

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (2): 262.doi: 10.7503/cjcu20180525
• Organic Chemistry • Previous Articles Next Articles
ZHANG Chenglu*( ), GONG Rongqing, YANG Jingyi, SUN Xiaona, LI Yilin, WANG Huayu, SONG Fulu, SUN Yuedong
), GONG Rongqing, YANG Jingyi, SUN Xiaona, LI Yilin, WANG Huayu, SONG Fulu, SUN Yuedong
Received:2018-07-24
Online:2019-02-10
Published:2018-12-27
Contact:
ZHANG Chenglu
E-mail:zhangchenglu@lnnu.edu.cn
Supported by:CLC Number:
TrendMD:
ZHANG Chenglu,GONG Rongqing,YANG Jingyi,SUN Xiaona,LI Yilin,WANG Huayu,SONG Fulu,SUN Yuedong. Synthesis of Novel Polyheterocyclic Molecules with 1,3,4-Oxadiazole Thioetheramide as Core Framework and Their Inhibitory Activity on Cdc25B and PTP1B†[J]. Chem. J. Chinese Universities, 2019, 40(2): 262.
| Compd. | m. p./℃ | Compd. | m. p./℃ |
|---|---|---|---|
| 1a | 173.1—173.9(173—174[ | 3c | 241.5—243.1(242—243[ |
| 1b | 113.7—114.8(114—115[ | 6a | 227.7—228.8(228—229[ |
| 1c | 114.1—114.9(114—115[ | 6b | 239.9—240.6(240—241[ |
| 2a | 289.1—289.7(289—290[ | 6c | 231.1—231.7(230—232[ |
| 2b | >300(>300[ | 6d | 243.6—244.2(243—244[ |
| 2c | 287.8—289.1(288—290[ | 6e | 162.5—163.1(162—163[ |
| 3a | 139.6—140.9(139—141[ | 6f | 169.6—170.9(170—171[ |
Table 1 Melting points of compounds 1a—1c, 2a—2c, 3a, 3c and 6a—6f
| Compd. | m. p./℃ | Compd. | m. p./℃ |
|---|---|---|---|
| 1a | 173.1—173.9(173—174[ | 3c | 241.5—243.1(242—243[ |
| 1b | 113.7—114.8(114—115[ | 6a | 227.7—228.8(228—229[ |
| 1c | 114.1—114.9(114—115[ | 6b | 239.9—240.6(240—241[ |
| 2a | 289.1—289.7(289—290[ | 6c | 231.1—231.7(230—232[ |
| 2b | >300(>300[ | 6d | 243.6—244.2(243—244[ |
| 2c | 287.8—289.1(288—290[ | 6e | 162.5—163.1(162—163[ |
| 3a | 139.6—140.9(139—141[ | 6f | 169.6—170.9(170—171[ |
| Compd. | Appearance | Yield(%) | m. p./℃ | IR(KBr), |
|---|---|---|---|---|
| 3b | White solid | 85.3 | 257.1—257.9 | 3419, 3033, 2960, 1694, 1630, 1578, 1519, 1453, 1276, 1172, 864 |
| 4a | White solid | 82.3 | 229.5—230.3 | 3412, 3025, 2979, 1715, 1618, 1499, 1401, 1250, 1172, 857 |
| 4b | White solid | 91.8 | 280.7—281.3 | 3416, 3036, 2971, 1702, 1616, 1566, 1513, 1428, 1172, 866 |
| 4c | White solid | 89.2 | 164.9—165.7 | 3326, 3051, 2967, 1708, 1611, 1572, 1506, 1394, 1173, 858 |
| 5a | Light yellow solid | 62.1 | 276.8—277.5 | 3418, 3025, 2934, 1611, 1493, 1401, 1362, 1238, 1165, 845 |
| 5b | Light yellow solid | 69.5 | >300 | 3431, 3051, 2966, 1604, 1499, 1388, 1335, 1244, 1159, 844 |
| 5c | Light yellow solid | 56.2 | 281.7—282.6 | 3431, 3084, 2934, 1605, 1499, 1414, 1362, 1250, 1165, 845 |
Table 2 Appearance, yields, melting points and IR data of compounds 3b, 4a—4c and 5a—5c
| Compd. | Appearance | Yield(%) | m. p./℃ | IR(KBr), |
|---|---|---|---|---|
| 3b | White solid | 85.3 | 257.1—257.9 | 3419, 3033, 2960, 1694, 1630, 1578, 1519, 1453, 1276, 1172, 864 |
| 4a | White solid | 82.3 | 229.5—230.3 | 3412, 3025, 2979, 1715, 1618, 1499, 1401, 1250, 1172, 857 |
| 4b | White solid | 91.8 | 280.7—281.3 | 3416, 3036, 2971, 1702, 1616, 1566, 1513, 1428, 1172, 866 |
| 4c | White solid | 89.2 | 164.9—165.7 | 3326, 3051, 2967, 1708, 1611, 1572, 1506, 1394, 1173, 858 |
| 5a | Light yellow solid | 62.1 | 276.8—277.5 | 3418, 3025, 2934, 1611, 1493, 1401, 1362, 1238, 1165, 845 |
| 5b | Light yellow solid | 69.5 | >300 | 3431, 3051, 2966, 1604, 1499, 1388, 1335, 1244, 1159, 844 |
| 5c | Light yellow solid | 56.2 | 281.7—282.6 | 3431, 3084, 2934, 1605, 1499, 1414, 1362, 1250, 1165, 845 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ |
|---|---|
| 3b | 9.46(s, 1H, NH), 7.98(d,J=8.4 Hz, 2H, PhH), 7.87(d, J=8.5 Hz, 2H, PhH), 3.82(s, 3H, OCH3), 3.52—3.49(m, 8H, pyrrolidine), 1.91(s, 8H, pyrrolidine) |
| 4a | 9.53(s, 1H, NH), 9.34(s, 1H, NH), 7.74(s, 4H, PhH), 4.42(s, 2H, NH2), 3.71(s, 8H, morpholine), 3.63(s, 8H, morpholine) |
| 4b | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.91(d,J=8.3 Hz, 2H, PhH), 7.74(d, J=8.3 Hz, 2H, PhH), 4.36(s, 2H, NH2), 3.50—3.45(m, 8H, pyrrolidine), 1.90(s, 8H, pyrrolidine) |
| 4c | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.74(q,J=8.8 Hz, 4H, PhH), 4.42(s, 2H, NH2), 3.71(d, J=4.35 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) |
| 5a | 14.49(s, 1H, SH), 9.59(s, 1H, NH), 7.93(d,J=8.6 Hz, 2H, PhH), 7.81(d, J=8.7 Hz, 2H, PhH), 3.74(s, 8H, morpholine), 3.65(s, 8H, morpholine) |
