

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (7): 1433.doi: 10.7503/cjcu20131233
• Organic Chemistry • Previous Articles Next Articles
CHEN Ran1, AN Zhongwei1,2,*( ), CHEN Xinbing1, CHEN Pei1
), CHEN Xinbing1, CHEN Pei1
Received:2013-12-16
Online:2014-07-10
Published:2014-06-10
Contact:
AN Zhongwei
E-mail:gmecazw@163.com
Supported by:CLC Number:
TrendMD:
CHEN Ran, AN Zhongwei, CHEN Xinbing, CHEN Pei. Syntheses and Properties of Four-ring Liquid Crystals with Ethylene and Internal Alkyne Bridge†[J]. Chem. J. Chinese Universities, 2014, 35(7): 1433.
| Compd. | m. p./℃ | Yield(%) | Appearance | MS, m/z(RI, %) |
|---|---|---|---|---|
| 2-F | 82.7—83.3 | 65 | Light yellow solid | 298(M+, 9), 283(100), 155(28), 115(11), 55(8) |
| 3-F | 87.1—88.2 | 70 | Light yellow solid | 312(M+, 13), 297(100), 159(15) |
| 4-F | 81.4—82.7 | 68 | Light yellow solid | 326(M+, 10), 311(100), 159(24), 155(43), 115(15) |
| 5-F | 79.3—79.8 | 72 | Light yellow solid | 340(M+, 11), 325(100), 155(40), 115(12), 55(7) |
| 2-2 | 159.3—161.5 | 45 | White solid | 454(M+, 100), 329(66), 204(21) |
| 3-2 | 150.1—152.3 | 43 | White solid | 468(M+, 100), 343(27), 329(32), 204(17) |
| 4-2 | 133.8—136.1 | 45 | White solid | 482(M+, 100), 357(19), 329(29), 204(16) |
| 5-2 | 127.5—130.0 | 36 | White solid | 496(M+, 100), 371(9), 357(9), 343(13), 329(16), 204(14) |
| 3-3 | 139.1—141.7 | 40 | White solid | 482(M+, 65), 371(30), 343(100), 204(41) |
| 4-3 | 150.8—153.4 | 40 | White solid | 496(M+, 100), 357(21), 343(29), 204(14) |
| 5-3 | 139.5—142.1 | 40 | White solid | 510(M+, 100), 371(14), 343(27), 204(13) |
| 4-4 | 154.6—156.8 | 40 | White solid | 510(M+, 100), 357(50), 204(24) |
| 5-4 | 143.7—146.1 | 38 | White solid | 524(M+, 100), 371(23), 357(22), 204(17) |
| 5-5 | 143.2—145.6 | 43 | White solid | 538(M+, 100), 371(29), 204(11) |
Table 1 Physical data and MS results of new compounds
| Compd. | m. p./℃ | Yield(%) | Appearance | MS, m/z(RI, %) |
|---|---|---|---|---|
| 2-F | 82.7—83.3 | 65 | Light yellow solid | 298(M+, 9), 283(100), 155(28), 115(11), 55(8) |
| 3-F | 87.1—88.2 | 70 | Light yellow solid | 312(M+, 13), 297(100), 159(15) |
| 4-F | 81.4—82.7 | 68 | Light yellow solid | 326(M+, 10), 311(100), 159(24), 155(43), 115(15) |
| 5-F | 79.3—79.8 | 72 | Light yellow solid | 340(M+, 11), 325(100), 155(40), 115(12), 55(7) |
| 2-2 | 159.3—161.5 | 45 | White solid | 454(M+, 100), 329(66), 204(21) |
| 3-2 | 150.1—152.3 | 43 | White solid | 468(M+, 100), 343(27), 329(32), 204(17) |
| 4-2 | 133.8—136.1 | 45 | White solid | 482(M+, 100), 357(19), 329(29), 204(16) |
| 5-2 | 127.5—130.0 | 36 | White solid | 496(M+, 100), 371(9), 357(9), 343(13), 329(16), 204(14) |
