , Xiao, J. , Yang, X. , Li, S. , Chem.Res. Chinese Universities, 2013, 29( 5), 906- 910 |
| [3] | Brigger, I. , Dubernet, C. , Couvreur, P. , Adv. Drug Delivery Rev., 2002, 54, 631- 651 |
| [4] | 王海, 张超, 张琳华, 刘兰霞, 郑义, 朱敦皖. 高等学校化学学报, 2014, 35( 10), 2239- 2245 |
| Wang, H. , Zhang, C. , Zhang L., H. , Liu L., X. , Zheng, Y. , Zhu D., W. , Chem. J. Chinese Universities, 2014, 35( 10), 2239- 2245 | |
| [5] | Bae Y., H. , Yin H., Q. , J. Controll Release, 2008, 131, 2- 4 |
| [6] | Burt H., M. , Zhang, X. , Toleikis, P. , Embree, L. , Hunter W., L. , Colloid Surface B, 1999, 16, 161- 171 |
| [7] | Savic, R. , Azzam, T. , Eisenberg, A. , Maysinger, D. , Langmuir, 2006, 22, 3570- 3578 |
| [8] | Torchilin, V. , Adv. Drug Deliv. Rev., 2011, 63, 131- 135 |
| [9] | Meng, F. , Cheng, R. , Deng, C. , Zhong, Z. , Mater. Today, 2012, 15, 436- 442 |
| [10] | Ladewig M., S. , Karl, S.E. , Hamelmann, V. , Helb H., M. , Scholl H. P., N. , Holz F., G. , Eter, N. , Graefe’s Arch. Clin. Exp.Ophthalmol., 2008, 246, 17- 25 |
| [11] | Krá, lová J. , Kejí , k Z. , Bí , za T. , Pouková, P. , Krá, l A. , Martá, sek P. , Krá, l V. , J. Med. Chem., 2010, 53, 128- 138 |
| [12] | Lin, W. , Kim, D. , Langmuir, 2011, 27, 12090- 12097 |
| [13] | Meng, F. , Hennink W., E. , Zhong, Z. , Biomaterials, 2009, 30, 2180- 2198 |
| [14] | Kim, D. , Lee E., S. , Oh K., T. , Zhong G., G. , Bae Y., H. , Small, 2008, 4, 2043- 2050 |
| [15] | Li Y., L. , Zhu, L. , Liu, Z. , Cheng, R. , Meng, F. , Cui, J. , Ji, S. , Zhong, Z. , Angew. Chem. Int. Ed., 2009, 48, 9914- 9918 |
| [16] | Wang Y., C. , Li, Y. , Sun T., M. , Xiong M., H. , Wu, J. , Yang Y., Y. , Wang, J. , Macromol. Rapid Commun., 2010, 31, 1201- 1206 |
| [17] | Cajot, S. , Lautram, N. , Passirani, C. , Jé, rôme C. , J. Controll Release, 2011, 152, 30- 36 |
| [18] | Rijcken C., J. , Snel C., J. , Schiffelers R., M. , CF van, Nostrum , Hennink W., E. , Biomaterials, 2007, 28, 5581- 5593 |
| [19] | Jiang J., Q. , Tong, X. , Morris, D. , Zhao, Y. , Macromolecules, 2006, 39, 4633- 4640 |
| [20] | Zhao, Y. , J. Mater. Chem., 2009, 19, 4887- 4895 |
| [21] | Goodwin A., P. , Mynar J., L. , Ma Y., Z. , Fleming G., R. , Frechet J. M., J. , J. Am. Chem. Soc., 2005, 127, 9952- 9953 |
| [22] | Lopez, A. , Monsan, P. , Biochimie, 1980, 62, 323- 329 |
| [23] | Kabanov A., V. , Nazarova I., R. , Astafieva I., V. , Batrakova E., V. , Alakhov V., Y. , Yaroslavov A., A. , Kabanov V., A. , Macromolecules, 1995, 28, 2303- 2314 |
| [24] | Hä, rtner S. , Kim H., C. , Hammpp, N. , J. Polym. Sci. Part A: Polym. Chem., 2007, 45, 2443- 2452 |
| [25] | Zhang, Y. , Xu, C. , Li, B. , RSC Adv., 2013, 3, 6044- 6050 |
| [26] | Moore J., S. , Stupp S., I. , Macromolecules, 1990, 23, 65- 70 |
| [27] | Lion, Y. , Delmelle, M. , van de Vorst, A. , Nature, 1976, 263, 442- 443 |
| [28] | Lion, Y. , Gandin, E. , van de vorst, A. , Photochem. Photobiol., 1980, 31, 305- 309 |
| [29] | Tong, R. , Hemmati H., D. , Langer, R. , Kohane D., S. , J. Am. Chem. Soc., 2012, 134, 8848- 8855 |
| [1] | ZHAO Yuhui, LI Mingle, LONG Saran, FAN Jiangli, PENG Xiaojun. Spectroscopic Characterization of Solvation Effect for a Polarity-Sensitive BDP [J]. Chem. J. Chinese Universities, 2020, 41(9): 2018. |