| 5b | 13.38(s, 1H, SH), 9.45(s, 1H, NH), 8.05(d,J=8.7 Hz, 2H, PhH), 7.98(d, J=8.55 Hz, 2H, PhH), 3.51—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) |
| 5c | 13.84(s, 1H, SH), 9.43(s, 1H, NH), 7.92(d,J=8.75 Hz, 2H, PhH), 7.78(d, J=8.7 Hz, 2H, PhH), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) |
Table 3 1H NMR data of compounds 3b, 4a—4c and 5a—5c
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ |
|---|---|
| 3b | 9.46(s, 1H, NH), 7.98(d,J=8.4 Hz, 2H, PhH), 7.87(d, J=8.5 Hz, 2H, PhH), 3.82(s, 3H, OCH3), 3.52—3.49(m, 8H, pyrrolidine), 1.91(s, 8H, pyrrolidine) |
| 4a | 9.53(s, 1H, NH), 9.34(s, 1H, NH), 7.74(s, 4H, PhH), 4.42(s, 2H, NH2), 3.71(s, 8H, morpholine), 3.63(s, 8H, morpholine) |
| 4b | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.91(d,J=8.3 Hz, 2H, PhH), 7.74(d, J=8.3 Hz, 2H, PhH), 4.36(s, 2H, NH2), 3.50—3.45(m, 8H, pyrrolidine), 1.90(s, 8H, pyrrolidine) |
| 4c | 9.52(s, 1H, NH), 9.19(s, 1H, NH), 7.74(q,J=8.8 Hz, 4H, PhH), 4.42(s, 2H, NH2), 3.71(d, J=4.35 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) |
| 5a | 14.49(s, 1H, SH), 9.59(s, 1H, NH), 7.93(d,J=8.6 Hz, 2H, PhH), 7.81(d, J=8.7 Hz, 2H, PhH), 3.74(s, 8H, morpholine), 3.65(s, 8H, morpholine) |
| 5b | 13.38(s, 1H, SH), 9.45(s, 1H, NH), 8.05(d,J=8.7 Hz, 2H, PhH), 7.98(d, J=8.55 Hz, 2H, PhH), 3.51—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) |
| 5c | 13.84(s, 1H, SH), 9.43(s, 1H, NH), 7.92(d,J=8.75 Hz, 2H, PhH), 7.78(d, J=8.7 Hz, 2H, PhH), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) |
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(positive-SIMS), m/z(calcd. ), [M+H]+ |
|---|---|---|---|---|
| TZOA-1 | White solid | 67.6 | 279.8—280.8 | 660.1935(660.1923) |
| TZOA-2 | White solid | 68.9 | 282.9—283.7 | 674.2088(674.2080) |
| TZOA-3 | Light pink solid | 73.9 | 273.1—273.9 | 690.2025(690.2029) |
| TZOA-4 | Brownish solid | 65.1 | 259.7—260.5 | 694.1542(694.1534) |
| TZOA-5 | Yellow solid | 73.2 | 287.6—288.5 | 705.1782(705.1774) |
| TZOA-6 | White solid | 76.1 | 253.2—253.8 | 728.1787(728.1797) |
| TZOA-7 | Yellow solid | 75.8 | 250.7—251.5 | 628.2014(628.2025) |
| TZOA-8 | White solid | 66.9 | 269.8—270.3 | 642.2191(642.2182) |
| TZOA-9 | White solid | 73.9 | 255.5—256.3 | 658.2138(658.2131) |
| TZOA-10 | White solid | 79.9 | 265.1—265.7 | 662.1640(662.1635) |
| TZOA-11 | Yellow solid | 77.5 | 262.6—263.2 | 673.1883(673.1876) |
| TZOA-12 | Yellow solid | 79.1 | 284.2—285.0 | 696.1907(696.1899) |
| TZOA-13 | Yellow solid | 78.2 | 252.7—253.5 | 656.2331(656.2338) |
| TZOA-14 | White solid | 76.8 | 248.9—249.6 | 670.2489(670.2495) |
| TZOA-15 | White solid | 78.2 | 271.7—272.3 | 686.2453(686.2444) |
| TZOA-16 | White solid | 73.1 | 254.3—254.9 | 690.1952(690.1948) |
| TZOA-17 | Yellow solid | 77.3 | >300 | 701.2195(701.2189) |
| TZOA-18 | White solid | 79.9 | >300 | 724.2219(724.2212) |
Table 4 Appearance, yields, melting points and HRMS data of compounds TZOA-1—TZOA-18
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(positive-SIMS), m/z(calcd. ), [M+H]+ |
|---|---|---|---|---|
| TZOA-1 | White solid | 67.6 | 279.8—280.8 | 660.1935(660.1923) |
| TZOA-2 | White solid | 68.9 | 282.9—283.7 | 674.2088(674.2080) |
| TZOA-3 | Light pink solid | 73.9 | 273.1—273.9 | 690.2025(690.2029) |
| TZOA-4 | Brownish solid | 65.1 | 259.7—260.5 | 694.1542(694.1534) |
| TZOA-5 | Yellow solid | 73.2 | 287.6—288.5 | 705.1782(705.1774) |
| TZOA-6 | White solid | 76.1 | 253.2—253.8 | 728.1787(728.1797) |
| TZOA-7 | Yellow solid | 75.8 | 250.7—251.5 | 628.2014(628.2025) |
| TZOA-8 | White solid | 66.9 | 269.8—270.3 | 642.2191(642.2182) |
| TZOA-9 | White solid | 73.9 | 255.5—256.3 | 658.2138(658.2131) |
| TZOA-10 | White solid | 79.9 | 265.1—265.7 | 662.1640(662.1635) |
| TZOA-11 | Yellow solid | 77.5 | 262.6—263.2 | 673.1883(673.1876) |
| TZOA-12 | Yellow solid | 79.1 | 284.2—285.0 | 696.1907(696.1899) |
| TZOA-13 | Yellow solid | 78.2 | 252.7—253.5 | 656.2331(656.2338) |
| TZOA-14 | White solid | 76.8 | 248.9—249.6 | 670.2489(670.2495) |
| TZOA-15 | White solid | 78.2 | 271.7—272.3 | 686.2453(686.2444) |
| TZOA-16 | White solid | 73.1 | 254.3—254.9 | 690.1952(690.1948) |
| TZOA-17 | Yellow solid | 77.3 | >300 | 701.2195(701.2189) |
| TZOA-18 | White solid | 79.9 | >300 | 724.2219(724.2212) |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
|---|---|---|