| 3-3 | 139.1—141.7 | 40 | White solid | 482(M+, 65), 371(30), 343(100), 204(41) |
| 4-3 | 150.8—153.4 | 40 | White solid | 496(M+, 100), 357(21), 343(29), 204(14) |
| 5-3 | 139.5—142.1 | 40 | White solid | 510(M+, 100), 371(14), 343(27), 204(13) |
| 4-4 | 154.6—156.8 | 40 | White solid | 510(M+, 100), 357(50), 204(24) |
| 5-4 | 143.7—146.1 | 38 | White solid | 524(M+, 100), 371(23), 357(22), 204(17) |
| 5-5 | 143.2—145.6 | 43 | White solid | 538(M+, 100), 371(29), 204(11) |
| Compd. | 1H NMR(CDCl3, 300 MHz), δ | 13C NMR(CDCl3, 75 MHz), δ |
|---|---|---|
| 2-F | 0.81—0.93(m, 7H), 1.11—1.27(m, 4H), 1.43—1.50(m, 2H), 1.60(s, 6H), 1.70—1.80(m, 4H), 2.51—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 11.5, 30.0, 31.6, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.0, 39.6, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 3-F | 0.84—0.98(m, 7H), 1.10—1.18(m, 4H), 1.25—1.34(m, 2H), 1.42—1.48(m, 2H), 1.60(s, 6H), 1.69—1.78(m, 4H), 2.54—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.0 Hz, 2H) | 14.4, 20.0, 31.6, 33.3, 33.3, 33.3, 37.5, 37.5, 39.1, 39.8, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 4-F | 0.80—0.96(m, 7H), 1.09—1.19(m, 4H), 1.23—1.32(m, 4H), 1.42—1.50(m, 2H), 1.60(s, 6H), 1.69—1.80(m, 4H), 2.53—2.62(m, 2H), 7.07(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 14.2, 23.2, 29.3, 31.6, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 65.6, 82.3, 93.3, 119.9, 128.3, 131.6, 143.6 |
| 5-F | 0.82—0.99(m, 7H), 1.07—1.20(m, 4H), 1.21—1.32(m, 6H), 1.45—1.54(m, 2H), 1.61(s, 6H), 1.69—1.79(m, 4H), 2.53—2.65(m, 2H), 7.10(d, J=8.0 Hz, 2H), 7.32(d, J=8.0 Hz, 2H) | 14.1, 22.7, 26.7, 31.6, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.8, 39.0, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 2-2 | 0.74—0.85(m, 14H), 1.07—1.17(m, 8H), 1.38—1.44(m, 4H), 1.65—1.73(m, 8H), 2.51—2.57(m, 4H), 7.05(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 11.5, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-2 | 0.76—0.93(m, 14H), 1.03—1.10(m, 4H), 1.12—1.21(m, 4H), 1.21—1.28(m, 2H), 1.38—1.46(m, 4H), 1.62—1.75(m, 8H), 2.49—2.60(m, 4H), 7.06(d, J=7.9 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 11.5, 14.4, 20.0, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.6, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-2 | 0.84—0.99(m, 14H), 1.12—1.21(m, 6H), 1.21—1.24(m, 2H), 1.25—1.31(m, 4H), 1.45—1.53(m, 4H), 1.69—1.83(m, 8H), 2.55—2.68(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.41(d, J=8.0 Hz, 4H) | 11.5, 14.1, 23.0, 29.3, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-2 | 0.83—0.96(m, 14H), 1.12—1.22(m, 8H), 1.23—1.30(m, 6H), 1.46—1.54(m, 4H), 1.70—1.83(m, 8H), 2.57—2.72(m, 4H), 7.13(d, J=7.4 Hz, 4H), 7.42(d, J=7.5 Hz, 4H) | 11.5, 