

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (2): 262.doi: 10.7503/cjcu20180525
张成路( ), 宫荣庆, 杨敬怡, 孙晓娜, 李奕嶙, 王华玉, 宋府璐, 孙越冬
), 宫荣庆, 杨敬怡, 孙晓娜, 李奕嶙, 王华玉, 宋府璐, 孙越冬
收稿日期:2018-07-24
出版日期:2019-02-10
发布日期:2018-12-27
作者简介:联系人简介: 张成路, 男, 博士, 教授, 主要从事有机合成研究. E-mail:
基金资助:
ZHANG Chenglu*( ), GONG Rongqing, YANG Jingyi, SUN Xiaona, LI Yilin, WANG Huayu, SONG Fulu, SUN Yuedong
), GONG Rongqing, YANG Jingyi, SUN Xiaona, LI Yilin, WANG Huayu, SONG Fulu, SUN Yuedong
Received:2018-07-24
Online:2019-02-10
Published:2018-12-27
Contact:
ZHANG Chenglu
E-mail:zhangchenglu@lnnu.edu.cn
Supported by:摘要:
首先利用含有三嗪的芳香酰肼(3)构筑了1,3,4-噁二唑衍生物(5), 然后将化合物5与含有1,3,4-噻二唑的衍生物(6)拼合合成了18个目标分子. 利用红外光谱(IR)、 核磁共振波谱(NMR)和高分辨质谱(HRMS)等技术对其结构进行了表征. 考察了目标分子对细胞分裂周期25磷酸酯酶B(Cdc25B)和蛋白酪氨酸磷酸酯酶1B(PTP1B)的抑制活性. 结果表明, 有8个目标分子的抑制活性优于其阳性对照物, 有望成为潜在的Cdc25B抑制剂; 有12个目标分子的抑制活性优于其对照物, 有望成为潜在的PTP1B抑制剂.
中图分类号:
TrendMD:
张成路, 宫荣庆, 杨敬怡, 孙晓娜, 李奕嶙, 王华玉, 宋府璐, 孙越冬. 1,3,4-噁二唑硫醚酰胺为核心骨架的新型多杂环分子的合成及对Cdc25B与PTP1B的抑制活性. 高等学校化学学报, 2019, 40(2): 262.
ZHANG Chenglu,GONG Rongqing,YANG Jingyi,SUN Xiaona,LI Yilin,WANG Huayu,SONG Fulu,SUN Yuedong. Synthesis of Novel Polyheterocyclic Molecules with 1,3,4-Oxadiazole Thioetheramide as Core Framework and Their Inhibitory Activity on Cdc25B and PTP1B†. Chem. J. Chinese Universities, 2019, 40(2): 262.
| Compd. | m. p./℃ | Compd. | m. p./℃ |
|---|---|---|---|
| 1a | 173.1—173.9(173—174[ | 3c | 241.5—243.1(242—243[ |
| 1b | 113.7—114.8(114—115[ | 6a | 227.7—228.8(228—229[ |
| 1c | 114.1—114.9(114—115[ | 6b | 239.9—240.6(240—241[ |
| 2a | 289.1—289.7(289—290[ | 6c | 231.1—231.7(230—232[ |
| 2b | >300(>300[ | 6d | 243.6—244.2(243—244[ |
| 2c | 287.8—289.1(288—290[ | 6e | 162.5—163.1(162—163[ |
| 3a | 139.6—140.9(139—141[ | 6f | 169.6—170.9(170—171[ |
Table 1 Melting points of compounds 1a—1c, 2a—2c, 3a, 3c and 6a—6f
| Compd. | m. p./℃ | Compd. | m. p./℃ |
|---|---|---|---|
| 1a | 173.1—173.9(173—174[ | 3c | 241.5—243.1(242—243[ |
| 1b | 113.7—114.8(114—115[ | 6a | 227.7—228.8(228—229[ |
| 1c | 114.1—114.9(114—115[ | 6b | 239.9—240.6(240—241[ |
| 2a | 289.1—289.7(289—290[ | 6c | 231.1—231.7(230—232[ |
| 2b | >300(>300[ | 6d | 243.6—244.2(243—244[ |
| 2c | 287.8—289.1(288—290[ | 6e | 162.5—163.1(162—163[ |
| 3a | 139.6—140.9(139—141[ | 6f | 169.6—170.9(170—171[ |
| Compd. | Appearance | Yield(%) | m. p./℃ | IR(KBr), |
|---|---|---|---|---|
| 3b | White solid | 85.3 | 257.1—257.9 | 3419, 3033, 2960, 1694, 1630, 1578, 1519, 1453, 1276, 1172, 864 |
| 4a | White solid | 82.3 | 229.5—230.3 | 3412, 3025, 2979, 1715, 1618, 1499, 1401, 1250, 1172, 857 |
| 4b | White solid | 91.8 | 280.7—281.3 | 3416, 3036, 2971, 1702, 1616, 1566, 1513, 1428, 1172, 866 |
| 4c | White solid | 89.2 | 164.9—165.7 | 3326, 3051, 2967, 1708, 1611, 1572, 1506, 1394, 1173, 858 |
| 5a | Light yellow solid | 62.1 | 276.8—277.5 | 3418, 3025, 2934, 1611, 1493, 1401, 1362, 1238, 1165, 845 |
| 5b | Light yellow solid | 69.5 | >300 | 3431, 3051, 2966, 1604, 1499, 1388, 1335, 1244, 1159, 844 |
| 5c | Light yellow solid | 56.2 | 281.7—282.6 | 3431, 3084, 2934, 1605, 1499, 1414, 1362, 1250, 1165, 845 |
Table 2 Appearance, yields, melting points and IR data of compounds 3b, 4a—4c and 5a—5c
| Compd. | Appearance | Yield(%) | m. p./℃ | IR(KBr), |
|---|---|---|---|---|
| 3b | White solid | 85.3 | 257.1—257.9 | 3419, 3033, 2960, 1694, 1630, 1578, 1519, 1453, 1276, 1172, 864 |
| 4a | White solid | 82.3 | 229.5—230.3 | 3412, 3025, 2979, 1715, 1618, 1499, 1401, 1250, 1172, 857 |
| 4b | White solid | 91.8 | 280.7—281.3 | 3416, 3036, 2971, 1702, 1616, 1566, 1513, 1428, 1172, 866 |
| 4c | White solid | 89.2 | 164.9—165.7 | 3326, 3051, 2967, 1708, 1611, 1572, 1506, 1394, 1173, 858 |
| 5a | Light yellow solid | 62.1 | 276.8—277.5 | 3418, 3025, 2934, 1611, 1493, 1401, 1362, 1238, 1165, 845 |
| 5b | Light yellow solid | 69.5 | >300 | 3431, 3051, 2966, 1604, 1499, 1388, 1335, 1244, 1159, 844 |