| TZOA-1 | 13.17(s, 1H, NH), 9.54(s, 1H, NH), 7.91—7.85(m, 6H, PhH), 7.50—7.42(m, 3H, PhH), 4.32(s, 2H, CH2), 3.72(d, J=4.15 Hz, 8H, morpholine), 3.64(d, J=4.05 Hz, 8H, morpholine) | 168.13, 165.06, 164.64, 163.94, 162.89, 159.77, 143.78, 131.51, 129.54, 129.06, 126.96, 126.44, 119.33, 115.44, 65.96, 43.38, 37.71 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-2 | 9.55(s, 1H, NH), 7.91(q,J=8.6 Hz, 4H, PhH), 7.73(d, J=7.65 Hz, 2H, PhH), 7.27(d, J=7.8 Hz, 2H, Ph), 4.18(s, 2H, CH2), 3.74(s, 8H, morpholine), 3.66(s, 8H, morpholine), 2.35(s, 3H, CH3) | 169.90, 168.85, 164.71, 164.66, 163.96, 157.59, 143.64, 138.00, 130.27, 129.38, 126.91, 125.93, 119.36, 115.66, 65.97, 43.38, 20.80 |
| TZOA-3 | 13.01(s, 1H, NH), 9.54(s, 1H, NH), 7.91(d,J=8.7 Hz, 2H, PhH), 7.86(d, J=8.6 Hz, 2H, PhH), 7.81(d, J=8.5 Hz, 2H, PhH), 7.04(d, J=8.5 Hz, 2H, Ph), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.72(d, J=3.6 Hz, 8H, morpholine), 3.64(d, J=3.55 Hz, 8H, morpholine) | 168.53, 164.93, 164.63, 163.93, 163.25, 160.10, 158.93, 143.73, 127.73, 126.94, 124.62, 119.32, 115.50, 114.42, 65.96, 55.23, 43.36, 38.47 |
| TZOA-4 | 13.16(s, 1H, NH), 9.55(s, 1H, NH), 7.92(t,J=8.4 Hz, 4H, PhH), 7.87(d, J=8.75 Hz, 2H, PhH), 7.58(d, J=8.05 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.73(d, J=3.3 Hz, 8H, morpholine), 3.65(s, 8H, morpholine) | 167.96, 165.09, 164.63, 163.94, 163.27, 162.79, 159.99, 143.80, 131.36, 129.66, 129.10, 126.97, 126.48, 119.32, 115.41, 65.96, 43.37, 37.49 |
| TZOA-5 | 9.53(s, 1H, NH), 8.29(d,J=8.5 Hz, 2H, PhH), 8.09(d, J=8.5 Hz, 2H, PhH), 7.88(q, J=8.75 Hz, 4H, PhH), 4.22(s, 2H, CH2), 3.72(d, J=4.05 Hz, 8H, morpholine), 3.64(d, J=3.9 Hz, 8H, morpholine) | 167.75, 165.12, 164.63, 163.93, 162.68, 160.23, 143.81, 131.20, 129.76, 129.11, 126.96, 126.53, 119.31, 115.38, 65.96, 43.36, 37.27 |
| TZOA-6 | 13.32(s, 1H, NH), 9.54(s, 1H, NH), 8.14(d,J=8.2 Hz, 2H, PhH), 7.90(d, J=8.75 Hz, 2H, PhH), 7.85(t, J=8.25 Hz, 4H, PhH), 4.43(s, 2H, CH2), 3.71(s, 8H, morpholine), 3.64(d, J=3.5 Hz, 8H, morpholine) | 167.59, 165.23, 164.62, 163.92, 162.36, 162.22, 159.26, 143.85, 134.62, 129.86, 129.61, 127.29, 126.97, 126.09, 126.07, 125.02, 122.85, 119.30, 115.32, 65.95, 43.36, 36.68 |
| TZOA-7 | 13.18(s, 1H, NH), 9.38(s, 1H, NH), 8.05(d,J=8.65 Hz, 2H, PhH), 7.89(d, J=7.7 Hz, 2H, PhH), 7.83(d, J =8.65 Hz, 2H, PhH), 7.50—7.43(m, 3H, PhH), 4.33(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.16, 164.75, 164.41, 163.04, 162.60, 159.01, 144.12, 131.44, 128.99, 128.67, 126.45, 126.01, 118.65, 114.55, 45.31, 37.79, 24.46, 24.29 |
| TZOA-8 | 9.37(s, 1H, NH), 8.06(d,J=8.4 Hz, 2H, PhH), 7.84(d, J=8.35 Hz, 2H, PhH), 7.72(d, J=7.6 Hz, 2H, PhH), 7.26(d, J=7.8 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 2.33(s, 3H, CH3), 1.89(s, 8H, pyrrolidine) | 169.78, 164.83, 163.75, 163.39, 162.95, 157.75, 144.36, 138.10, 130.15, 129.40, 126.77, 125.97, 119.00, 115.05, 45.66, 24.82, 24.65, 20.82 |
| TZOA-9 | 13.00(s, 1H, NH), 9.37(s, 1H, NH), 8.05(d,J=8.9 Hz, 2H, PhH), 7.83—7.79(m, 4H, PhH), 7.04(d, J=8.9 Hz, 2H, PhH), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.64, 165.01, 163.38, 163.17, 162.94, 160.06, 158.79, 144.43, 127.71, 126.79, 124.68, 118.99, 114.93, 114.41, 55.23, 45.64, 38.52, 24.81, 24.64 |
| TZOA-10 | 9.37(s, 1H, NH), 8.06(d,J=8.3 Hz, 2H, PhH), 7.84(d, J=8.25 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.17(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 169.55, 168.12, 164.53, 163.22, 163.05, 162.62, 144.02, 132.73, 131.22, 128.59, 127.27, 126.44, 118.66, 114.69, 45.33, 24.47, 24.31 |
| TZOA-11 | 9.36(s, 1H, NH), 8.31(d,J=8.65 Hz, 2H, PhH), 8.14(d, J=8.65 Hz, 2H, PhH), 8.04(d, J=8.65 Hz, 2H, PhH), 7.82(t, J=8.45 Hz, 2H, PhH), 4.31(s, 2H, CH2), 3.50—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 169.89, 168.97, 164.47, 163.39, 163.04, 162.60, 155.91, 144.00, 132.55, 131.44, 128.55, 127.19, 126.43, 118.66, 114.71, 45.29, 24.48, 24.30 |
| TZOA-12 | 13.23(s, 1H, NH), 9.38(s, 1H, NH), 8.12(d, J=8.25 Hz, 2H, PhH), 8.05(d, J=8.6 Hz, 2H, PhH), 7.85—7.81(m, 4H, PhH), 4.36(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 167.99, 164.86, 163.04, 162.60, 162.23, 158.04, 144.15, 134.81, 129.16, 128.90, 126.72, 126.47, 125.67, 125.64, 124.73, 122.57, 118.64, 114.49, 45.30, 37.14, 24.46, 24.29 |
| TZOA-13 | 9.38(s, 1H, NH), 7.93(d, J=8.15 Hz, 2H, PhH), 7.87(d, J=8.55 Hz, 2H, PhH), 7.83(d, J=7.6 Hz, 2H, PhH), 7.44(t,J=7.65 Hz, 2H, PhH), 7.38—7.35(m, 1H, PhH), 4.17(s, 2H, CH2), 3.73(t, J=4.75 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.79, 165.18, 164.35, 164.05, 162.62, 160.20, 144.18, 131.24, 129.73, 129.10, 126.91, 126.52, 119.04, 115.08, 43.65, 37.31, 25.33, 24.31 |