14.1, 22.7, 26.7, 30.0, 32.3, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-3 | 0.70—0.92(m, 14H), 1.01—1.16(m, 8H), 1.18—1.30(m, 4H), 1.36—1.47(m, 4H), 1.55—1.79(m, 8H), 2.44—2.65(m, 4H), 7.05(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.4, 20.0, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-3 | 0.72—0.93(m, 14H), 0.98—1.14(m, 8H), 1.16—1.29(m, 6H), 1.36—1.47(m, 4H), 1.59—1.83(m, 8H), 2.43—2.64(m, 4H), 7.06(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.1, 14.4, 20.0, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-3 | 0.77—0.94(m, 14H), 1.05—1.11(m, 8H), 1.16—1.23(m, 8H), 1.38—1.44(m, 4H), 1.63—1.72(m, 8H), 2.50—2.57(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 14.1, 14.4, 20.0, 22.7, 26.7, 32.3, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-4 | 0.84—0.95(m, 14H), 0.98—1.13(m, 8H), 1.15—1.28(m, 8H), 1.37—1.46(m, 4H), 1.58—1.79(m, 8H), 2.36—2.69(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.8 Hz, 4H) | 14.1, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-4 | 0.74—0.93(m, 14H), 1.02—1.12(m, 8H), 1.15—1.26(m, 10H), 1.35—1.47(m, 6H), 1.62—1.73(m, 8H), 2.48—2.60(m, 4H), 7.05(d, J=7.2 Hz, 4H), 7.34(d, J=7.0 Hz, 4H) | 14.1, 22.7, 23.0, 26.7, 29.3, 32.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-5 | 0.82—1.00(m, 14H), 1.10—1.19(m, 8H), 1.20—1.33(m, 12H), 1.45—1.53(m, 4H), 1.68—1.84(m, 8H), 2.52—2.71(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.42(d, J=8.0 Hz, 4H) | 14.1, 22.7, 26.7, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
Table 2 1H NMR and 13C NMR data of new compounds
| Compd. | 1H NMR(CDCl3, 300 MHz), δ | 13C NMR(CDCl3, 75 MHz), δ |
|---|---|---|
| 2-F | 0.81—0.93(m, 7H), 1.11—1.27(m, 4H), 1.43—1.50(m, 2H), 1.60(s, 6H), 1.70—1.80(m, 4H), 2.51—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 11.5, 30.0, 31.6, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.0, 39.6, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 3-F | 0.84—0.98(m, 7H), 1.10—1.18(m, 4H), 1.25—1.34(m, 2H), 1.42—1.48(m, 2H), 1.60(s, 6H), 1.69—1.78(m, 4H), 2.54—2.63(m, 2H), 7.08(d, J=8.0 Hz, 2H), 7.31(d, J=8.0 Hz, 2H) | 14.4, 20.0, 31.6, 33.3, 33.3, 33.3, 37.5, 37.5, 39.1, 39.8, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 4-F | 0.80—0.96(m, 7H), 1.09—1.19(m, 4H), 1.23—1.32(m, 4H), 1.42—1.50(m, 2H), 1.60(s, 6H), 1.69—1.80(m, 4H), 2.53—2.62(m, 2H), 7.07(d, J=8.0 Hz, 2H), 7.31(d, J=8.1 Hz, 2H) | 14.2, 23.2, 29.3, 31.6, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 65.6, 82.3, 93.3, 119.9, 128.3, 131.6, 143.6 |
| 5-F | 0.82—0.99(m, 7H), 1.07—1.20(m, 4H), 1.21—1.32(m, 6H), 1.45—1.54(m, 2H), 1.61(s, 6H), 1.69—1.79(m, 4H), 2.53—2.65(m, 2H), 7.10(d, J=8.0 Hz, 2H), 7.32(d, J=8.0 Hz, 2H) | 14.1, 22.7, 26.7, 31.6, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.8, 39.0, 65.6, 82.3, 93.2, 119.9, 128.3, 131.6, 143.6 |
