

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (7): 1117.doi: 10.7503/cjcu20170043
• Articles: Inorganic Chemistry • Previous Articles Next Articles
SUN Kaifang, CAI Cheng, HOU Zongsheng, WANG Ying, REN Qizhi*( )
)
Received:2017-01-18
Online:2017-07-10
Published:2017-06-20
Contact:
REN Qizhi
E-mail:qzren@sjtu.edu.cn
Supported by:CLC Number:
TrendMD:
SUN Kaifang, CAI Cheng, HOU Zongsheng, WANG Ying, REN Qizhi. Synthesis, Characterization and Catalytic Properties of Series of Water-soluble Sulfonated Porphyrins†[J]. Chem. J. Chinese Universities, 2017, 38(7): 1117.
| TPP | TPPS | TMPP | TMPPS | TCPP | TCPPS | 3-TMPP | 3-TMPPS | Assignment |
|---|---|---|---|---|---|---|---|---|
| 3315.6 | 3298.5 | 3317.9 | 3318.2 | 3315.9 | 3316.8 | ν(N—H)(pyrrole) | ||
| 3424.5 | 3414.2 | 3427.0 | 3426.2 | 3438.7 | ν(O—H) | |||
| 3058.4 | 2934.2 | 2921.6 | 2931.0 | 3068.0 | 2970.8 | 2926.0 | 2908.1 | ν(C—H) |
| 3024.7 | 2859.8 | 2833.4 | 2836.1 | 2919.3 | 2920.2 | 2831.6 | ||
| 1593.7 | 1653.1 | 1607.4 | 1699.6 | 1596.7 | 1624.3 | 1630.0 | 1637.0 | ν(C |
| 1556.0 | 1534.5 | 1508.7 | 1591.0 | 1485.9 | 1464.8 | 1592.5 | ||
| 798.8 | 740.1 | 800.2 | 883.5 | 799.4 | 880.5 | 799.8 | 808.9 | ν(C—H)+ |
| 698.2 | 796.9 | 794.8 | δ(C—H)(phenyl) | |||||
| 967.2 | 910.8 | 964.5 | 975.1 | 964.8 | 964.2 | 973.2 | 977.5 | δ(N—H)(pyrrole) |
| 878.1 | 850.8 | 841.2 | 841.6 | 856.0 | 810.6 | 878.4 | 865.5 | π(C—H)(pyrrole) |
| 1252.4 | 1173.4 | 1188.5 | 1251.9 | ν(SO2) | ||||
| 1132.9 | 1151.8 | 1120.7 | 1150.7 | |||||
| 1024.4 | 1091.3 | 1015.1 | 1061.8 | |||||
| 1247.2 | 1248.8 | 1251.9 | 1258.0 | ν(C—O)(ph—O—C) | ||||
| 1092.0 | 1090.0 | ν(C—Cl) |
Table 1 IR spectra data(ν˙/cm-1) of water soluble sulfonated porphyrins
| TPP | TPPS | TMPP | TMPPS | TCPP | TCPPS | 3-TMPP | 3-TMPPS | Assignment |
|---|---|---|---|---|---|---|---|---|
| 3315.6 | 3298.5 | 3317.9 | 3318.2 | 3315.9 | 3316.8 | ν(N—H)(pyrrole) | ||
| 3424.5 | 3414.2 | 3427.0 | 3426.2 | 3438.7 | ν(O—H) | |||
| 3058.4 | 2934.2 | 2921.6 | 2931.0 | 3068.0 | 2970.8 | 2926.0 | 2908.1 | ν(C—H) |
| 3024.7 | 2859.8 | 2833.4 | 2836.1 | 2919.3 | 2920.2 | 2831.6 | ||
| 1593.7 | 1653.1 | 1607.4 | 1699.6 | 1596.7 | 1624.3 | 1630.0 | 1637.0 | ν(C |
| 1556.0 | 1534.5 | 1508.7 | 1591.0 | 1485.9 | 1464.8 | 1592.5 | ||
| 798.8 | 740.1 | 800.2 | 883.5 | 799.4 | 880.5 | 799.8 | 808.9 | ν(C—H)+ |
| 698.2 | 796.9 | 794.8 | δ(C—H)(phenyl) | |||||
| 967.2 | 910.8 | 964.5 | 975.1 | 964.8 | 964.2 | 973.2 | 977.5 | δ(N—H)(pyrrole) |
| 878.1 | 850.8 | 841.2 | 841.6 | 856.0 | 810.6 | 878.4 | 865.5 | π(C—H)(pyrrole) |
| 1252.4 | 1173.4 | 1188.5 | 1251.9 | ν(SO2) | ||||
| 1132.9 | 1151.8 | 1120.7 | 1150.7 | |||||
| 1024.4 | 1091.3 | 1015.1 | 1061.8 | |||||
| 1247.2 | 1248.8 | 1251.9 | 1258.0 | ν(C—O)(ph—O—C) | ||||
| 1092.0 | 1090.0 | ν(C—Cl) |
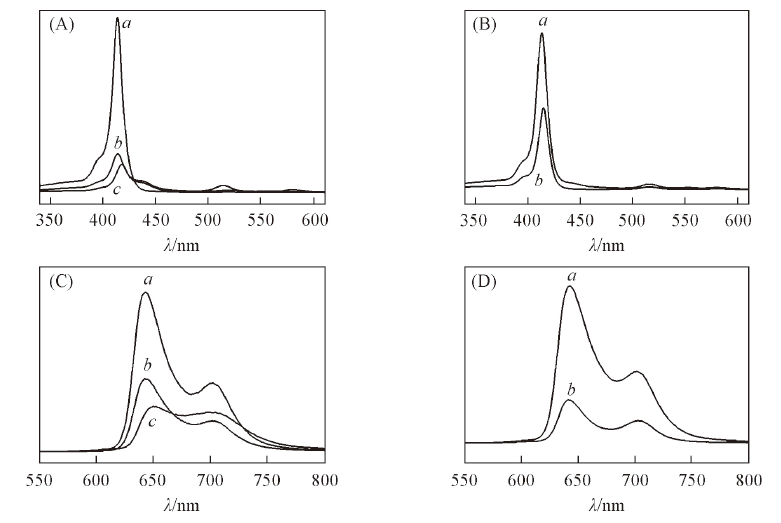
Fig.4 UV-Vis spectra(A, B) and fluorescence emission spectra(C, D) of sulfonated porphyrins (A, C) a. TDCPPS; b. TCPPS; c. TMPPS. (B, D) a. TPPS; b. 3-TMPPS.