| TZOA-14 | 9.38(s, 1H, NH), 7.92(d,J=7.55 Hz, 2H, PhH), 7.86(d, J=7.9 Hz, 2H, PhH), 7.71(d, J=6.95 Hz, 2H, PhH), 7.25(d, J=6.85 Hz, 2H, PhH), 4.16(s, 2H, CH2), 3.72(s, 8H, piperidine), 2.33(s, 3H, CH3), 1.62—1.51(m, 12H, piperidine) | 169.90, 168.84, 164.75, 164.37, 164.07, 163.90, 157.58, 144.01, 137.98, 130.28, 129.38, 126.87, 125.93, 119.07, 115.34, 43.67, 25.33, 24.32, 20.80 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-15 | 13.05(s, 1H, NH), 9.39(s, 1H, NH), 7.91(d,J=8.55 Hz, 2H, PhH), 7.83(q, J=5.45 Hz, 4H, PhH), 7.06(d, J=8.55 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.82(s, 3H, OCH3), 3.71(t, J=4.5 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) | 166.84, 164.89, 163.99, 163.69, 162.05, 160.30, 143.86, 127.79, 126.58, 123.15, 120.64, 118.68, 114.67, 114.24, 54.96, 43.30, 36.43, 24.98, 23.96 |
| TZOA-16 | 9.38(s, 1H, NH), 7.92(d,J=8.7 Hz, 2H, PhH), 7.86(t,J=8.55 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) | 170.27, 169.41, 164.78, 164.37, 164.06,163.81, 156.23, 144.02, 132.89, 131.82, 128.89, 127.53, 126.86, 119.07, 115.32, 43.66, 25.33, 24.32 |
| TZOA-17 | 9.38(s, 1H, NH), 8.29(d,J=8.85 Hz, 2H, PhH), 8.09(d, J=8.25 Hz, 2H, PhH), 7.92(d, J=8.25 Hz, 2H, PhH), 7.86(d, J=8.55 Hz, 2H, PhH), 4.20(s, 2H, CH2), 3.73(t, J=4.8 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.30, 165.29, 164.38, 164.33, 164.07, 162.26, 159.71, 144.22, 143.58, 134.59, 129.67, 129.23, 128.30, 126.92, 126.03, 119.02, 118.00, 114.99, 43.62, 36.59, 25.33, 24.32 |
| TZOA-18 | 13.30(s, 1H, NH), 9.39(s, 1H, NH), 8.15(d,J=8.1 Hz, 2H, PhH), 7.91(d, J=8.4 Hz, 2H, PhH), 7.87(d, J=8.1 Hz, 2H, PhH), 7.83(d, J=8.6 Hz, 2H, PhH), 4.43(s, 2H, CH2), 3.71(t, J=4.65 Hz, 8H, piperidine), 1.61—1.50(m, 12H, piperidine) | 167.18, 164.96, 164.00, 163.70, 161.89, 159.00, 143.88, 134.23, 129.57, 129.31, 126.98, 126.58, 125.73, 122.51, 118.69, 114.66, 43.31, 36.24, 24.98, 23.96 |
Table 5 1H NMR and 13C NMR data of compounds TZOA-1—TZOA-18
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
|---|---|---|
| TZOA-1 | 13.17(s, 1H, NH), 9.54(s, 1H, NH), 7.91—7.85(m, 6H, PhH), 7.50—7.42(m, 3H, PhH), 4.32(s, 2H, CH2), 3.72(d, J=4.15 Hz, 8H, morpholine), 3.64(d, J=4.05 Hz, 8H, morpholine) | 168.13, 165.06, 164.64, 163.94, 162.89, 159.77, 143.78, 131.51, 129.54, 129.06, 126.96, 126.44, 119.33, 115.44, 65.96, 43.38, 37.71 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-2 | 9.55(s, 1H, NH), 7.91(q,J=8.6 Hz, 4H, PhH), 7.73(d, J=7.65 Hz, 2H, PhH), 7.27(d, J=7.8 Hz, 2H, Ph), 4.18(s, 2H, CH2), 3.74(s, 8H, morpholine), 3.66(s, 8H, morpholine), 2.35(s, 3H, CH3) | 169.90, 168.85, 164.71, 164.66, 163.96, 157.59, 143.64, 138.00, 130.27, 129.38, 126.91, 125.93, 119.36, 115.66, 65.97, 43.38, 20.80 |
| TZOA-3 | 13.01(s, 1H, NH), 9.54(s, 1H, NH), 7.91(d,J=8.7 Hz, 2H, PhH), 7.86(d, J=8.6 Hz, 2H, PhH), 7.81(d, J=8.5 Hz, 2H, PhH), 7.04(d, J=8.5 Hz, 2H, Ph), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.72(d, J=3.6 Hz, 8H, morpholine), 3.64(d, J=3.55 Hz, 8H, morpholine) | 168.53, 164.93, 164.63, 163.93, 163.25, 160.10, 158.93, 143.73, 127.73, 126.94, 124.62, 119.32, 115.50, 114.42, 65.96, 55.23, 43.36, 38.47 |
| TZOA-4 | 13.16(s, 1H, NH), 9.55(s, 1H, NH), 7.92(t,J=8.4 Hz, 4H, PhH), 7.87(d, J=8.75 Hz, 2H, PhH), 7.58(d, J=8.05 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.73(d, J=3.3 Hz, 8H, morpholine), 3.65(s, 8H, morpholine) | 167.96, 165.09, 164.63, 163.94, 163.27, 162.79, 159.99, 143.80, 131.36, 129.66, 129.10, 126.97, 126.48, 119.32, 115.41, 65.96, 43.37, 37.49 |
| TZOA-5 | 9.53(s, 1H, NH), 8.29(d,J=8.5 Hz, 2H, PhH), 8.09(d, J=8.5 Hz, 2H, PhH), 7.88(q, J=8.75 Hz, 4H, PhH), 4.22(s, 2H, CH2), 3.72(d, J=4.05 Hz, 8H, morpholine), 3.64(d, J=3.9 Hz, 8H, morpholine) | 167.75, 165.12, 164.63, 163.93, 162.68, 160.23, 143.81, 131.20, 129.76, 129.11, 126.96, 126.53, 119.31, 115.38, 65.96, 43.36, 37.27 |
| TZOA-6 | 13.32(s, 1H, NH), 9.54(s, 1H, NH), 8.14(d,J=8.2 Hz, 2H, PhH), 7.90(d, J=8.75 Hz, 2H, PhH), 7.85(t, J=8.25 Hz, 4H, PhH), 4.43(s, 2H, CH2), 3.71(s, 8H, morpholine), 3.64(d, J=3.5 Hz, 8H, morpholine) | 167.59, 165.23, 164.62, 163.92, 162.36, 162.22, 159.26, 143.85, 134.62, 129.86, 129.61, 127.29, 126.97, 126.09, 126.07, 125.02, 122.85, 119.30, 115.32, 65.95, 43.36, 36.68 |