| 2-2 | 0.74—0.85(m, 14H), 1.07—1.17(m, 8H), 1.38—1.44(m, 4H), 1.65—1.73(m, 8H), 2.51—2.57(m, 4H), 7.05(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 11.5, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-2 | 0.76—0.93(m, 14H), 1.03—1.10(m, 4H), 1.12—1.21(m, 4H), 1.21—1.28(m, 2H), 1.38—1.46(m, 4H), 1.62—1.75(m, 8H), 2.49—2.60(m, 4H), 7.06(d, J=7.9 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 11.5, 14.4, 20.0, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.6, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-2 | 0.84—0.99(m, 14H), 1.12—1.21(m, 6H), 1.21—1.24(m, 2H), 1.25—1.31(m, 4H), 1.45—1.53(m, 4H), 1.69—1.83(m, 8H), 2.55—2.68(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.41(d, J=8.0 Hz, 4H) | 11.5, 14.1, 23.0, 29.3, 30.0, 32.8, 32.8, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-2 | 0.83—0.96(m, 14H), 1.12—1.22(m, 8H), 1.23—1.30(m, 6H), 1.46—1.54(m, 4H), 1.70—1.83(m, 8H), 2.57—2.72(m, 4H), 7.13(d, J=7.4 Hz, 4H), 7.42(d, J=7.5 Hz, 4H) | 11.5, 14.1, 22.7, 26.7, 30.0, 32.3, 32.8, 32.8, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.6, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 3-3 | 0.70—0.92(m, 14H), 1.01—1.16(m, 8H), 1.18—1.30(m, 4H), 1.36—1.47(m, 4H), 1.55—1.79(m, 8H), 2.44—2.65(m, 4H), 7.05(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.4, 20.0, 33.3, 33.3, 33.3, 37.5, 37.6, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-3 | 0.72—0.93(m, 14H), 0.98—1.14(m, 8H), 1.16—1.29(m, 6H), 1.36—1.47(m, 4H), 1.59—1.83(m, 8H), 2.43—2.64(m, 4H), 7.06(d, J=8.0 Hz, 4H), 7.34(d, J=8.0 Hz, 4H) | 14.1, 14.4, 20.0, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-3 | 0.77—0.94(m, 14H), 1.05—1.11(m, 8H), 1.16—1.23(m, 8H), 1.38—1.44(m, 4H), 1.63—1.72(m, 8H), 2.50—2.57(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.9 Hz, 4H) | 14.1, 14.4, 20.0, 22.7, 26.7, 32.3, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 39.8, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 4-4 | 0.84—0.95(m, 14H), 0.98—1.13(m, 8H), 1.15—1.28(m, 8H), 1.37—1.46(m, 4H), 1.58—1.79(m, 8H), 2.36—2.69(m, 4H), 7.06(d, J=7.8 Hz, 4H), 7.34(d, J=7.8 Hz, 4H) | 14.1, 23.0, 29.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.8, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-4 | 0.74—0.93(m, 14H), 1.02—1.12(m, 8H), 1.15—1.26(m, 10H), 1.35—1.47(m, 6H), 1.62—1.73(m, 8H), 2.48—2.60(m, 4H), 7.05(d, J=7.2 Hz, 4H), 7.34(d, J=7.0 Hz, 4H) | 14.1, 22.7, 23.0, 26.7, 29.3, 32.3, 33.3, 33.3, 33.3, 37.2, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| 5-5 | 0.82—1.00(m, 14H), 1.10—1.19(m, 8H), 1.20—1.33(m, 12H), 1.45—1.53(m, 4H), 1.68—1.84(m, 8H), 2.52—2.71(m, 4H), 7.13(d, J=8.0 Hz, 4H), 7.42(d, J=8.0 Hz, 4H) | 14.1, 22.7, 26.7, 32.2, 33.3, 33.3, 33.3, 37.5, 37.5, 37.9, 39.1, 99.0, 120.6, 128.4, 131.5, 143.5 |
