

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (3): 523.doi: 10.7503/cjcu20140895
• Physical Chemistry • Previous Articles Next Articles
CHAO Jingdi2, HE Hong1,2,*( ), SONG Liyun2, FANG Yujiao2, LIANG Quanming1, ZHANG Guizhen1,2, QIU Wenge1,2, ZHANG Ran2
), SONG Liyun2, FANG Yujiao2, LIANG Quanming1, ZHANG Guizhen1,2, QIU Wenge1,2, ZHANG Ran2
Received:2014-10-09
Online:2015-03-10
Published:2015-01-30
Contact:
HE Hong
E-mail:hehong@bjut.edu.cn
Supported by:CLC Number:
TrendMD:
CHAO Jingdi, HE Hong, SONG Liyun, FANG Yujiao, LIANG Quanming, ZHANG Guizhen, QIU Wenge, ZHANG Ran. Promotional Effect of Pr-Doping on the NH3-SCR Activity over the V2O5-MoO3/TiO2 Catalyst†[J]. Chem. J. Chinese Universities, 2015, 36(3): 523.
| Sample | Content of Pr(%) | SBET/(m2·g-1) | Pore volume/(cm3·g-1) | Crystallite size/nm |
|---|---|---|---|---|
| TiO2 | 34.8 | 0.06 | 29.8 | |
| 2Pr-TiO2 | 2 | 76.0 | 0.12 | 11.6 |
| 4Pr-TiO2 | 4 | 123.2 | 0.19 | 12.0 |
| 8Pr-TiO2 | 8 | 101.5 | 0.16 | 11.7 |
| 3V6MoTi | 15.7 | 0.03 | 33.6 | |
| 3V6Mo2PrTi | 2 | 64.5 | 0.10 | 12.2 |
| 3V6Mo4PrTi | 4 | 85.0 | 0.14 | 12.7 |
| 3V6Mo8PrTi | 8 | 87.6 | 0.16 | 12.6 |
Table 1 SBET, pore volume and average crystallite size of the Pr6O11-TiO2 supports and V2O5-MoO3/Pr6O11-TiO2 catalysts
| Sample | Content of Pr(%) | SBET/(m2·g-1) | Pore volume/(cm3·g-1) | Crystallite size/nm |
|---|---|---|---|---|
| TiO2 | 34.8 | 0.06 | 29.8 | |
| 2Pr-TiO2 | 2 | 76.0 | 0.12 | 11.6 |
| 4Pr-TiO2 | 4 | 123.2 | 0.19 | 12.0 |
| 8Pr-TiO2 | 8 | 101.5 | 0.16 | 11.7 |
| 3V6MoTi | 15.7 | 0.03 | 33.6 | |
| 3V6Mo2PrTi | 2 | 64.5 | 0.10 | 12.2 |
| 3V6Mo4PrTi | 4 | 85.0 | 0.14 | 12.7 |
| 3V6Mo8PrTi | 8 | 87.6 | 0.16 | 12.6 |
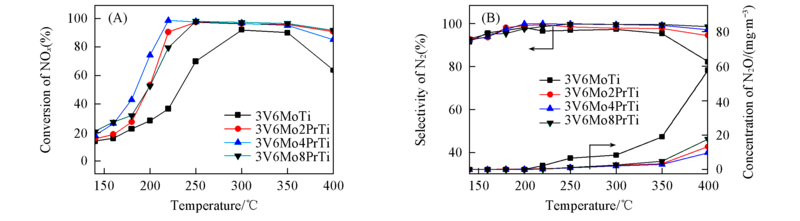
Fig.2 Conversion of NOx(A), selectivity of N2 and production of N2O(B) over the V2O5-MoO3/Pr6O11-TiO2 catalysts Test condition: 760 mg/m3 NH3, 1340 mg/m3 NO, 8%O2, He balance, space velocity: 3×104 h-1.