| TZOA-7 | 13.18(s, 1H, NH), 9.38(s, 1H, NH), 8.05(d,J=8.65 Hz, 2H, PhH), 7.89(d, J=7.7 Hz, 2H, PhH), 7.83(d, J =8.65 Hz, 2H, PhH), 7.50—7.43(m, 3H, PhH), 4.33(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.16, 164.75, 164.41, 163.04, 162.60, 159.01, 144.12, 131.44, 128.99, 128.67, 126.45, 126.01, 118.65, 114.55, 45.31, 37.79, 24.46, 24.29 |
| TZOA-8 | 9.37(s, 1H, NH), 8.06(d,J=8.4 Hz, 2H, PhH), 7.84(d, J=8.35 Hz, 2H, PhH), 7.72(d, J=7.6 Hz, 2H, PhH), 7.26(d, J=7.8 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 2.33(s, 3H, CH3), 1.89(s, 8H, pyrrolidine) | 169.78, 164.83, 163.75, 163.39, 162.95, 157.75, 144.36, 138.10, 130.15, 129.40, 126.77, 125.97, 119.00, 115.05, 45.66, 24.82, 24.65, 20.82 |
| TZOA-9 | 13.00(s, 1H, NH), 9.37(s, 1H, NH), 8.05(d,J=8.9 Hz, 2H, PhH), 7.83—7.79(m, 4H, PhH), 7.04(d, J=8.9 Hz, 2H, PhH), 4.28(s, 2H, CH2), 3.81(s, 3H, OCH3), 3.51—3.47(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 168.64, 165.01, 163.38, 163.17, 162.94, 160.06, 158.79, 144.43, 127.71, 126.79, 124.68, 118.99, 114.93, 114.41, 55.23, 45.64, 38.52, 24.81, 24.64 |
| TZOA-10 | 9.37(s, 1H, NH), 8.06(d,J=8.3 Hz, 2H, PhH), 7.84(d, J=8.25 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.17(s, 2H, CH2), 3.52—3.48(m, 8H, pyrrolidine), 1.89(s, 8H, pyrrolidine) | 169.55, 168.12, 164.53, 163.22, 163.05, 162.62, 144.02, 132.73, 131.22, 128.59, 127.27, 126.44, 118.66, 114.69, 45.33, 24.47, 24.31 |
| TZOA-11 | 9.36(s, 1H, NH), 8.31(d,J=8.65 Hz, 2H, PhH), 8.14(d, J=8.65 Hz, 2H, PhH), 8.04(d, J=8.65 Hz, 2H, PhH), 7.82(t, J=8.45 Hz, 2H, PhH), 4.31(s, 2H, CH2), 3.50—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 169.89, 168.97, 164.47, 163.39, 163.04, 162.60, 155.91, 144.00, 132.55, 131.44, 128.55, 127.19, 126.43, 118.66, 114.71, 45.29, 24.48, 24.30 |
| TZOA-12 | 13.23(s, 1H, NH), 9.38(s, 1H, NH), 8.12(d, J=8.25 Hz, 2H, PhH), 8.05(d, J=8.6 Hz, 2H, PhH), 7.85—7.81(m, 4H, PhH), 4.36(s, 2H, CH2), 3.51—3.47(m, 8H, pyrrolidine), 1.88(s, 8H, pyrrolidine) | 167.99, 164.86, 163.04, 162.60, 162.23, 158.04, 144.15, 134.81, 129.16, 128.90, 126.72, 126.47, 125.67, 125.64, 124.73, 122.57, 118.64, 114.49, 45.30, 37.14, 24.46, 24.29 |
| TZOA-13 | 9.38(s, 1H, NH), 7.93(d, J=8.15 Hz, 2H, PhH), 7.87(d, J=8.55 Hz, 2H, PhH), 7.83(d, J=7.6 Hz, 2H, PhH), 7.44(t,J=7.65 Hz, 2H, PhH), 7.38—7.35(m, 1H, PhH), 4.17(s, 2H, CH2), 3.73(t, J=4.75 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.79, 165.18, 164.35, 164.05, 162.62, 160.20, 144.18, 131.24, 129.73, 129.10, 126.91, 126.52, 119.04, 115.08, 43.65, 37.31, 25.33, 24.31 |
| TZOA-14 | 9.38(s, 1H, NH), 7.92(d,J=7.55 Hz, 2H, PhH), 7.86(d, J=7.9 Hz, 2H, PhH), 7.71(d, J=6.95 Hz, 2H, PhH), 7.25(d, J=6.85 Hz, 2H, PhH), 4.16(s, 2H, CH2), 3.72(s, 8H, piperidine), 2.33(s, 3H, CH3), 1.62—1.51(m, 12H, piperidine) | 169.90, 168.84, 164.75, 164.37, 164.07, 163.90, 157.58, 144.01, 137.98, 130.28, 129.38, 126.87, 125.93, 119.07, 115.34, 43.67, 25.33, 24.32, 20.80 |
| Compd. | 1H NMR(500 MHz, DMSO-d6), δ | 13C NMR(125 MHz, DMSO-d6), δ |
| TZOA-15 | 13.05(s, 1H, NH), 9.39(s, 1H, NH), 7.91(d,J=8.55 Hz, 2H, PhH), 7.83(q, J=5.45 Hz, 4H, PhH), 7.06(d, J=8.55 Hz, 2H, PhH), 4.37(s, 2H, CH2), 3.82(s, 3H, OCH3), 3.71(t, J=4.5 Hz, 8H, piperidine), 1.62—1.50(m, 12H, piperidine) | 166.84, 164.89, 163.99, 163.69, 162.05, 160.30, 143.86, 127.79, 126.58, 123.15, 120.64, 118.68, 114.67, 114.24, 54.96, 43.30, 36.43, 24.98, 23.96 |
| TZOA-16 | 9.38(s, 1H, NH), 7.92(d,J=8.7 Hz, 2H, PhH), 7.86(t,J=8.55 Hz, 4H, PhH), 7.50(d, J=8.3 Hz, 2H, PhH), 4.18(s, 2H, CH2), 3.72(s, 8H, piperidine), 1.62—1.51(m, 12H, piperidine) | 170.27, 169.41, 164.78, 164.37, 164.06,163.81, 156.23, 144.02, 132.89, 131.82, 128.89, 127.53, 126.86, 119.07, 115.32, 43.66, 25.33, 24.32 |
| TZOA-17 | 9.38(s, 1H, NH), 8.29(d,J=8.85 Hz, 2H, PhH), 8.09(d, J=8.25 Hz, 2H, PhH), 7.92(d, J=8.25 Hz, 2H, PhH), 7.86(d, J=8.55 Hz, 2H, PhH), 4.20(s, 2H, CH2), 3.73(t, J=4.8 Hz, 8H, piperidine), 1.63—1.51(m, 12H, piperidine) | 167.30, 165.29, 164.38, 164.33, 164.07, 162.26, 159.71, 144.22, 143.58, 134.59, 129.67, 129.23, 128.30, 126.92, 126.03, 119.02, 118.00, 114.99, 43.62, 36.59, 25.33, 24.32 |
| TZOA-18 | 13.30(s, 1H, NH), 9.39(s, 1H, NH), 8.15(d,J=8.1 Hz, 2H, PhH), 7.91(d, J=8.4 Hz, 2H, PhH), 7.87(d, J=8.1 Hz, 2H, PhH), 7.83(d, J=8.6 Hz, 2H, PhH), 4.43(s, 2H, CH2), 3.71(t, J=4.65 Hz, 8H, piperidine), 1.61—1.50(m, 12H, piperidine) | 167.18, 164.96, 164.00, 163.70, 161.89, 159.00, 143.88, 134.23, 129.57, 129.31, 126.98, 126.58, 125.73, 122.51, 118.69, 114.66, 43.31, 36.24, 24.98, 23.96 |