| 5c | Light yellow solid | 56.2 | 281.7—282.6 | 3431, 3084, 2934, 1605, 1499, 1414, 1362, 1250, 1165, 845 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ |
|---|---|
| 3b | 9.46(s, 1H, NH), 7.98(d,J=8.4 Hz, 2H, PhH), 7.87(d, J=8.5 Hz, 2H, PhH), 3.82(s, 3H, OCH3), 3.52—3.49(m, 8H, pyrrolidine), 1.91(s, 8H, pyrrolidine) |
| 4a | 9.53(s, 1H, NH), 9.34(s, 1H, NH), 7.74(s, 4H, PhH), 4.42(s, 2H, NH2), 3.71(s, 8H, morpholine), 3.63(s, 8H, morpholine) |
| 4b | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.91(d,J=8.3 Hz, 2H, PhH), 7.74(d, J=8.3 Hz, 2H, PhH), 4.36(s, 2H, NH2), 3.50—3.45(m, 8H, pyrrolidine), 1.90(s, 8H, pyrrolidine) |
| 4c | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.74(q,J=8.8 Hz, 4H, PhH), 4.42(s, 2H, NH2), 3.71(d, J=4.35 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) |
| 5a | 14.49(s, 1H, SH), 9.59(s, 1H, NH), 7.93(d,J=8.6 Hz, 2H, PhH), 7.81(d, J=8.7 Hz, 2H, PhH), 3.74(s, 8H, morpholine), 3.65(s, 8H, morpholine) |
| 5b | 13.38(s, 1H, SH), 9.45(s, 1H, NH), 8.05(d,J=8.7 Hz, 2H, PhH), 7.98(d, J=8.55 Hz, 2H, PhH), 3.51—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) |
| 5c | 13.84(s, 1H, SH), 9.43(s, 1H, NH), 7.92(d,J=8.75 Hz, 2H, PhH), 7.78(d, J=8.7 Hz, 2H, PhH), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) |
Table 3 1H NMR data of compounds 3b, 4a—4c and 5a—5c
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ |
|---|---|
| 3b | 9.46(s, 1H, NH), 7.98(d,J=8.4 Hz, 2H, PhH), 7.87(d, J=8.5 Hz, 2H, PhH), 3.82(s, 3H, OCH3), 3.52—3.49(m, 8H, pyrrolidine), 1.91(s, 8H, pyrrolidine) |
| 4a | 9.53(s, 1H, NH), 9.34(s, 1H, NH), 7.74(s, 4H, PhH), 4.42(s, 2H, NH2), 3.71(s, 8H, morpholine), 3.63(s, 8H, morpholine) |
| 4b | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.91(d,J=8.3 Hz, 2H, PhH), 7.74(d, J=8.3 Hz, 2H, PhH), 4.36(s, 2H, NH2), 3.50—3.45(m, 8H, pyrrolidine), 1.90(s, 8H, pyrrolidine) |
| 4c | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.74(q,J=8.8 Hz, 4H, PhH), 4.42(s, 2H, NH2), 3.71(d, J=4.35 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) |
| 5a | 14.49(s, 1H, SH), 9.59(s, 1H, NH), 7.93(d,J=8.6 Hz, 2H, PhH), 7.81(d, J=8.7 Hz, 2H, PhH), 3.74(s, 8H, morpholine), 3.65(s, 8H, morpholine) |
| 5b | 13.38(s, 1H, SH), 9.45(s, 1H, NH), 8.05(d,J=8.7 Hz, 2H, PhH), 7.98(d, J=8.55 Hz, 2H, PhH), 3.51—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) |
| 5c | 13.84(s, 1H, SH), 9.43(s, 1H, NH), 7.92(d,J=8.75 Hz, 2H, PhH), 7.78(d, J=8.7 Hz, 2H, PhH), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) |
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(positive-SIMS), m/z(calcd. ), [M+H]+ |
|---|---|---|---|---|
| TZOA-1 | White solid | 67.6 | 279.8—280.8 | 660.1935(660.1923) |
| TZOA-2 | White solid | 68.9 | 282.9—283.7 | 674.2088(674.2080) |
| TZOA-3 | Light pink solid | 73.9 | 273.1—273.9 | 690.2025(690.2029) |
| TZOA-4 | Brownish solid | 65.1 | 259.7—260.5 | 694.1542(694.1534) |
| TZOA-5 | Yellow solid | 73.2 | 287.6—288.5 | 705.1782(705.1774) |
| TZOA-6 | White solid | 76.1 | 253.2—253.8 | 728.1787(728.1797) |
| TZOA-7 | Yellow solid | 75.8 | 250.7—251.5 | 628.2014(628.2025) |
| TZOA-8 | White solid | 66.9 | 269.8—270.3 | 642.2191(642.2182) |
| TZOA-9 | White solid | 73.9 | 255.5—256.3 | 658.2138(658.2131) |
| TZOA-10 | White solid | 79.9 | 265.1—265.7 | 662.1640(662.1635) |
| TZOA-11 | Yellow solid | 77.5 | 262.6—263.2 | 673.1883(673.1876) |
| TZOA-12 | Yellow solid | 79.1 | 284.2—285.0 | 696.1907(696.1899) |
| TZOA-13 | Yellow solid | 78.2 | 252.7—253.5 | 656.2331(656.2338) |
| TZOA-14 | White solid | 76.8 | 248.9—249.6 | 670.2489(670.2495) |
| TZOA-15 | White solid | 78.2 | 271.7—272.3 | 686.2453(686.2444) |
| TZOA-16 | White solid | 73.1 | 254.3—254.9 | 690.1952(690.1948) |
| TZOA-17 | Yellow solid | 77.3 | >300 | 701.2195(701.2189) |
| TZOA-18 | White solid | 79.9 | >300 | 724.2219(724.2212) |
Table 4 Appearance, yields, melting points and HRMS data of compounds TZOA-1—TZOA-18