| Porphyrin | Blankb | TPPS | TMPPS | TCPPS | 3-TMPPS | TDCPPS |
|---|---|---|---|---|---|---|
| 103k/min-1 | 3 | 80 | 64 | 18 | 31 | 17 |
| Conversion rate(%) | 19 | 91 | 95 | 81 | 92 | 78 |
Table 2 Dynamic data of 1,5-dihydroxynaphthalene photocatalyzed by water-soluble porphyrinsa
| Porphyrin | Blankb | TPPS | TMPPS | TCPPS | 3-TMPPS | TDCPPS |
|---|---|---|---|---|---|---|
| 103k/min-1 | 3 | 80 | 64 | 18 | 31 | 17 |
| Conversion rate(%) | 19 | 91 | 95 | 81 | 92 | 78 |
| [1] | Meunier B., de Visser S. P., Shaik S., Chem. Rev., 2004, 104(9), 3947—3980 |
| [2] | Castro K. A., Simões M. M., Neves M. G. P., Catal. Sci. Technol., 2014, 4(1), 129—141 |
| [3] | Hu Y. P., Shan D. L., Lu X. Q., Chem. J. Chinese Universities,2016, 37(6), 1082—1087 |
| (胡一平, 陕多亮, 卢小泉.高等学校化学学报, 2016, 37(6), 1082—1087) | |
| [4] | Liu S. S., Lu J. F., Wang M. K., J. Electrochem., 2016, 22(4), 340—355 |
| (刘双双, 鲁建峰, 王鸣魁.电化学, 2016, 22(4), 340—355) | |
| [5] | Tabata M., Nakajima K., Nyarko E., J. Inorg. Biochem., 2000, 78(4), 383—389 |
| [6] | Pedrosa L. F., de Souza M. C., Faustino M. A. F., Australian J. Chem., 2011, 64(7), 939—944 |
| [7] | Monteiro C. J., Pereira M. M., Azenha M. E., Photochem. Photobiol. Sci., 2005, 4(8), 617—624 |
| [8] | Long S. R., Wan Y., Xia A. D., Acta Chim. Sinica,2015, 73(7), 723—728) |
| (龙飒然, 宛岩, 夏安东.化学学报, 2015, 73(7), 723—728) | |
| [9] | Nimri S., Keinan E., J. Am. Chem. Soc., 1999, 121(39), 8978—8982 |
| [10] | Steinberg-Yfrach G., Rigaud J. L., Durantini E. N., Nature,1998, 392(6675), 479—482 |
| [11] | D’Oliveira J. C., Al-Sayyed G., Pichat P., Environ. Sci. Technol., 1990, 24(7), 990—996 |
| [12] | Wei B., Zhang Q. L., Liu Y. Z., Chem. Eng., 2016, 44(5), 11—16 |
| (魏冰, 张巧玲,刘有智.化学工程, 2016, 44(5), 11—16) | |
| [13] | Sun X. J., Appl. Chem. Ind., 2016, 45(5), 866—868 |
| (孙秀君. 应用化工, 2016,45(5), 866—868) | |
| [14] | Liu Q. Y., Liu L., Cui F. F., Environ. Sci. Technol., 2008, 31(7), 116—119 |
| (刘琼玉, 刘立, 崔菲菲.环境科学与技术, 2008, 31(7), 116—119) | |
| [15] | Nowakowska M., Kępczyński M., J. Photoch. Photobio.A,1998, 116(3), 251—256 |
| [16] | Skurlatov Y. I., Ernestova L., Vichutinskaya E., J. Photoch. Photobio.A,1997, 107(1), 207—213 |
| [17] | Lente G., Espenson J.H.,Chem. Commun., 2003, (10), 1162—1163 |
| [18] | DeRosa M. C., Crutchley R. J., Coordin. Chem. Rev., 2002, 2(33), 351—371 |
| [19] | Murtinho D., Pineiro M., Pereira M.M., J. Chem. Soc. Perkin Trans. 2, 2000, (12), 2441—2447 |
| [20] | Ren Q.Z., Yao Y., Ding X. J.,Chem. Commun., 2009, (31), 4732—4734 |
| [21] | Liu S. Y., Ren Q. Z., Ding X. J., Chem. J. Chinese Universities,2009, 30(7), 1272—1278 |
| (刘双艳, 任奇志,丁晓健.高等学校化学学报, 2009, 30(7), 1272—1278) | |
| [22] | Wang Y., Ren Q. Z., Guo L. T., Chem. J. Chinese Universities,2013, 34(7), 1576—1584 |