| Compd. | Cdc25Ba | PTP1Bc | ||
|---|---|---|---|---|
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 1a | 1.55±1.34 | NAd | 2.99±3.88 | NA |
| 1b | 0.16±0.66 | NA | 1.01±0.05 | NA |
| 1c | 18.62±2.83 | NA | 24.47±1.22 | NA |
| 2a | 0.67±0.17 | NA | 4.77±6.25 | NA |
| 2b | 53.53±2.79 | 10.37±0.63 | 38.79±5.97 | NA |
| 2c | 37.76±7.95 | NA | 22.35±0.77 | NA |
| 3a | 4.85±0.23 | NA | 5.45±0.39 | NA |
| 3b | 53.48±1.06 | 5.62±1.52 | 84.63±4.76 | 9.18±0.43 |
| 3c | 0.32±1.52 | NA | 24.07±1.96 | NA |
| 4a | 1.64±0.69 | NA | 4.91±0.60 | NA |
| 4b | 8.93±3.84 | NA | 1.21±0.52 | NA |
| 4c | 1.41±3.43 | NA | 6.63±3.14 | NA |
| 5a | 61.81±3.58 | 12.82±1.46 | 31.13±4.69 | NA |
| 5b | 60.00±1.00 | 6.64±1.04 | 60.53±2.00 | 19.71±2.84 |
| 5c | 64.49±1.60 | 6.41±0.49 | 84.47±0.52 | 6.64±0.16 |
| 6a | 0.34±0.39 | NA | 15.64±5.71 | NA |
| 6b | 36.68±2.83 | NA | 85.71±2.80 | 12.24±0.11 |
| 6c | 0.02±0.35 | NA | 85.22±1.87 | 20.58±0.06 |
| Compd. | Cdc25Ba | PTP1Bc | ||
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 6d | 14.99±1.04 | NA | 91.00±1.53 | 7.21±0.11 |
| 6e | 64.59±0.69 | 9.92±0.74 | 87.74±2.21 | 17.28±2.24 |
| 6f | 25.69±2.93 | NA | 88.47±1.38 | 11.81±0.47 |
| TZOA-1 | 4.28±0.98 | NA | 31.91±0.81 | NA |
| TZOA-2 | 44.10±0.00 | NA | 31.77±0.32 | NA |
| TZOA-3 | 0.37±1.02 | NA | 31.85±1.09 | NA |
| TZOA-4 | 69.32±2.77 | 6.66±1.77 | 40.60±1.18 | NA |
| TZOA-5 | 70.23±3.06 | 3.32±0.06 | 44.83±4.20 | NA |
| TZOA-6 | 77.53±1.20 | 3.01±0.19 | 48.10±0.92 | NA |
| TZOA-7 | 74.97±0.17 | 3.36±0.13 | 84.42±1.00 | 7.04±0.73 |
| TZOA-8 | 24.30±4.25 | NA | 86.75±1.65 | 15.11±0.98 |
| TZOA-9 | 57.61±0.91 | 9.38±0.99 | 84.66±0.90 | 9.32±1.86 |
| TZOA-10 | 63.73±4.13 | 5.49±0.48 | 84.70±8.06 | 3.24±0.06 |
| TZOA-11 | 92.25±0.21 | 0.71±0.04 | 93.40±0.39 | 2.37±0.38 |
| TZOA-12 | 89.07±1.18 | 1.12±0.13 | 89.16±6.93 | 8.50±0.07 |
| TZOA-13 | 90.28±0.07 | 1.04±0.25 | 77.33±0.73 | 26.18±6.77 |
| TZOA-14 | 93.37±1.09 | 0.88±0.15 | 88.15±0.14 | 4.78±0.24 |
| TZOA-15 | 94.54±1.50 | 0.86±0.50 | 88.78±0.56 | 4.22±0.64 |
| TZOA-16 | 91.13±0.06 | 1.02±0.12 | 89.73±3.35 | 4.36±0.78 |
| TZOA-17 | 96.18±0.16 | 0.66±0.33 | 92.71±0.74 | 6.12±0.58 |
| TZOA-18 | 99.23±0.09 | 0.46±0.34 | 91.52±0.39 | 3.04±0.02 |
Table 6 Inhibitory activities of compounds 1—6 and TZOA-1—TZOA-18 against Cdc25B and PTP1B
| Compd. | Cdc25Ba | PTP1Bc | ||
|---|---|---|---|---|
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 1a | 1.55±1.34 | NAd | 2.99±3.88 | NA |
| 1b | 0.16±0.66 | NA | 1.01±0.05 | NA |
| 1c | 18.62±2.83 | NA | 24.47±1.22 | NA |
| 2a | 0.67±0.17 | NA | 4.77±6.25 | NA |
| 2b | 53.53±2.79 | 10.37±0.63 | 38.79±5.97 | NA |
| 2c | 37.76±7.95 | NA | 22.35±0.77 | NA |
| 3a | 4.85±0.23 | NA | 5.45±0.39 | NA |
| 3b | 53.48±1.06 | 5.62±1.52 | 84.63±4.76 | 9.18±0.43 |
| 3c | 0.32±1.52 | NA | 24.07±1.96 | NA |
| 4a | 1.64±0.69 | NA | 4.91±0.60 | NA |
| 4b | 8.93±3.84 | NA | 1.21±0.52 | NA |
| 4c | 1.41±3.43 | NA | 6.63±3.14 | NA |
| 5a | 61.81±3.58 | 12.82±1.46 | 31.13±4.69 | NA |
| 5b | 60.00±1.00 | 6.64±1.04 | 60.53±2.00 | 19.71±2.84 |
| 5c | 64.49±1.60 | 6.41±0.49 | 84.47±0.52 | 6.64±0.16 |
| 6a | 0.34±0.39 | NA | 15.64±5.71 | NA |
| 6b | 36.68±2.83 | NA | 85.71±2.80 | 12.24±0.11 |
| 6c | 0.02±0.35 | NA | 85.22±1.87 | 20.58±0.06 |
| Compd. | Cdc25Ba | PTP1Bc | ||
| Inhibition rate(%)b | IC50/(μg·mL-1) | Inhibition rate (%)b | IC50/(μg·mL-1) | |
| 6d | 14.99±1.04 | NA | 91.00±1.53 | 7.21±0.11 |
| 6e | 64.59±0.69 | 9.92±0.74 | 87.74±2.21 | 17.28±2.24 |
| 6f | 25.69±2.93 | NA | 88.47±1.38 | 11.81±0.47 |
| TZOA-1 | 4.28±0.98 | NA | 31.91±0.81 | NA |
| TZOA-2 | 44.10±0.00 | NA | 31.77±0.32 | NA |
| TZOA-3 | 0.37±1.02 | NA | 31.85±1.09 | NA |
| TZOA-4 | 69.32±2.77 | 6.66±1.77 | 40.60±1.18 | NA |
| TZOA-5 | 70.23±3.06 | 3.32±0.06 | 44.83±4.20 | NA |
| TZOA-6 | 77.53±1.20 | 3.01±0.19 | 48.10±0.92 | NA |
| TZOA-7 | 74.97±0.17 | 3.36±0.13 | 84.42±1.00 | 7.04±0.73 |
| TZOA-8 | 24.30±4.25 | NA | 86.75±1.65 | 15.11±0.98 |
| TZOA-9 | 57.61±0.91 | 9.38±0.99 | 84.66±0.90 | 9.32±1.86 |
| TZOA-10 | 63.73±4.13 | 5.49±0.48 | 84.70±8.06 | 3.24±0.06 |
| TZOA-11 | 92.25±0.21 | 0.71±0.04 | 93.40±0.39 | 2.37±0.38 |
| TZOA-12 | 89.07±1.18 | 1.12±0.13 | 89.16±6.93 | 8.50±0.07 |
| TZOA-13 | 90.28±0.07 | 1.04±0.25 | 77.33±0.73 | 26.18±6.77 |
| TZOA-14 | 93.37±1.09 | 0.88±0.15 | 88.15±0.14 | 4.78±0.24 |