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(positive-SIMS), m/z(calcd. ), [M+H]+ |
|---|---|---|---|---|
| TZOA-1 | White solid | 67.6 | 279.8—280.8 | 660.1935(660.1923) |
| TZOA-2 | White solid | 68.9 | 282.9—283.7 | 674.2088(674.2080) |
| TZOA-3 | Light pink solid | 73.9 | 273.1—273.9 | 690.2025(690.2029) |
| TZOA-4 | Brownish solid | 65.1 | 259.7—260.5 | 694.1542(694.1534) |
| TZOA-5 | Yellow solid | 73.2 | 287.6—288.5 | 705.1782(705.1774) |
| TZOA-6 | White solid | 76.1 | 253.2—253.8 | 728.1787(728.1797) |
| TZOA-7 | Yellow solid | 75.8 | 250.7—251.5 | 628.2014(628.2025) |
| TZOA-8 | White solid | 66.9 | 269.8—270.3 | 642.2191(642.2182) |
| TZOA-9 | White solid | 73.9 | 255.5—256.3 | 658.2138(658.2131) |
| TZOA-10 | White solid | 79.9 | 265.1—265.7 | 662.1640(662.1635) |
| TZOA-11 | Yellow solid | 77.5 | 262.6—263.2 | 673.1883(673.1876) |
| TZOA-12 | Yellow solid | 79.1 | 284.2—285.0 | 696.1907(696.1899) |
| TZOA-13 | Yellow solid | 78.2 | 252.7—253.5 | 656.2331(656.2338) |
| TZOA-14 | White solid | 76.8 | 248.9—249.6 | 670.2489(670.2495) |
| TZOA-15 | White solid | 78.2 | 271.7—272.3 | 686.2453(686.2444) |
| TZOA-16 | White solid | 73.1 | 254.3—254.9 | 690.1952(690.1948) |
| TZOA-17 | Yellow solid | 77.3 | >300 | 701.2195(701.2189) |
| TZOA-18 | White solid | 79.9 | >300 | 724.2219(724.2212) |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
|---|---|---|
| TZOA-1 | 13.17(s, 1H, NH), 9.54(s, 1H, NH), 7.91—7.85(m, 6H, PhH), 7.50—7.42(m, 3H, PhH), 4.32(s, 2H, CH2), 3.72(d, J=4.15 Hz, 8H, morpholine), 3.64(d, J=4.05 Hz, 8H, morpholine) | 168.13, 165.06, 164.64, 163.94, 162.89, 159.77, 143.78, 131.51, 129.54, 129.06, 126.96, 126.44, 119.33, 115.44, 65.96, 43.38, 37.71 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-2 | 9.55(s, 1H, NH), 7.91(q,J=8.6 Hz, 4H, PhH), 7.73(d, J=7.65 Hz, 2H, PhH), 7.27(d, J=7.8 Hz, 2H, Ph), 4.18(s, 2H, CH2), 3.74(s, 8H, morpholine), 3.66(s, 8H, morpholine), 2.35(s, 3H, CH3) | 169.90, 168.85, 164.71, 164.66, 163.96, 157.59, 143.64, 138.00, 130.27, 129.38, 126.91, 125.93, 119.36, 115.66, 65.97, 43.38, 20.80 |
| TZOA-3 | 13.01(s, 1H, NH), 9.54(s, 1H, NH), 7.91(d,J=8.7 Hz, 2H, PhH), 7.86(d, J=8.6 Hz, 2H, PhH), 7.81(d, J=8.5 Hz, 2H, PhH), 7.04(d, J=8.5 Hz, 2H, Ph), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.72(d, J=3.6 Hz, 8H, morpholine), 3.64(d, J=3.55 Hz, 8H, morpholine) | 168.53, 164.93, 164.63, 163.93, 163.25, 160.10, 158.93, 143.73, 127.73, 126.94, 124.62, 119.32, 115.50, 114.42, 65.96, 55.23, 43.36, 38.47 |
| TZOA-4 | 13.16(s, 1H, NH), 9.55(s, 1H, NH), 7.92(t,J=8.4 Hz, 4H, PhH), 7.87(d, J=8.75 Hz, 2H, PhH), 7.58(d, J=8.05 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.73(d, J=3.3 Hz, 8H, morpholine), 3.65(s, 8H, morpholine) | 167.96, 165.09, 164.63, 163.94, 163.27, 162.79, 159.99, 143.80, 131.36, 129.66, 129.10, 126.97, 126.48, 119.32, 115.41, 65.96, 43.37, 37.49 |
| TZOA-5 | 9.53(s, 1H, NH), 8.29(d,J=8.5 Hz, 2H, PhH), 8.09(d, J=8.5 Hz, 2H, PhH), 7.88(q, J=8.75 Hz, 4H, PhH), 4.22(s, 2H, CH2), 3.72(d, J=4.05 Hz, 8H, morpholine), 3.64(d, J=3.9 Hz, 8H, morpholine) | 167.75, 165.12, 164.63, 163.93, 162.68, 160.23, 143.81, 131.20, 129.76, 129.11, 126.96, 126.53, 119.31, 115.38, 65.96, 43.36, 37.27 |
| TZOA-6 | 13.32(s, 1H, NH), 9.54(s, 1H, NH), 8.14(d,J=8.2 Hz, 2H, PhH), 7.90(d, J=8.75 Hz, 2H, PhH), 7.85(t, J=8.25 Hz, 4H, PhH), 4.43(s, 2H, CH2), 3.71(s, 8H, morpholine), 3.64(d, J=3.5 Hz, 8H, morpholine) | 167.59, 165.23, 164.62, 163.92, 162.36, 162.22, 159.26, 143.85, 134.62, 129.86, 129.61, 127.29, 126.97, 126.09, 126.07, 125.02, 122.85, 119.30, 115.32, 65.95, 43.36, 36.68 |
| TZOA-7 | 13.18(s, 1H, NH), 9.38(s, 1H, NH), 8.05(d,J=8.65 Hz, 2H, PhH), 7.89(d, J=7.7 Hz, 2H, PhH), 7.83(d, J =8.65 Hz, 2H, PhH), 7.50—7.43(m, 3H, PhH), 4.33(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.16, 164.75, 164.41, 163.04, 162.60, 159.01, 144.12, 131.44, 128.99, 128.67, 126.45, 126.01, 118.65, 114.55, 45.31, 37.79, 24.46, 24.29 |