| [2] | YANG Jin,CAO Yan,ZHANG Naidong. Co-sensitization in the Visible Light/H2O2 System † [J]. Chem. J. Chinese Universities, 2020, 41(3): 505. |
| [3] | TONG Chunyi, TANG Fengxia, LIU Bin, LIAO Hongdong, LIU Xuanming. Preparation of Micro-spheres Co-carried Anti-tumor Drug and Sensitizer(5-Fu-COS/SeNP) and Their Inhibiting Tumorous Cellular Growths Activity† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1603. |
| [4] | LIU Tao, ZHANG Chong-Lei, CHEN Ping, TANG Guo-Qing, LIN Lie*. Interaction Between Methylene Blue and ctDNA by Time-resolved Spectroscopy [J]. Chem. J. Chinese Universities, 2011, 32(8): 1854. |
| [5] | LÜ Qing-Luan, ZHANG Miao, YUE Ning-Ning, GONG Bin, WANG Huai-You*. Production Mechanism Study of Singlet Oxygen in Copper Ion-Catalyzed Curcumin Using Fluorimetric Method and Its Determination in the Presence of Superoxide Anion Radical [J]. Chem. J. Chinese Universities, 2009, 30(3): 460. |
| [6] | YIN Jing-Bo, CHEN Hong-Dan, LUO Kun, ZHUANG Xiu-Li, CHEN Xue-Si, CAO Tian. Preparation and Properties of Biodegradable Microspheres Containing 5-Fluorouracil [J]. Chem. J. Chinese Universities, 2005, 26(6): 1174. |
| [7] | ZHANG Qiu-Lin, ZENG Ji-Chao., XU Xin-Hua, ZHOU Bing, LU Rui-Liang, CHEN Xiong, LI Yan-Jie . Synthesis and Biologic Activities of Glycerothiophospholipid Conjugate of Tegafur Containing Aryl-selenium Group in the Glycerol Skeleton [J]. Chem. J. Chinese Universities, 2005, 26(11): 2046. |
| [8] | LI Juan, LI Jing, QI Yan-Fei, WANG Hong-Fang, WANG En-Bo, HU Chang-Wen, XU Lin, WU Xin-Yu. Synthesis and Anti-liver Cancer Activity of 5-Fluorouracil Salt of 12-Tungstoboric Acid [J]. Chem. J. Chinese Universities, 2004, 25(6): 1010. |
| [9] | CHEN Jian-Yong, LIU Guan-Feng, SHEN Zhi-Quan . Embedment and Deliverance of 5-Fluorouracil from Blended Membrane of Silk Fibroin and 5-Fluorouracil [J]. Chem. J. Chinese Universities, 1999, 20(10): 1646. |
| [10] | MAO Man-Jun, TIAN Xuan, CHEN Yao-Zu. Syntheses and Antitumor Activities of Spin-labeled5-Fluorouracil Derivatives [J]. Chem. J. Chinese Universities, 1998, 19(3): 395. |
| [11] | FAN Chang-Lie, HU Bin, ZHUO Ren-Xi, DU Yang . Studies on the Tyrosine-Based Polyphosphate Antitumor Drugs [J]. Chem. J. Chinese Universities, 1997, 18(1): 149. |
| [12] | LUO Xuan-Gan, ZHUO Ren-Xi, LI Man-Qing . Studies of the Synthesis and Antitumor Activity of 5-Fluorouracil Derivatives Containing D-Aminoglucose Residue [J]. Chem. J. Chinese Universities, 1996, 17(9): 1416. |
| [13] | LING Ke-Qing. Photochemical Synthesis of 1,2-Dihydro-3H-indol-3-ones [J]. Chem. J. Chinese Universities, 1996, 17(6): 920. |
| [14] | LUO Yi, ZHUO Ren-xi, FAN Chang-Lie. Synthesis of Crosslinked Polyphosphates and Drug Release [J]. Chem. J. Chinese Universities, 1995, 16(10): 1633. |
| [15] | SUN Chan-Jun, WANG Yi-Gui, CHEN Zai-Cheng, XUE Peng, HU Wei-Feng, XU Bei-Li, ZHAO Yao-Ran, WANG Mei-Ling. Studies on the Synthesis of Glycosides(Ⅳ)─Synthesis of N-Glucuronides of 5-Fluorouracil and Their Antitumor Activities [J]. Chem. J. Chinese Universities, 1994, 15(8): 1168. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||