| TZOA-15 | 94.54±1.50 | 0.86±0.50 | 88.78±0.56 | 4.22±0.64 |
| TZOA-16 | 91.13±0.06 | 1.02±0.12 | 89.73±3.35 | 4.36±0.78 |
| TZOA-17 | 96.18±0.16 | 0.66±0.33 | 92.71±0.74 | 6.12±0.58 |
| TZOA-18 | 99.23±0.09 | 0.46±0.34 | 91.52±0.39 | 3.04±0.02 |
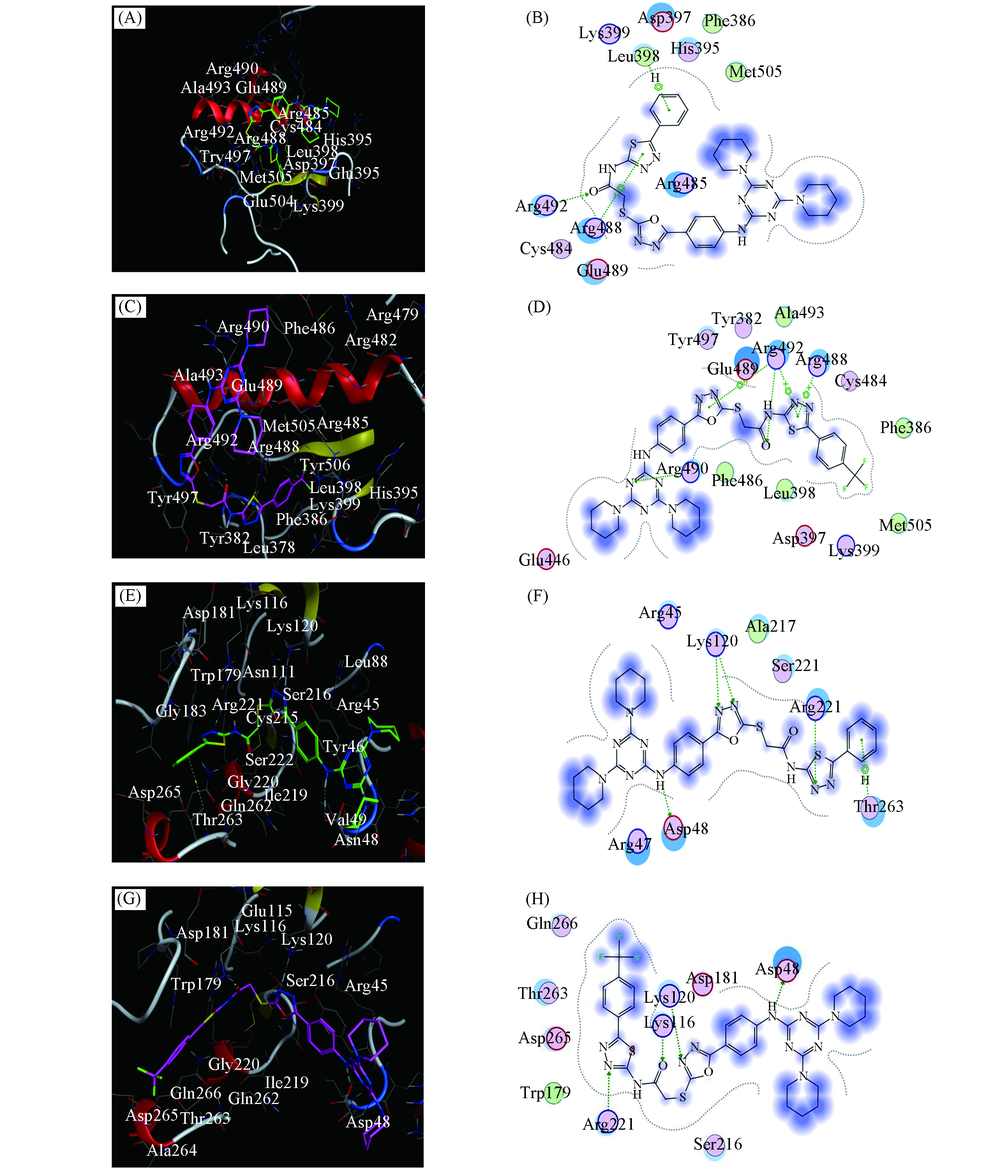
Fig.2 Molecular docking of compound TZOA-13, TZOA-18 with Cdc25B and PTP1B, respectively(A, B) TZOA-13 with Cdc25B; (C, D) TZOA-18 with Cdc25B; (E, F) TZOA-13 with PTP1B; (G, H) TZOA-18 with PTP1B.
| [1] | Kmar R., Kmar A., Jain S., Kaushik D., Eur. J. Med. Chem., 2011, 46(9), 3543-3550 |
| [2] | Husain A., Ahmad A., Alam M. M., Ajmal M., Ahuja P., Eur. J. Med. Chem., 2009, 44(9), 3798-3804 |
| [3] | Zhang X. M., Qiu M., Sun J., Zhang Y. B., Yang Y. S., Wang X. L., Tang J. F., Zhu H. L., Bioorg. Med. Chem., 2011, 19(21), 6518-6524 |
| [4] | Shingalapur R. V., Hosamani K. M., Keri R. S., Hugar M. H., Eur. J. Med. Chem.,2010, 45(5), 1753-1759 |
| [5] | Wang F. H., Qin Z. L., Huang Q., Front. Chem. China,2006, 1(1), 112-114 |
| [6] | Naresh K. R., Poornachandra Y., Nagender P., Santhosh K. G., Krishna S. D., Ganesh K. C., Narsaiah B., Bioorg. Med. Chem. Lett., 2016, 26, 4829-4831 |
| [7] | Almasirad A., Mousavi Z., Tajik M., Assarzadeh M. J., Shafiee A., DARU-J. Pharm. Sci., 2014, 22(22), 1-8 |
| [8] | Chaudhary S. K., Chaudhary M., Chaudhari A., Parmar S. S., J. Pharm. Sci., 1978, 67(11), 1507-1509 |
| [9] | El-Emam A. A., Al-Deeb O. A., Al-Omar M., Lehmann J., Bioorg. Med. Chem., 2004, 12(19), 5107-5113 |
| [10] | Dolman S. J., Gosselin F., O’Shea P. D., Davies I. W., J. Org. Chem., 2006, 71(25), 9548-9551 |
| [11] | Biftu T., Feng D. D., Liang G. B., Kuo H., Qian X., Naylor E. M., Colandrea V. J., Candelore M. R., Cascieri M. A., Colwell L. F., Forrest M. J., Hom G. J., Maclntyre D. E., Stearns R. A., Strader C. D., Wyvratt M. J., Fisher M. H., Weber A.E., Bioorg. Med. Chem. Lett., 2000, 10(13), 1431-1434 |
| [12] | Velázquez C., Rao P. N. P., McDonald R., Knaus E. E., Bioorg. Med. Chem., 2005, 13(8), 2749-2757 |
| [13] | Era M., Adachi C., Tsutsui T., Saito S., Chem. Phys. Lett., 1991, 178(56), 488-490 |
| [14] | Bentiss F., Traisnel M., Lagrenee M., Corros. Sci., 2000, 42(1), 127-146 |
| [15] | Yang X. H., Wen Q., Zhao T. T., Sun J., Li X., Xing M., Lu X., Zhu H. L., Bioorg. Med. Chem.,2012, 20, 1181-1187 |
| [16] | Liu T. T., Wan Y. C., Liu R. S., Ma L., Li M. Y., Fang H., Chem. Res. Chinese Universities,2016, 32(5), 768-774 |
| [17] | Wang C. X., Song H. L., Liu W. Q., Xu C. L., Chem. Res. Chinese Universities,2016, 32(4), 615-620 |
| [18] | Gür M., Muglu H., Çavus M. S., Güder A., Sayiner H. S., Kandemirli F., J. Mole. Struct.,2017, 1134, 40-50 |
| [19] | Tang J. Y., Liu J. B., Wu F. Y., Bioorg. Chem., 2016, 69, 29-36 |
| [20] | Solankee A., Kapadia K., C’iric A., Sokovic M., Doytchinova I., Geronikaki A., Eur. J. Med. Chem., 2010, 45(2), 510-518 |
| [21] | Saczewski F., Bułakowska A., Bednarski P.,Grunert R., Eur. J. Med. Chem., 2006, 41(2), 219-225 |
| [22] | Zacharie B., Abbott S. D., Bienvenu J. F., Cameron A. D., Cloutier J., Duceppe J. S., Ezzitouni A., Fortin D., Houde K., Lauzon C., Moreau N., Perron V., Wilb N., Asselin M., Doueet A., Fafard M. E., Gaudreau D., Grouix B., Sarra B. F., St A. N., Gagnon L., Penney C. L., J. Med. Chem., 2010, 53(3), 1138-1145 |
| [23] | Singla P., Luxami V., Puai K., Eur. J. Med. Chem., 2016, 117, 59-69 |
| [24] | Mishra A., Kaushik N. K., Verma A. K., Gupta R., Eur. J. Med. Chem.,2008, 43(10), 2189-2196 |
| [25] | Guo Y., Synthesis and Activity of Novel Bicyclic Heterocycle Modified Amide Sulfide/Mannich Base Derivatives, Liaoning Normal University, Dalian, 2016 |
| (国阳. 新型双杂环修饰酰胺硫醚/曼尼希碱衍生物的合成及活性研究, 大连: 辽宁师范大学, 2016) | |
| [26] | Rakse M., Karthikeyan C., Deora G. S., Moorthy N. S. H. N., Rathore V., Rawat A. K., SrivastavaA. K., Trivedi P., Eur. J. Med. Chem., 2013, 70, 469-476 |
| [27] | Zhang J., Ji F. J., Gu Y., Zhang X. Y., Qiao S. X., Pharmacol. Rep., 2014, 66(3), 515-519 |
| [28] | Hu M., Design and Synthesis of Potential PTP1B/SHP2/Cdc25B Carbohydrate Small Molecule Inhibitor Based on CuAAC, East China University of Science and Technology, Shanghai, 2011 |
| (胡敏. 基于CuAAC的潜在PTP1B/SHP2/Cdc25B糖类小分子抑制剂的设计与合成, 上海: 华东理工大学, 2011) | |
| [29] | Cao X. R., Yang X. Y., Wang P. X., Liang Y., Liu F., Tuerhong M., Jin D. Q., Xu J., Lee D. H., Ohizumi Y., Guo Y. Q., Bioorg. Chem.,2017, 75, 139-148 |