| TZOA-8 | 9.37(s, 1H, NH), 8.06(d,J=8.4 Hz, 2H, PhH), 7.84(d, J=8.35 Hz, 2H, PhH), 7.72(d, J=7.6 Hz, 2H, PhH), 7.26(d, J=7.8 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 2.33(s, 3H, CH3), 1.89(s, 8H, pyrrolidine) | 169.78, 164.83, 163.75, 163.39, 162.95, 157.75, 144.36, 138.10, 130.15, 129.40, 126.77, 125.97, 119.00, 115.05, 45.66, 24.82, 24.65, 20.82 |
| TZOA-9 | 13.00(s, 1H, NH), 9.37(s, 1H, NH), 8.05(d,J=8.9 Hz, 2H, PhH), 7.83—7.79(m, 4H, PhH), 7.04(d, J=8.9 Hz, 2H, PhH), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.64, 165.01, 163.38, 163.17, 162.94, 160.06, 158.79, 144.43, 127.71, 126.79, 124.68, 118.99, 114.93, 114.41, 55.23, 45.64, 38.52, 24.81, 24.64 |
| TZOA-10 | 9.37(s, 1H, NH), 8.06(d,J=8.3 Hz, 2H, PhH), 7.84(d, J=8.25 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.17(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 169.55, 168.12, 164.53, 163.22, 163.05, 162.62, 144.02, 132.73, 131.22, 128.59, 127.27, 126.44, 118.66, 114.69, 45.33, 24.47, 24.31 |
| TZOA-11 | 9.36(s, 1H, NH), 8.31(d,J=8.65 Hz, 2H, PhH), 8.14(d, J=8.65 Hz, 2H, PhH), 8.04(d, J=8.65 Hz, 2H, PhH), 7.82(t, J=8.45 Hz, 2H, PhH), 4.31(s, 2H, CH2), 3.50—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 169.89, 168.97, 164.47, 163.39, 163.04, 162.60, 155.91, 144.00, 132.55, 131.44, 128.55, 127.19, 126.43, 118.66, 114.71, 45.29, 24.48, 24.30 |
| TZOA-12 | 13.23(s, 1H, NH), 9.38(s, 1H, NH), 8.12(d, J=8.25 Hz, 2H, PhH), 8.05(d, J=8.6 Hz, 2H, PhH), 7.85—7.81(m, 4H, PhH), 4.36(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 167.99, 164.86, 163.04, 162.60, 162.23, 158.04, 144.15, 134.81, 129.16, 128.90, 126.72, 126.47, 125.67, 125.64, 124.73, 122.57, 118.64, 114.49, 45.30, 37.14, 24.46, 24.29 |
| TZOA-13 | 9.38(s, 1H, NH), 7.93(d, J=8.15 Hz, 2H, PhH), 7.87(d, J=8.55 Hz, 2H, PhH), 7.83(d, J=7.6 Hz, 2H, PhH), 7.44(t,J=7.65 Hz, 2H, PhH), 7.38—7.35(m, 1H, PhH), 4.17(s, 2H, CH2), 3.73(t, J=4.75 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.79, 165.18, 164.35, 164.05, 162.62, 160.20, 144.18, 131.24, 129.73, 129.10, 126.91, 126.52, 119.04, 115.08, 43.65, 37.31, 25.33, 24.31 |
| TZOA-14 | 9.38(s, 1H, NH), 7.92(d,J=7.55 Hz, 2H, PhH), 7.86(d, J=7.9 Hz, 2H, PhH), 7.71(d, J=6.95 Hz, 2H, PhH), 7.25(d, J=6.85 Hz, 2H, PhH), 4.16(s, 2H, CH2), 3.72(s, 8H, piperidine), 2.33(s, 3H, CH3), 1.62—1.51(m, 12H, piperidine) | 169.90, 168.84, 164.75, 164.37, 164.07, 163.90, 157.58, 144.01, 137.98, 130.28, 129.38, 126.87, 125.93, 119.07, 115.34, 43.67, 25.33, 24.32, 20.80 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-15 | 13.05(s, 1H, NH), 9.39(s, 1H, NH), 7.91(d,J=8.55 Hz, 2H, PhH), 7.83(q, J=5.45 Hz, 4H, PhH), 7.06(d, J=8.55 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.82(s, 3H, OCH3), 3.71(t, J=4.5 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) | 166.84, 164.89, 163.99, 163.69, 162.05, 160.30, 143.86, 127.79, 126.58, 123.15, 120.64, 118.68, 114.67, 114.24, 54.96, 43.30, 36.43, 24.98, 23.96 |
| TZOA-16 | 9.38(s, 1H, NH), 7.92(d,J=8.7 Hz, 2H, PhH), 7.86(t,J=8.55 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) | 170.27, 169.41, 164.78, 164.37, 164.06,163.81, 156.23, 144.02, 132.89, 131.82, 128.89, 127.53, 126.86, 119.07, 115.32, 43.66, 25.33, 24.32 |
| TZOA-17 | 9.38(s, 1H, NH), 8.29(d,J=8.85 Hz, 2H, PhH), 8.09(d, J=8.25 Hz, 2H, PhH), 7.92(d, J=8.25 Hz, 2H, PhH), 7.86(d, J=8.55 Hz, 2H, PhH), 4.20(s, 2H, CH2), 3.73(t, J=4.8 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.30, 165.29, 164.38, 164.33, 164.07, 162.26, 159.71, 144.22, 143.58, 134.59, 129.67, 129.23, 128.30, 126.92, 126.03, 119.02, 118.00, 114.99, 43.62, 36.59, 25.33, 24.32 |
| TZOA-18 | 13.30(s, 1H, NH), 9.39(s, 1H, NH), 8.15(d,J=8.1 Hz, 2H, PhH), 7.91(d, J=8.4 Hz, 2H, PhH), 7.87(d, J=8.1 Hz, 2H, PhH), 7.83(d, J=8.6 Hz, 2H, PhH), 4.43(s, 2H, CH2), 3.71(t, J=4.65 Hz, 8H, piperidine), 1.61—1.50(m, 12H, piperidine) | 167.18, 164.96, 164.00, 163.70, 161.89, 159.00, 143.88, 134.23, 129.57, 129.31, 126.98, 126.58, 125.73, 122.51, 118.69, 114.66, 43.31, 36.24, 24.98, 23.96 |