| n | m | Transition temperature /℃[Enthalpy change/(kJ·mol-1)] | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Heating processa | Cooling processb | ||||||||
| Cr | SA | N | I | I | N | SA | Cr | ||
| 2 | 2 | 69.9(10.7) | 160.8(23.2) | 220.4(4.0) | 219.9(-3.8) | 159.4(-20.5) | |||
| 3 | 2 | 50.7(1.9) | 151.4(20.1) | 233.5(6.2) | 232.4(-5.1) | 149.0(-15.9) | |||
| 4 | 2 | 73.1(2.6) | | 135.2(16.1) | 229.0(5.6) | 227.4(-5.3) | 132.3(-15.6) | ||
| 5 | 2 | 78.4(24.7) | 128.3(10.0) | 225.8(4.6) | 225.2(-4.4) | 127.2(-9.4) | |||
| 3 | 3 | 89.4(13.0) | 140.5(10.4) | 240.1(1.7) | 239.6(-2.1) | 138.4(-10.4) | |||
| 4 | 3 | 58.5(14.6) | 152.2(17.1) | 240.4(5.1) | 239.9(-4.6) | | 150.9(-16.5) | ||
| 5 | 3 | 88.0(24.0) | 140.2(12.0) | 239.3(4.4) | 238.8(-4.9) | 138.9(-10.1) | |||
| 4 | 4 | 66.8(17.5) | 155.4(6.6) | 235.7(3.9) | 235.3(-3.8) | 154.3(-16.1) | |||
| 5 | 4 | 79.5(22.1) | 144.9(0.6) | 146.4(7.9) | 234.8(4.5) | 234.3(-4.3) | 146.0(-4.2) | 144.3(-8.9) | 40.9(-18.5) |
| 5 | 5 | 117.6(32.6) | 144.5(8.9) | 156.8(5.1) | 234.0(5.0) | 233.8(-4.9) | 156.5(-4.9) | 141.8(-9.7) | 95.0(-29.5) |
Table 3 Phase transition temperature and enthalpy change of compounds n-m
| n | m | Transition temperature /℃[Enthalpy change/(kJ·mol-1)] | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Heating processa | Cooling processb | ||||||||
| Cr | SA | N | I | I | N | SA | Cr | ||
| 2 | 2 | 69.9(10.7) | 160.8(23.2) | 220.4(4.0) | 219.9(-3.8) | 159.4(-20.5) | |||
| 3 | 2 | 50.7(1.9) | 151.4(20.1) | 233.5(6.2) | 232.4(-5.1) | 149.0(-15.9) | |||
| 4 | 2 | 73.1(2.6) | | 135.2(16.1) | 229.0(5.6) | 227.4(-5.3) | 132.3(-15.6) | ||
| 5 | 2 | 78.4(24.7) | 128.3(10.0) | 225.8(4.6) | 225.2(-4.4) | 127.2(-9.4) | |||
| 3 | 3 | 89.4(13.0) | 140.5(10.4) | 240.1(1.7) | 239.6(-2.1) | 138.4(-10.4) | |||
| 4 | 3 | 58.5(14.6) | 152.2(17.1) | 240.4(5.1) | 239.9(-4.6) | | 150.9(-16.5) | ||
| 5 | 3 | 88.0(24.0) | 140.2(12.0) | 239.3(4.4) | 238.8(-4.9) | 138.9(-10.1) | |||
| 4 | 4 | 66.8(17.5) | 155.4(6.6) | 235.7(3.9) | 235.3(-3.8) | 154.3(-16.1) | |||
| 5 | 4 | 79.5(22.1) | 144.9(0.6) | 146.4(7.9) | 234.8(4.5) | 234.3(-4.3) | 146.0(-4.2) | 144.3(-8.9) | 40.9(-18.5) |
| 5 | 5 | 117.6(32.6) | 144.5(8.9) | 156.8(5.1) | 234.0(5.0) | 233.8(-4.9) | 156.5(-4.9) | 141.8(-9.7) | 95.0(-29.5) |
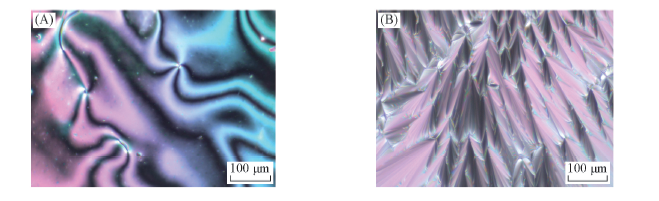
Fig.2 Polarizing optical micrographs of compound 5-5(200×) (A) A schlieren texture of the nematic phase at 226 ℃ on heating; (B) smectic A phase at 156 ℃ on cooling.