| [30] | Matsuno T., Kato M., Tsuchida Y., Takahashi M., Yaguchi S., Terada S., Chem. Pharm. Bull., 1997, 45(2), 291-296 |
| [31] | Zhu C.A., Synthesis and Properties of Novel Y-Type 1,2,4-Triazole/Triazolothiadiazole-1,3,5-Triazine Derivatives, Liaoning Normal University, Dalian, 2015 |
| (朱长安. 新Y型1,2,4-三唑/三唑并噻二唑-1,3,5-三嗪衍生物的合成及其性能研究, 大连: 辽宁师范大学, 2015) | |
| [32] | Zhang C. L., Li C. Y., Yang M., Zhu C. A., Sun X. N., Li Y. Z., Chinese J. Appl. Chem.,2017, 34(4), 385-393 |
| (张成路, 李传银, 杨蒙, 朱长安, 孙晓娜, 李益政. 应用化学, 2017, 34(4), 385-393) | |
| [33] | Li D. J., Long D. Q., Fu H. Q., Chem. Inform., 2006, 181(3), 519-526 |
| [34] | Karaman N., Sıcak Y., Tok T. T., Öztürk M., Karaküçük I. A., Dikmen M., Koçyiği K. B., Oruç-EmreE. E., Eur. J. Med. Chem., 2016, 124, 270-283 |
| [35] | Ábrahámi R. A., Kiss L., Barrio P., Fülöp F., Tetrahedron,2016, 72(47), 7526-7535 |
| [1] | WANG Linshuo, LI Kunjie, LIU Yumin, ZHAO Ruihong, LI Qing, QIAN Xin, ZHANG Fan, XUE Zhiwei. Theoretical Studies of Triphenyl-s-triazine Groups Regulating Photoelectric Properties of Sensitizing Dyes† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1653. |
| [2] | PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe [J]. Chem. J. Chinese Universities, 2020, 41(2): 228. |
| [3] | ZHANG Chenglu, SUN Xiaona, LI Chuanyin, CAI Jiying, WANG Jing, LI Yizheng, WANG Huayu. Synthesis and Bioactivities Evaluation of Novel 1-(1,3,5-Triazin-6-yl)-3-methyl-4-(5-phenyl-1,3,4-oxadiazol-2-yl) pyrazole† [J]. Chem. J. Chinese Universities, 2017, 38(10): 1764. |
| [4] | ZHANG Chenglu, WANG Xue, HU Xue, SUN Lijie, QU Ruifeng, GUO Yang, CHAI Jinhua, ZHU Chang’an. Syntheses and Biological Activities of V-Shaped Symmetrical Triazolothiazine Oxadiazole Derivatives† [J]. Chem. J. Chinese Universities, 2015, 36(3): 463. |
| [5] | ZHANG Chenglu, SUN Lijie, WU Feiyu, QU Ruifeng, ZHU Chang’an, WANG Xue, CHAI Jinhua, GUO Yang, HU Xue. Synthesis and Bioactivity of 1,4-Bis[3-aliphatic/aryl -1,2,4-triazole[3,4-b]-1,3,4-thiatriazole-6-yl] Aryl Derivatives† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1445. |
| [6] | ZHAO Lian, YUAN Xiang-Guo, ZHAO Jian-Peng, CHEN Hua*, LIU Li, LI Xiao-Liu, CAO Ke-Qiang. Design, Synthesis and Antifungal Activity Against Valsa Mali of the Triamino Substitued Triazines Bearing Aminopyrimidine Group [J]. Chem. J. Chinese Universities, 2011, 32(12): 2795. |
| [7] |
XIAO Shen-Chu, ZHENG Liang-Bin, XIE Wen-Zhong, LIN Wei-Ying, LI Qing-Hong, GUO Can-Cheng* . Simple One-pot Synthesis of Triazine-porphyrins [J]. Chem. J. Chinese Universities, 2009, 30(3): 478. |
| [8] | PAN Xiao-Xia, SUI Yu, XU Hong-Jie, YIN Jie, LIU Yan-Gang, GAO Jian, ZHU Zi-Kang, HUANG De-Yin, WANG Zong-Guang . One-step Synthesis of High Tg Organo-soluble Side-chain Fluorinated Polyimides Containing a Triazine-azo Chromophore [J]. Chem. J. Chinese Universities, 2002, 23(7): 1433. |
| [9] | DUAN Yue-Qin, LIU Xiao-Hang, GAO Xian-Hao, LI Chen-Xi, LI Guo-Hua, HE Bing-Lin. Studies on Molecular Recognition of Triazine Resin to Nucleic Base [J]. Chem. J. Chinese Universities, 2001, 22(S1): 214. |
| [10] | SUI Yu, GUO Xiao-Xia, YIN Jie, LIU Yan-Gang, GAO Jian-ZHU, Zi-Kang, HUANG De-Yin, WANG Zong-Guang. Synthesis and Characterization of High TgOrgano-Soluble Side-Chain Copolyimides Containing a Triazine-Azo Chromophore [J]. Chem. J. Chinese Universities, 2000, 21(12): 1944. |
| [11] | ZOU Jian-Ping, ZENG Run-Sheng, WANG Ai-Fen, LU Zhong-E . Synthesis and Reaction of α-Thiocarbonylthioformmorpholine [J]. Chem. J. Chinese Universities, 1998, 19(11): 1771. |
| [12] | LU Zhong-E, ZENG Run-Sheng, XI Hai-Tao. Synthesis of 3-Phenyl-5-substituted Anilino-6-methyl-1,2,4-triazine [J]. Chem. J. Chinese Universities, 1995, 16(8): 1257. |
| [13] | SHA Yao-Wu, CAI Meng-Shen. Chemistry of 1,2,4-Triazines (XXIV)──Synthesis of Pyrazolo-(5,4-e)-1,2,4-triazine Derivatives [J]. Chem. J. Chinese Universities, 1995, 16(4): 563. |
| [14] | SHA Yao-Wu, CAI Meng-Shen . Chemistry of 1,2,4-Triazines(XXII)──Synthesis of Imidazolo-(5,4e)-1,2,4-triazine [J]. Chem. J. Chinese Universities, 1994, 15(5): 708. |
| [15] | HAN Mei, SHI Xu-Ling, YANG Zhen-Jun, CAI Meng-Shen, CHENG Tie-Ming. A Study of 1,2,4-Triazines (XX) ——Study on Structures of Products from a Series of Substituted Benzene Sulfonation of 3-Methylthio-5-hydroxy-1,2,4-triazine by Spectrum Analysis [J]. Chem. J. Chinese Universities, 1993, 14(9): 1254. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||