Table 5 1H NMR and 13C NMR data of compounds TZOA-1—TZOA-18
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
|---|---|---|
| TZOA-1 | 13.17(s, 1H, NH), 9.54(s, 1H, NH), 7.91—7.85(m, 6H, PhH), 7.50—7.42(m, 3H, PhH), 4.32(s, 2H, CH2), 3.72(d, J=4.15 Hz, 8H, morpholine), 3.64(d, J=4.05 Hz, 8H, morpholine) | 168.13, 165.06, 164.64, 163.94, 162.89, 159.77, 143.78, 131.51, 129.54, 129.06, 126.96, 126.44, 119.33, 115.44, 65.96, 43.38, 37.71 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-2 | 9.55(s, 1H, NH), 7.91(q,J=8.6 Hz, 4H, PhH), 7.73(d, J=7.65 Hz, 2H, PhH), 7.27(d, J=7.8 Hz, 2H, Ph), 4.18(s, 2H, CH2), 3.74(s, 8H, morpholine), 3.66(s, 8H, morpholine), 2.35(s, 3H, CH3) | 169.90, 168.85, 164.71, 164.66, 163.96, 157.59, 143.64, 138.00, 130.27, 129.38, 126.91, 125.93, 119.36, 115.66, 65.97, 43.38, 20.80 |
| TZOA-3 | 13.01(s, 1H, NH), 9.54(s, 1H, NH), 7.91(d,J=8.7 Hz, 2H, PhH), 7.86(d, J=8.6 Hz, 2H, PhH), 7.81(d, J=8.5 Hz, 2H, PhH), 7.04(d, J=8.5 Hz, 2H, Ph), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.72(d, J=3.6 Hz, 8H, morpholine), 3.64(d, J=3.55 Hz, 8H, morpholine) | 168.53, 164.93, 164.63, 163.93, 163.25, 160.10, 158.93, 143.73, 127.73, 126.94, 124.62, 119.32, 115.50, 114.42, 65.96, 55.23, 43.36, 38.47 |
| TZOA-4 | 13.16(s, 1H, NH), 9.55(s, 1H, NH), 7.92(t,J=8.4 Hz, 4H, PhH), 7.87(d, J=8.75 Hz, 2H, PhH), 7.58(d, J=8.05 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.73(d, J=3.3 Hz, 8H, morpholine), 3.65(s, 8H, morpholine) | 167.96, 165.09, 164.63, 163.94, 163.27, 162.79, 159.99, 143.80, 131.36, 129.66, 129.10, 126.97, 126.48, 119.32, 115.41, 65.96, 43.37, 37.49 |
| TZOA-5 | 9.53(s, 1H, NH), 8.29(d,J=8.5 Hz, 2H, PhH), 8.09(d, J=8.5 Hz, 2H, PhH), 7.88(q, J=8.75 Hz, 4H, PhH), 4.22(s, 2H, CH2), 3.72(d, J=4.05 Hz, 8H, morpholine), 3.64(d, J=3.9 Hz, 8H, morpholine) | 167.75, 165.12, 164.63, 163.93, 162.68, 160.23, 143.81, 131.20, 129.76, 129.11, 126.96, 126.53, 119.31, 115.38, 65.96, 43.36, 37.27 |
| TZOA-6 | 13.32(s, 1H, NH), 9.54(s, 1H, NH), 8.14(d,J=8.2 Hz, 2H, PhH), 7.90(d, J=8.75 Hz, 2H, PhH), 7.85(t, J=8.25 Hz, 4H, PhH), 4.43(s, 2H, CH2), 3.71(s, 8H, morpholine), 3.64(d, J=3.5 Hz, 8H, morpholine) | 167.59, 165.23, 164.62, 163.92, 162.36, 162.22, 159.26, 143.85, 134.62, 129.86, 129.61, 127.29, 126.97, 126.09, 126.07, 125.02, 122.85, 119.30, 115.32, 65.95, 43.36, 36.68 |
| TZOA-7 | 13.18(s, 1H, NH), 9.38(s, 1H, NH), 8.05(d,J=8.65 Hz, 2H, PhH), 7.89(d, J=7.7 Hz, 2H, PhH), 7.83(d, J =8.65 Hz, 2H, PhH), 7.50—7.43(m, 3H, PhH), 4.33(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.16, 164.75, 164.41, 163.04, 162.60, 159.01, 144.12, 131.44, 128.99, 128.67, 126.45, 126.01, 118.65, 114.55, 45.31, 37.79, 24.46, 24.29 |
| TZOA-8 | 9.37(s, 1H, NH), 8.06(d,J=8.4 Hz, 2H, PhH), 7.84(d, J=8.35 Hz, 2H, PhH), 7.72(d, J=7.6 Hz, 2H, PhH), 7.26(d, J=7.8 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 2.33(s, 3H, CH3), 1.89(s, 8H, pyrrolidine) | 169.78, 164.83, 163.75, 163.39, 162.95, 157.75, 144.36, 138.10, 130.15, 129.40, 126.77, 125.97, 119.00, 115.05, 45.66, 24.82, 24.65, 20.82 |
| TZOA-9 | 13.00(s, 1H, NH), 9.37(s, 1H, NH), 8.05(d,J=8.9 Hz, 2H, PhH), 7.83—7.79(m, 4H, PhH), 7.04(d, J=8.9 Hz, 2H, PhH), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.64, 165.01, 163.38, 163.17, 162.94, 160.06, 158.79, 144.43, 127.71, 126.79, 124.68, 118.99, 114.93, 114.41, 55.23, 45.64, 38.52, 24.81, 24.64 |
| TZOA-10 | 9.37(s, 1H, NH), 8.06(d,J=8.3 Hz, 2H, PhH), 7.84(d, J=8.25 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.17(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 169.55, 168.12, 164.53, 163.22, 163.05, 162.62, 144.02, 132.73, 131.22, 128.59, 127.27, 126.44, 118.66, 114.69, 45.33, 24.47, 24.31 |