| Sample | A(V4+)/A(V2p) | A(Oα)/A(O1s) |
|---|---|---|
| 3V6MoTi | 0.12 | 0.15 |
| 3V6Mo4PrTi | 0.11 | 0.20 |
Table 2 XPS data of the catalysts*
| Sample | A(V4+)/A(V2p) | A(Oα)/A(O1s) |
|---|---|---|
| 3V6MoTi | 0.12 | 0.15 |
| 3V6Mo4PrTi | 0.11 | 0.20 |
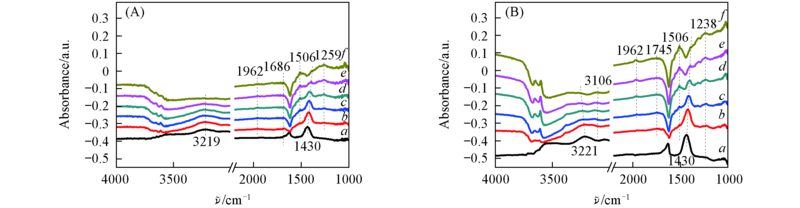
Fig.5 In situ DRIFTS spectra of NH3 adsorption over the 3V6MoTi(A) and 3V6Mo4PrTi(B) catalysts Temperature/℃: a. 40; b. 100; c. 150; d. 200; e. 250; f. 300.
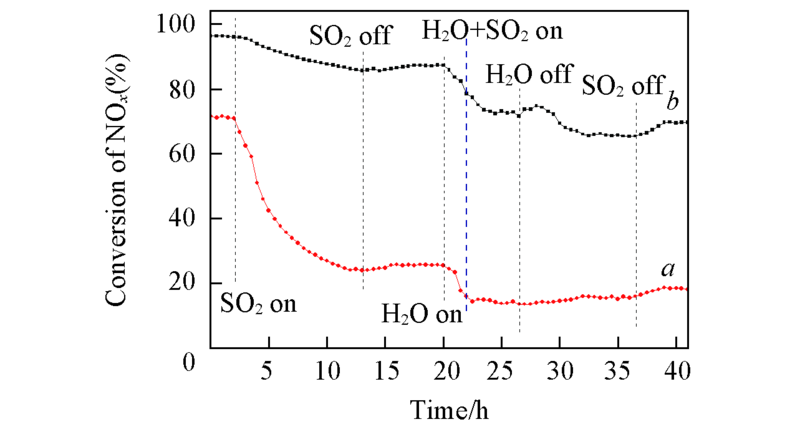
Fig.7 Effects of SO2 and H2O on the NOx conversion over 3V6MoTi(a) and 3V6Mo4PrTi(b) catalysts at 260 ℃ Reaction condition: NH3(760 mg/m3), NO(1340 mg/m3), O2(8%), SO2(1430 mg/m3), H2O(5%), N2 balance, space velocity: 3×104 h-1.
| [1] | Liu B. B., Zhang G. Z., He H., Li J. Z., Zi X. H., Qiu W. G., Dai H. X., Chem. J. Chinese Universities,2013, 34(8), 1936—1944 |
| (刘北北, 张桂臻, 何洪, 李金洲, 訾学红, 邱文革, 戴洪兴. 高等学校化学学报, 2013, 34(8), 1936—1944) | |
| [2] | Zhang S. L., Li H. Y., Zhong Q., Appl. Catal. A: Gen., 2012, 435/436, 156—162 |
| [3] | Li H. Y., Zhang S. L., Zhong Q., J. Colloid Interface Sci., 2013, 402, 190—195 |
| [4] | Pan Y. X., Zhao W., Zhong Q., Cai W., Li H. Y., J. Environ. Sci., 2013, 25(8), 1703—1711 |
| [5] | Zhao W., Zhong Q., Pan Y. X., Zhang R., Chem. Eng. J., 2013, 228, 815—823 |
| [6] | Shu Y., Wang H. C., Zhu J. W., Zhang F., Chem. Res. Chinese Universities,2014, 30(6), 1005—1010 |
| [7] | Shen B. X., Liu T., Zhao N., Yang X. Y., Deng L. D., J. Environ. Sci., 2010, 22(9), 1447—1454 |
| [8] | Amiridis M. D., Wachs I. E., Deo G., Jehng J. M., Kim D. S., J. Catal., 1996, 161(1), 247—253 |
| [9] | Lee K. J., Kumar P. A., Maqbool M. S., Rao K. N., Song K. H., Ha H. P., Appl. Catal. B: Environ., 2013, 142/143, 705—717 |
| [10] | Lee K. J., Maqbool M. S., Kumar P. A., Song K. H., Ha H. P., Catal. Lett., 2013, 143(10), 988—995 |
| [11] | Chen L., Li J. H., Ge M. F., Environ. Sci. Technol., 2010, 44(24), 9590—9596 |
| [12] | Li Q., Hou X. X., Yang H. S., Ma Z. X., Zheng J. W., Liu F., Zhang X. B., Yuang Z. Y., J. Mol. Catal. A: Chem., 2012, 356, 121—127 |
| [13] | Chen L., Li J. H., Ge M. F., J. Phys. Chem. C,2009, 113(50), 21177—21184 |
| [14] | Liu C. X., Chen L., Li J. H., Ma L., Arandiyan H., Du Y., Xu J. Y., Hao J. M., Environ. Sci. Technol., 2012, 46(11), 6182—6189 |