| Compd. | Structure | Transition temperature/℃ | ||||||
|---|---|---|---|---|---|---|---|---|
| Cr | SX1 | SX2 | SH | SB | N | I | ||
| 1 | 66 | 122 | 150.5 | 174 | 197.5 | 231 | ||
| 2[ | 76 | (63.5) | 148 | 190.5 | 215 | 228 | ||
| 3 | 185 | Above 300 | ||||||
| 4[ | 202 | Above 300 | ||||||
| 5 | 127 | | 155 | 210 | 220 | 325 | ||
| 6[ | 57 | | 62 | 226 | 248 | 312 | ||
| 3-3 | 140.5 | | 240.1 | |||||
| 5-3 | 140.2 | 239.3 | ||||||
Table 4 Phase transition temperatures of compounds 1—6, 3-3 and 5-3*
| Compd. | Structure | Transition temperature/℃ | ||||||
|---|---|---|---|---|---|---|---|---|
| Cr | SX1 | SX2 | SH | SB | N | I | ||
| 1 | 66 | 122 | 150.5 | 174 | 197.5 | 231 | ||
| 2[ | 76 | (63.5) | 148 | 190.5 | 215 | 228 | ||
| 3 | 185 | Above 300 | ||||||
| 4[ | 202 | Above 300 | ||||||
| 5 | 127 | | 155 | 210 | 220 | 325 | ||
| 6[ | 57 | | 62 | 226 | 248 | 312 | ||
| 3-3 | 140.5 | | 240.1 | |||||
| 5-3 | 140.2 | 239.3 | ||||||
| Compd. | 1 | 2-2 | 3-2 | 3-3 | 4-2 | 4-3 | 4-4 | 5-2 | 5-3 | 5-4 | 5-5 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Δn | 0.1998 | 0.3398 | 0.3358 | 0.3278 | 0.3278 | 0.3238 | 0.3198 | 0.3238 | 0.3198 | 0.3158 | 0.3118 |
Table 5 Optical anisotropies of compounds 1 and n-m
| Compd. | 1 | 2-2 | 3-2 | 3-3 | 4-2 | 4-3 | 4-4 | 5-2 | 5-3 | 5-4 | 5-5 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Δn | 0.1998 | 0.3398 | 0.3358 | 0.3278 | 0.3278 | 0.3238 | 0.3198 | 0.3238 | 0.3198 | 0.3158 | 0.3118 |
| [1] | Thomas G., Klaus G., Bernd R., Angew. Chem. Int. Ed., 2013, 52, 2—14 |
| [2] | Seung H. L., Surjya S. B., Heui S. J., Jeong K. U., J. Mater. Chem., 2012, 22(24), 11893—11903 |
| [3] | Kendhale A. M., Schenning A. P. H. J., Debije M. G., J. Mater. Chem. A, 2013, 1, 229—232 |
| [4] | Li L. W., Bryant D., Heugten T. V., Bos P. J., Opt. Express, 2013, 21(7), 8371—8381 |
| [5] | Zhao Y. L., Nan F. Y., Appl. Phys. Lett., 2013, 102(13), 131108-1—131108-5 |
| [6] | Reuter M., Garbat K., Vieweg N., Fischer B. M., Dᶏbrowski R., Koch M., Dziaduszek J., Urban S., J. Mater. Chem.C, 2013, 1(29), 4457—4463 |
| [7] | Buchnev O., Wallauer J., Walther M., Kaczmarek M., Zheludev N. I., Fedotov V. A., Appl. Phys. Lett., 2013, 103(14), 141904-1—141904-4 |
| [8] | Reuter M., Vieweg N., Fischer B. M., Mikulicz M., Koch M., Garbat K., Dᶏbrowski R., APL Mater., 2013, 1(1), 012107-1—012107-7 |
| [9] | Vieweg N., Jansen C., Koch M., Liquid Crystals and Their Applications in the THz Frequency Range, Springer Series in Optical Sciences, Berlin, 2013, 301—326 |
| [10] | Dziaduszek J., Kula P., Dᶏbrowski R., Drzewinski W., Garbat K., Urban S., Gauza S., Liq. Cryst., 2012, 39(2), 239—247 |
| [11] | Jiang Y., An Z. W., Chen P., Chen X. B., Zheng M. Y., Liq. Cryst., 2012, 39(4), 457—465 |