| TZOA-11 | 9.36(s, 1H, NH), 8.31(d,J=8.65 Hz, 2H, PhH), 8.14(d, J=8.65 Hz, 2H, PhH), 8.04(d, J=8.65 Hz, 2H, PhH), 7.82(t, J=8.45 Hz, 2H, PhH), 4.31(s, 2H, CH2), 3.50—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 169.89, 168.97, 164.47, 163.39, 163.04, 162.60, 155.91, 144.00, 132.55, 131.44, 128.55, 127.19, 126.43, 118.66, 114.71, 45.29, 24.48, 24.30 |
| TZOA-12 | 13.23(s, 1H, NH), 9.38(s, 1H, NH), 8.12(d, J=8.25 Hz, 2H, PhH), 8.05(d, J=8.6 Hz, 2H, PhH), 7.85—7.81(m, 4H, PhH), 4.36(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 167.99, 164.86, 163.04, 162.60, 162.23, 158.04, 144.15, 134.81, 129.16, 128.90, 126.72, 126.47, 125.67, 125.64, 124.73, 122.57, 118.64, 114.49, 45.30, 37.14, 24.46, 24.29 |
| TZOA-13 | 9.38(s, 1H, NH), 7.93(d, J=8.15 Hz, 2H, PhH), 7.87(d, J=8.55 Hz, 2H, PhH), 7.83(d, J=7.6 Hz, 2H, PhH), 7.44(t,J=7.65 Hz, 2H, PhH), 7.38—7.35(m, 1H, PhH), 4.17(s, 2H, CH2), 3.73(t, J=4.75 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.79, 165.18, 164.35, 164.05, 162.62, 160.20, 144.18, 131.24, 129.73, 129.10, 126.91, 126.52, 119.04, 115.08, 43.65, 37.31, 25.33, 24.31 |
| TZOA-14 | 9.38(s, 1H, NH), 7.92(d,J=7.55 Hz, 2H, PhH), 7.86(d, J=7.9 Hz, 2H, PhH), 7.71(d, J=6.95 Hz, 2H, PhH), 7.25(d, J=6.85 Hz, 2H, PhH), 4.16(s, 2H, CH2), 3.72(s, 8H, piperidine), 2.33(s, 3H, CH3), 1.62—1.51(m, 12H, piperidine) | 169.90, 168.84, 164.75, 164.37, 164.07, 163.90, 157.58, 144.01, 137.98, 130.28, 129.38, 126.87, 125.93, 119.07, 115.34, 43.67, 25.33, 24.32, 20.80 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-15 | 13.05(s, 1H, NH), 9.39(s, 1H, NH), 7.91(d,J=8.55 Hz, 2H, PhH), 7.83(q, J=5.45 Hz, 4H, PhH), 7.06(d, J=8.55 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.82(s, 3H, OCH3), 3.71(t, J=4.5 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) | 166.84, 164.89, 163.99, 163.69, 162.05, 160.30, 143.86, 127.79, 126.58, 123.15, 120.64, 118.68, 114.67, 114.24, 54.96, 43.30, 36.43, 24.98, 23.96 |
| TZOA-16 | 9.38(s, 1H, NH), 7.92(d,J=8.7 Hz, 2H, PhH), 7.86(t,J=8.55 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) | 170.27, 169.41, 164.78, 164.37, 164.06,163.81, 156.23, 144.02, 132.89, 131.82, 128.89, 127.53, 126.86, 119.07, 115.32, 43.66, 25.33, 24.32 |
| TZOA-17 | 9.38(s, 1H, NH), 8.29(d,J=8.85 Hz, 2H, PhH), 8.09(d, J=8.25 Hz, 2H, PhH), 7.92(d, J=8.25 Hz, 2H, PhH), 7.86(d, J=8.55 Hz, 2H, PhH), 4.20(s, 2H, CH2), 3.73(t, J=4.8 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.30, 165.29, 164.38, 164.33, 164.07, 162.26, 159.71, 144.22, 143.58, 134.59, 129.67, 129.23, 128.30, 126.92, 126.03, 119.02, 118.00, 114.99, 43.62, 36.59, 25.33, 24.32 |
| TZOA-18 | 13.30(s, 1H, NH), 9.39(s, 1H, NH), 8.15(d,J=8.1 Hz, 2H, PhH), 7.91(d, J=8.4 Hz, 2H, PhH), 7.87(d, J=8.1 Hz, 2H, PhH), 7.83(d, J=8.6 Hz, 2H, PhH), 4.43(s, 2H, CH2), 3.71(t, J=4.65 Hz, 8H, piperidine), 1.61—1.50(m, 12H, piperidine) | 167.18, 164.96, 164.00, 163.70, 161.89, 159.00, 143.88, 134.23, 129.57, 129.31, 126.98, 126.58, 125.73, 122.51, 118.69, 114.66, 43.31, 36.24, 24.98, 23.96 |
| Compd. | Cdc25Ba | PTP1Bc | ||
|---|---|---|---|---|
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 1a | 1.55±1.34 | NAd | 2.99±3.88 | NA |
| 1b | 0.16±0.66 | NA | 1.01±0.05 | NA |
| 1c | 18.62±2.83 | NA | 24.47±1.22 | NA |
| 2a | 0.67±0.17 | NA | 4.77±6.25 | NA |
| 2b | 53.53±2.79 | 10.37±0.63 | 38.79±5.97 | NA |
| 2c | 37.76±7.95 | NA | 22.35±0.77 | NA |
| 3a | 4.85±0.23 | NA | 5.45±0.39 | NA |
| 3b | 53.48±1.06 | 5.62±1.52 | 84.63±4.76 | 9.18±0.43 |
| 3c | 0.32±1.52 | NA | 24.07±1.96 | NA |
| 4a | 1.64±0.69 | NA | 4.91±0.60 | NA |
| 4b | 8.93±3.84 | NA | 1.21±0.52 | NA |
| 4c | 1.41±3.43 | NA | 6.63±3.14 | NA |
| 5a | 61.81±3.58 | 12.82±1.46 | 31.13±4.69 | NA |
| 5b | 60.00±1.00 | 6.64±1.04 | 60.53±2.00 | 19.71±2.84 |
| 5c | 64.49±1.60 | 6.41±0.49 | 84.47±0.52 | 6.64±0.16 |
| 6a | 0.34±0.39 | NA | 15.64±5.71 | NA |
| 6b | 36.68±2.83 | NA | 85.71±2.80 | 12.24±0.11 |