| [15] | Casanova M., Rocchini E., Trovarelli A., Schermanz K., Begsteiger I., J. Alloys Compd., 2006, 408—412, 1108—1112 |
| [16] | Casanova M., Schermanz K., Llorca J., Trovarelli A., Catal. Today,2012, 184(1), 227—236 |
| [17] | Gamba O., Moreno S., Molina R., Int. J. Hydrogen Energy,2011, 36(2), 1540—1550 |
| [18] | Gallego G. S., Marín J. G., Batiot-Dupeyrat C., Barrault J., Mondragón F., Appl. Catal. A: Gen., 2009, 369(1/2), 97—103 |
| [19] | Zhang Y. Y., Zhang J., Xu Q., Yan S., Zhao S. L., Luo G. X., Li C., Mater. Res. Bull., 2014, 53, 107—115 |
| [20] | Spadavecchia F., Cappelletti G., Ardizzone S., Ceotto M., Azzola M. S., Lo Presti L., Cerrato G., Falciola L., J. Phys. Chem. C,2012, 116(43), 23083—23093 |
| [21] | Huang H. Y., Gu X. T., Zhou J. H., Ji K., Liu H. L., Feng Y. Y., Catal. Commun., 2009, 11(1), 58—61 |
| [22] | Song L. Y., Zhan Z. C., Liu X. J., He H., Qiu W. G., Zi X. H., Chin. J. Catal., 2014, 35(7), 1030—1035 |
| (宋丽云, 展宗城, 刘晓军, 何洪, 邱文革, 訾学红. 催化学报, 2014, 35(7), 1030—1035) | |
| [23] | Gao X., Jiang Y., Zhong Y., Luo Z. Y., Cen K. F., J. Hazard. Mater., 2010, 174(1—3), 734—739 |
| [24] | Kim M. H., Ham S. W., Top. Catal., 2010, 53(7), 597—607 |
| [25] | Reddy B. M., Rao K. N., Reddy G. K., Bharali P., J. Mol. Catal. A: Chem., 2006, 253(1/2), 44—51 |
| [26] | Camposeco R., Castillo S., Mugica V., Mejia-Centeno I., Marin J., Chem. Eng. J., 2014, 242, 313—320 |
| [27] | Chiang T. H., Yeh H. C., J. Alloys Compd., 2014, 585, 535—541 |
| [28] | Guan B., Lin H., Zhu L., Tian B., Huang Z., Chem. Eng. J., 2012, 181/182, 307—322 |
| [29] | Shan W. P., Liu F. D., He H., Shi X. Y., Zhang C. B., Appl. Catal. B: Environ., 2012, 115/116, 100—106 |
| [30] | Liu J. D., Huang Z. G., Li Y. Z., Guo Q. Q., Li Q. Y., Chem. J. Chinese Universities,2014, 35(3), 589—595 |
| (刘建东, 黄张根, 李摇哲, 郭倩倩, 李巧艳. 高等学校化学学报, 2014, 35(3), 589—595) | |
| [31] | Wu Z. B., Jiang B. Q., Liu Y., Wang H. Q., Jin R. B., Environ. Sci. Technol., 2007, 41(16), 5812—5817 |
| [32] | Wang Y., Peng J., Zhou C., Lim Z. Y., Wu C. Z., Ye S., Wang W. G., Int. J. Hydrogen Energy,2014, 39(2) , 778—787 |
| [33] | Shi L. S., Chen W. F., Zhou X. Z., Zhao F. J., Li Y. X., Ceram. Int., 2014, 40(6), 8569—8574 |
| [34] | Chen L., Li J. H., Ge M. F., Chem. Eng. J., 2011, 170(2/3), 531—537 |
| [35] | Gutiérrez-Alejandre A., Ramírez J., Busca G., Langmuir,1998, 14(3), 630—639 |