| [12] | Khoo I. C., Webster S., Kubo S., Youngblood W. J., Liou J. D., Mallouk T. E., Lin P., Hagan D. J., van Stryland E. W., J. Mater. Chem., 2009, 19(40), 7525—7531 |
| [13] | Fu Y. H., Chem. J. Chinese Universities, 2012, 33(7), 1490—1492 |
| (傅育红. 高等学校化学学报, 2012, 33(7), 1490—1492) | |
| [14] | Qiao W. Q., Chem. Res. Chinese Universities, 2012, 28(2), 239—241 |
| [15] | Chow H.F., Wan C. W., Low K. H., Yeung Y. Y., J. Org. Chem., 2001, 66, 1910—1913 |
| [16] | Cao X. Y., Zhang W., Zi H., Pei J., Org. Lett., 2004, 6(26), 4845—4848 |
| [17] | Zoltán N., Péter N., András K., Org. Lett., 2004, 6(26), 4917—4920 |
| [18] | Dietrich D., Lothar R., Textures of Liquid Crystal, Verlag Chemie, Weinheim, 1979, 170—212 |
| [19] | Hu M. G., An Z. W., Li J., Chin. J. Liq. Cryst. Displays, 2007, 22(2), 151—155 |
| (胡明刚, 安忠维, 李建. 液晶与显示, 2007, 22(2), 151—155) | |
| [20] | Dᶏbrowski R., Dziaduszek J., Czuprynski K., SPIE-The International Society for Optical Engineering, 1995, 2372, 235—241 |
| [21] | Takatsu H., Sasaki M., Tanaka Y., Sato H., Tolan-type Nematic Liquid Crystalline Compounds, GB 2155465A, 1985-09-25 |
| [22] | Chen X. B., An Z. W., Petrochemical Technology & Application, 2001, 19(6), 393—394 |
| (陈新兵, 安忠维. 石化技术与应用, 2001, 19(6), 393—394) | |
| [23] | Gauza S., Wang H. Y., Wen C. H.,Wu S. T., Alexander J. S., Dᶏbrowski R., Jpn. J. Appl. Phys., 2003, 42(6A), 3463—3466 |
| (Ed.: P, H, V, K) |
| [1] | HUANG Zhiyu, CHEN Honghui, MA Wenle, HUANG Yi, ZHU Dan, CHEN Yongsheng. Research Progress on Terahertz Stealth and Shielding Materials† [J]. Chem. J. Chinese Universities, 2019, 40(6): 1103. |
| [2] | LI Juanli, PENG Zenghui, LI Jian, HU Minggang, AN Zhongwei, ZHANG Lu. Synthesis and Properties of Novel Isothiocyanatotolane Liquid Crystals with Terminal Difluorovinyl Substitute† [J]. Chem. J. Chinese Universities, 2017, 38(10): 1788. |
| [3] | FANG Hongxia, ZHANG Qi, ZHANG Huili, QIN Dan, HONG Zhi, DU Yong. Terahertz Spectroscopic and Density Functional Theoretical Analysis of Cocrystal Between Olmesartan and Benzoic Acid† [J]. Chem. J. Chinese Universities, 2015, 36(12): 2504. |
| [4] | HE Ya-Ning, WANG Hao-Peng, WANG Xiao-Gong, ZHOU Qi-Xiang . Synthesis and Photo-responsive Behavior of a Novel Main-chain Azo Polyester [J]. Chem. J. Chinese Universities, 2003, 24(12): 2308. |
| [5] | JIN Ming, YANG Qing-Xin, LU Ran, PAN Ling-Yun, ZHAO Ying-Ying . Preparation of Azophenyl-substituted Pyrazoline Derivatives and Studies on Photoinduced Birefringence of Doped PMMA Films [J]. Chem. J. Chinese Universities, 2003, 24(1): 143. |
| [6] | ZHANG Jie, CHEN Xue-Nian, ZHANG Yu-Hua, YIN Yuan-Qi, HUANG Xiao-Ying. Synthesis and Characterization of C2Mo2Clusters Containing a Functionally Substituted Ligand [J]. Chem. J. Chinese Universities, 1999, 20(9): 1329. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||