| 6c | 0.02±0.35 | NA | 85.22±1.87 | 20.58±0.06 |
| Compd. | Cdc25Ba | PTP1Bc | ||
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 6d | 14.99±1.04 | NA | 91.00±1.53 | 7.21±0.11 |
| 6e | 64.59±0.69 | 9.92±0.74 | 87.74±2.21 | 17.28±2.24 |
| 6f | 25.69±2.93 | NA | 88.47±1.38 | 11.81±0.47 |
| TZOA-1 | 4.28±0.98 | NA | 31.91±0.81 | NA |
| TZOA-2 | 44.10±0.00 | NA | 31.77±0.32 | NA |
| TZOA-3 | 0.37±1.02 | NA | 31.85±1.09 | NA |
| TZOA-4 | 69.32±2.77 | 6.66±1.77 | 40.60±1.18 | NA |
| TZOA-5 | 70.23±3.06 | 3.32±0.06 | 44.83±4.20 | NA |
| TZOA-6 | 77.53±1.20 | 3.01±0.19 | 48.10±0.92 | NA |
| TZOA-7 | 74.97±0.17 | 3.36±0.13 | 84.42±1.00 | 7.04±0.73 |
| TZOA-8 | 24.30±4.25 | NA | 86.75±1.65 | 15.11±0.98 |
| TZOA-9 | 57.61±0.91 | 9.38±0.99 | 84.66±0.90 | 9.32±1.86 |
| TZOA-10 | 63.73±4.13 | 5.49±0.48 | 84.70±8.06 | 3.24±0.06 |
| TZOA-11 | 92.25±0.21 | 0.71±0.04 | 93.40±0.39 | 2.37±0.38 |
| TZOA-12 | 89.07±1.18 | 1.12±0.13 | 89.16±6.93 | 8.50±0.07 |
| TZOA-13 | 90.28±0.07 | 1.04±0.25 | 77.33±0.73 | 26.18±6.77 |
| TZOA-14 | 93.37±1.09 | 0.88±0.15 | 88.15±0.14 | 4.78±0.24 |
| TZOA-15 | 94.54±1.50 | 0.86±0.50 | 88.78±0.56 | 4.22±0.64 |
| TZOA-16 | 91.13±0.06 | 1.02±0.12 | 89.73±3.35 | 4.36±0.78 |
| TZOA-17 | 96.18±0.16 | 0.66±0.33 | 92.71±0.74 | 6.12±0.58 |
| TZOA-18 | 99.23±0.09 | 0.46±0.34 | 91.52±0.39 | 3.04±0.02 |
Table 6 Inhibitory activities of compounds 1—6 and TZOA-1—TZOA-18 against Cdc25B and PTP1B
| Compd. | Cdc25Ba | PTP1Bc | ||
|---|---|---|---|---|
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 1a | 1.55±1.34 | NAd | 2.99±3.88 | NA |
| 1b | 0.16±0.66 | NA | 1.01±0.05 | NA |
| 1c | 18.62±2.83 | NA | 24.47±1.22 | NA |
| 2a | 0.67±0.17 | NA | 4.77±6.25 | NA |
| 2b | 53.53±2.79 | 10.37±0.63 | 38.79±5.97 | NA |
| 2c | 37.76±7.95 | NA | 22.35±0.77 | NA |
| 3a | 4.85±0.23 | NA | 5.45±0.39 | NA |
| 3b | 53.48±1.06 | 5.62±1.52 | 84.63±4.76 | 9.18±0.43 |
| 3c | 0.32±1.52 | NA | 24.07±1.96 | NA |
| 4a | 1.64±0.69 | NA | 4.91±0.60 | NA |
| 4b | 8.93±3.84 | NA | 1.21±0.52 | NA |
| 4c | 1.41±3.43 | NA | 6.63±3.14 | NA |
| 5a | 61.81±3.58 | 12.82±1.46 | 31.13±4.69 | NA |
| 5b | 60.00±1.00 | 6.64±1.04 | 60.53±2.00 | 19.71±2.84 |
| 5c | 64.49±1.60 | 6.41±0.49 | 84.47±0.52 | 6.64±0.16 |
| 6a | 0.34±0.39 | NA | 15.64±5.71 | NA |
| 6b | 36.68±2.83 | NA | 85.71±2.80 | 12.24±0.11 |
| 6c | 0.02±0.35 | NA | 85.22±1.87 | 20.58±0.06 |
| Compd. | Cdc25Ba | PTP1Bc | ||
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 6d | 14.99±1.04 | NA | 91.00±1.53 | 7.21±0.11 |
| 6e | 64.59±0.69 | 9.92±0.74 | 87.74±2.21 | 17.28±2.24 |
| 6f | 25.69±2.93 | NA | 88.47±1.38 | 11.81±0.47 |
| TZOA-1 | 4.28±0.98 | NA | 31.91±0.81 | NA |
| TZOA-2 | 44.10±0.00 | NA | 31.77±0.32 | NA |
| TZOA-3 | 0.37±1.02 | NA | 31.85±1.09 | NA |
| TZOA-4 | 69.32±2.77 | 6.66±1.77 | 40.60±1.18 | NA |
| TZOA-5 | 70.23±3.06 | 3.32±0.06 | 44.83±4.20 | NA |
| TZOA-6 | 77.53±1.20 | 3.01±0.19 | 48.10±0.92 | NA |
| TZOA-7 | 74.97±0.17 | 3.36±0.13 | 84.42±1.00 | 7.04±0.73 |
| TZOA-8 | 24.30±4.25 | NA | 86.75±1.65 | 15.11±0.98 |
| TZOA-9 | 57.61±0.91 | 9.38±0.99 | 84.66±0.90 | 9.32±1.86 |
| TZOA-10 | 63.73±4.13 | 5.49±0.48 | 84.70±8.06 | 3.24±0.06 |
| TZOA-11 | 92.25±0.21 | 0.71±0.04 | 93.40±0.39 | 2.37±0.38 |
| TZOA-12 | 89.07±1.18 | 1.12±0.13 | 89.16±6.93 | 8.50±0.07 |
| TZOA-13 | 90.28±0.07 | 1.04±0.25 | 77.33±0.73 | 26.18±6.77 |
| TZOA-14 | 93.37±1.09 | 0.88±0.15 | 88.15±0.14 | 4.78±0.24 |
| TZOA-15 | 94.54±1.50 | 0.86±0.50 | 88.78±0.56 | 4.22±0.64 |
| TZOA-16 | 91.13±0.06 | 1.02±0.12 | 89.73±3.35 | 4.36±0.78 |
| TZOA-17 | 96.18±0.16 | 0.66±0.33 | 92.71±0.74 | 6.12±0.58 |
| TZOA-18 | 99.23±0.09 | 0.46±0.34 | 91.52±0.39 | 3.04±0.02 |
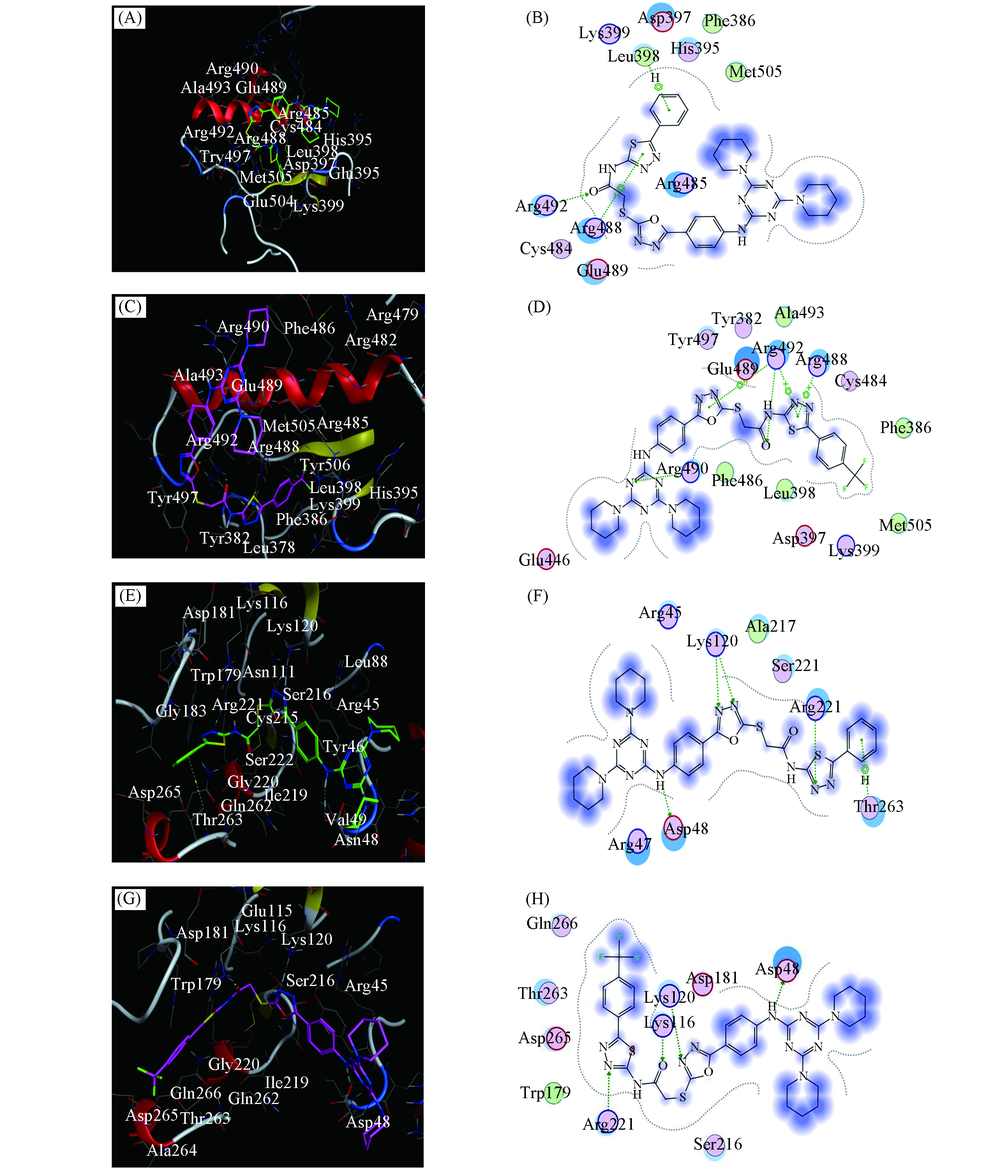
Fig.2 Molecular docking of compound TZOA-13, TZOA-18 with Cdc25B and PTP1B, respectively(A, B) TZOA-13 with Cdc25B; (C, D) TZOA-18 with Cdc25B; (E, F) TZOA-13 with PTP1B; (G, H) TZOA-18 with PTP1B.