| [36] | Pârvulescu V. I., Boghosian S., Pârvulescu V., Jung S. M., Grange P., J. Catal., 2003, 217(1), 172—185 |
| [37] | Liu Z. M., Zhang S. X., Li J. H., Ma L. L., Appl. Catal. B: Environ., 2014, 144, 90—95 |
| [38] | Kijlstra W. S., Brands D. S., Smit H. I., Poels E. K., Bliek A., J. Catal., 1997, 171(1), 219—230 |
| [39] | Centeno M. A., Carrizosa I., Odriozola J. A., Appl. Catal. B: Environ., 1998, 19(1), 67—73 |
| [40] | Peng Y., Liu C. X., Zhang X. Y., Li J. H., Appl. Catal. B: Environ., 2013, 140, 276—282 |
| [41] | Long R. Q., Yang R. T., J. Catal., 1999, 186(2), 254—268 |
| [42] | Topsøe N. Y., Topsøe H., Dumesic J. A., J. Catal., 1995, 151(1), 226—240 |
| [43] | Topsøe N. Y., Science, 1994, 265(5176), 1217—1219 |
| [44] | Zhang Y. M., Zhang H., Zhou L., H. W., Chem. Res. Chinese Universities,2014, 30(2), 279—283 |
| [45] | Pena D. A., Uphade B. S., Reddy E. P., Smirniotis P. G., J. Phys. Chem. B,2004, 108(28), 9927—9936 |
| [1] | ZHANG Ling,DUAN Hongchang,TAN Zhengguo,WU Qinming,MENG Xiangju,XIAO Fengshou. Recent Advances in the Preparation of 8MR Zeolites for the Selective Catalytic Reduction of NOx(NH3-SCR) in Diesel Engines † [J]. Chem. J. Chinese Universities, 2020, 41(1): 19. |
| [2] | LIU Ning, WANG Jiqiong, CHEN Biaohua, LI Yingxia, ZHANG Runduo. Study of Eight Membered Ring Zeolitic Catalyst of Cu/SAPO-35 over NH3-SCR† [J]. Chem. J. Chinese Universities, 2016, 37(10): 1817. |
| [3] | LIU Jiandong, HUANG Zhanggen, LI Zhe, GUO Qianqian, LI Qiaoyan. Ce Modification on Mn/TiO2/cordierite Monolithic Catalyst for Low-temperature NOx Reduction† [J]. Chem. J. Chinese Universities, 2014, 35(3): 589. |
| [4] | CHANG Gang, WANG Bin-Ju, ZHANG Jun, XIA Wen-Sheng*, WAN Hui-Lin*. Effect of Inclusion of Water Molecules Associated on Mechanism of H2O2+NH3H2O+ONH3 [J]. Chem. J. Chinese Universities, 2010, 31(9): 1820. |
| [5] | LIN Tao, XU Hai-Di, LI Wei, ZHANG Qiu-Lin, GONG Mao-Chu, CHEN Yao-Qiang*. Preparation of Mn-Fe/ZrO2-TiO2 Monolith Catalyst and Its Properties for Low-Temperature NH3-SCR Reaction [J]. Chem. J. Chinese Universities, 2009, 30(11): 2240. |
| [6] |
LIN Qi-Chun, LIN Wei-Ming, HAO Ji-Ming, LI Jun-Hua, FU Li-Xin.
Effect of Support Acidity on Selective Catalytic Reduction of NO by C3H6 over Cu-based Pillared Clay Catalyst [J]. Chem. J. Chinese Universities, 2006, 27(1): 85. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||