| [1] | Kmar R., Kmar A., Jain S., Kaushik D., Eur. J. Med. Chem., 2011, 46(9), 3543-3550 |
| [2] | Husain A., Ahmad A., Alam M. M., Ajmal M., Ahuja P., Eur. J. Med. Chem., 2009, 44(9), 3798-3804 |
| [3] | Zhang X. M., Qiu M., Sun J., Zhang Y. B., Yang Y. S., Wang X. L., Tang J. F., Zhu H. L., Bioorg. Med. Chem., 2011, 19(21), 6518-6524 |
| [4] | Shingalapur R. V., Hosamani K. M., Keri R. S., Hugar M. H., Eur. J. Med. Chem.,2010, 45(5), 1753-1759 |
| [5] | Wang F. H., Qin Z. L., Huang Q., Front. Chem. China,2006, 1(1), 112-114 |
| [6] | Naresh K. R., Poornachandra Y., Nagender P., Santhosh K. G., Krishna S. D., Ganesh K. C., Narsaiah B., Bioorg. Med. Chem. Lett., 2016, 26, 4829-4831 |
| [7] | Almasirad A., Mousavi Z., Tajik M., Assarzadeh M. J., Shafiee A., DARU-J. Pharm. Sci., 2014, 22(22), 1-8 |
| [8] | Chaudhary S. K., Chaudhary M., Chaudhari A., Parmar S. S., J. Pharm. Sci., 1978, 67(11), 1507-1509 |
| [9] | El-Emam A. A., Al-Deeb O. A., Al-Omar M., Lehmann J., Bioorg. Med. Chem., 2004, 12(19), 5107-5113 |
| [10] | Dolman S. J., Gosselin F., O’Shea P. D., Davies I. W., J. Org. Chem., 2006, 71(25), 9548-9551 |
| [11] | Biftu T., Feng D. D., Liang G. B., Kuo H., Qian X., Naylor E. M., Colandrea V. J., Candelore M. R., Cascieri M. A., Colwell L. F., Forrest M. J., Hom G. J., Maclntyre D. E., Stearns R. A., Strader C. D., Wyvratt M. J., Fisher M. H., Weber A.E., Bioorg. Med. Chem. Lett., 2000, 10(13), 1431-1434 |
| [12] | Velázquez C., Rao P. N. P., McDonald R., Knaus E. E., Bioorg. Med. Chem., 2005, 13(8), 2749-2757 |
| [13] | Era M., Adachi C., Tsutsui T., Saito S., Chem. Phys. Lett., 1991, 178(56), 488-490 |
| [14] | Bentiss F., Traisnel M., Lagrenee M., Corros. Sci., 2000, 42(1), 127-146 |
| [15] | Yang X. H., Wen Q., Zhao T. T., Sun J., Li X., Xing M., Lu X., Zhu H. L., Bioorg. Med. Chem.,2012, 20, 1181-1187 |
| [16] | Liu T. T., Wan Y. C., Liu R. S., Ma L., Li M. Y., Fang H., Chem. Res. Chinese Universities,2016, 32(5), 768-774 |
| [17] | Wang C. X., Song H. L., Liu W. Q., Xu C. L., Chem. Res. Chinese Universities,2016, 32(4), 615-620 |
| [18] | Gür M., Muglu H., Çavus M. S., Güder A., Sayiner H. S., Kandemirli F., J. Mole. Struct.,2017, 1134, 40-50 |
| [19] | Tang J. Y., Liu J. B., Wu F. Y., Bioorg. Chem., 2016, 69, 29-36 |
| [20] | Solankee A., Kapadia K., C’iric A., Sokovic M., Doytchinova I., Geronikaki A., Eur. J. Med. Chem., 2010, 45(2), 510-518 |
| [21] | Saczewski F., Bułakowska A., Bednarski P.,Grunert R., Eur. J. Med. Chem., 2006, 41(2), 219-225 |
| [22] | Zacharie B., Abbott S. D., Bienvenu J. F., Cameron A. D., Cloutier J., Duceppe J. S., Ezzitouni A., Fortin D., Houde K., Lauzon C., Moreau N., Perron V., Wilb N., Asselin M., Doueet A., Fafard M. E., Gaudreau D., Grouix B., Sarra B. F., St A. N., Gagnon L., Penney C. L., J. Med. Chem., 2010, 53(3), 1138-1145 |
| [23] | Singla P., Luxami V., Puai K., Eur. J. Med. Chem., 2016, 117, 59-69 |
| [24] | Mishra A., Kaushik N. K., Verma A. K., Gupta R., Eur. J. Med. Chem.,2008, 43(10), 2189-2196 |
| [25] | Guo Y., Synthesis and Activity of Novel Bicyclic Heterocycle Modified Amide Sulfide/Mannich Base Derivatives, Liaoning Normal University, Dalian, 2016 |
| (国阳. 新型双杂环修饰酰胺硫醚/曼尼希碱衍生物的合成及活性研究, 大连: 辽宁师范大学, 2016) | |
| [26] | Rakse M., Karthikeyan C., Deora G. S., Moorthy N. S. H. N., Rathore V., Rawat A. K., SrivastavaA. K., Trivedi P., Eur. J. Med. Chem., 2013, 70, 469-476 |
| [27] | Zhang J., Ji F. J., Gu Y., Zhang X. Y., Qiao S. X., Pharmacol. Rep., 2014, 66(3), 515-519 |
| [28] | Hu M., Design and Synthesis of Potential PTP1B/SHP2/Cdc25B Carbohydrate Small Molecule Inhibitor Based on CuAAC, East China University of Science and Technology, Shanghai, 2011 |
| (胡敏. 基于CuAAC的潜在PTP1B/SHP2/Cdc25B糖类小分子抑制剂的设计与合成, 上海: 华东理工大学, 2011) | |
| [29] | Cao X. R., Yang X. Y., Wang P. X., Liang Y., Liu F., Tuerhong M., Jin D. Q., Xu J., Lee D. H., Ohizumi Y., Guo Y. Q., Bioorg. Chem.,2017, 75, 139-148 |
| [30] | Matsuno T., Kato M., Tsuchida Y., Takahashi M., Yaguchi S., Terada S., Chem. Pharm. Bull., 1997, 45(2), 291-296 |
| [31] | Zhu C.A., Synthesis and Properties of Novel Y-Type 1,2,4-Triazole/Triazolothiadiazole-1,3,5-Triazine Derivatives, Liaoning Normal University, Dalian, 2015 |
| (朱长安. 新Y型1,2,4-三唑/三唑并噻二唑-1,3,5-三嗪衍生物的合成及其性能研究, 大连: 辽宁师范大学, 2015) | |
| [32] | Zhang C. L., Li C. Y., Yang M., Zhu C. A., Sun X. N., Li Y. Z., Chinese J. Appl. Chem.,2017, 34(4), 385-393 |
| (张成路, 李传银, 杨蒙, 朱长安, 孙晓娜, 李益政. 应用化学, 2017, 34(4), 385-393) | |
| [33] | Li D. J., Long D. Q., Fu H. Q., Chem. Inform., 2006, 181(3), 519-526 |
| [34] | Karaman N., Sıcak Y., Tok T. T., Öztürk M., Karaküçük I. A., Dikmen M., Koçyiği K. B., Oruç-EmreE. E., Eur. J. Med. Chem., 2016, 124, 270-283 |
| [35] | Ábrahámi R. A., Kiss L., Barrio P., Fülöp F., Tetrahedron,2016, 72(47), 7526-7535 |
| [1] | 王琳硕, 李昆杰, 刘玉敏, 赵瑞红, 李青, 钱鑫, 张帆, 薛志伟. 三苯基均三嗪基团调控敏化染料光电性能的理论研究[J]. 高等学校化学学报, 2020, 41(7): 1653. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||