| (王颖, 任奇志, 郭琳童.高等学校化学学报, 2013, 34(7), 1576—1584) | |
| [23] | Song X. F., She Y. B., Liu R., Chemical Reagents,2011, 33(3), 193—195 |
| (宋旭锋, 佘远斌, 刘燃.化学试剂, 2011, 33(3), 193—195) | |
| [24] | Wang T., Han S. T., J. Hebei Normal University (Natural Science Edition), 2007, 4, 488—490 |
| (王涛, 韩士田. 河北师范大学学报(自然科学版), 2007, 4, 488—490) | |
| [25] | Monteiro C. J., Pereira M. M., Azenha M. E., Photochem. Photobiol. Sci., 2005, 4(8), 617—624 |
| [26] | Jin T. S., Zhang J. S., Wang A. Q., Chinese J. Org. Chem., 2005, 25(3), 335—338 |
| (靳通收, 张建设, 王爱卿.有机化学, 2005, 25(3), 335—338) | |
| [27] | Shi D. Q., Zhuang Q. Y., Chen J., Chin. J. Org. Chem., 2003, 23(7), 694—696 |
| (史达清, 庄启亚, 陈景.有机化学, 2003, 23(7), 694—696) | |
| [28] | Tang F. L., Chen X. H., Shang Y. Y., Chemical Reagents,1991, 13(3), 141—144 |
| (汤福隆, 陈秀华, 尚杨玉.化学试剂, 1991, 13(3), 141—144) | |
| [29] | Wang T., Han S. T., Hebei. J. Ind. Sci. Technol., 2005, 22(4), 239—244 |
| (王涛,韩士田. 河北工业科技, 2005, 22(4), 239—244) | |
| [30] | Fu X.C., Shen W. X., Yao T. Y., Hou W. H., Physical Chemistry, Higher Education Press, Beijing, 2005 |
| (傅献彩, 沈文霞, 姚天扬, 侯文华. 物理化学,北京:高等教育出版社, 2005) | |
| [31] | Silva E., Pereira M. M., Burrows H. D., Photochem. Photobiol. Sci., 2004, 3(2), 200—204 |
| [32] | Wu Y. Q., Ouyang R. Z., Chin. J. Chem. Phys., 1999, 12(1), 93—98 |
| (吴谊群, 欧阳瑞珍. 化学物理学报, 1999, 12(1), 93—98) |
| [1] | GONG Yanxi, WANG Jianbing, CHAI Buyu, HAN Yuanchun, MA Yunfei, JIA Chaomin. Preparation of Potassium Doped g-C3N4 Thin Film Photoanode and Its Application in Photoelectrocatalytic Oxidation of Diclofenac Sodium in Water [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220005. |
| [2] | LUO Jinyuan, CHEN Linlin, WANG Yi, LI Hong. Fabrication and Performance of a Novel Visible Light-driven Fuel Cell Based on Photocatalytic Oxidation of Uric Acid by CdS Nanoparticles and Electrocatalytic Reduction of Oxygen by a Copper(Ⅱ) Complex† [J]. Chem. J. Chinese Universities, 2015, 36(12): 2468. |
| [3] | YANG Qiu-Jing, XU Zi-Li, XIE Chao, XUE Bao-Yong, DU Yao-Guo, ZHANG Jia-Hua . Effect of Eu 3+ Doping on the Photocatalytic Activity of Nanoparticles TiO2 [J]. Chem. J. Chinese Universities, 2004, 25(9): 1711. |
| [4] | AI Shi-Yun, LI Jia-Qing, YANG Ya, CHAI Yi-Hao, SUN Zheng-Dong, JIN Li-Tong . Determination of Chemical Oxygen Demand with a New Photocatalytic Oxidation System [J]. Chem. J. Chinese Universities, 2004, 25(5): 823. |
| [5] | JING Li-Qiang, XU Zi-Li, DU Yao-Guo, SUN Xiao-Jun, WANG Ling, ZHOU Xiu-Qing, SHAN Hong-Yan, CAI Wei-Min. Investigation on Photocatalytic Oxidation Degradation of n-C7H16 over ZnO Ultrafine Particles [J]. Chem. J. Chinese Universities, 2002, 23(5): 871. |
| [6] | LI Dan-Zhen, ZHENG Yi, FU Xian-Zhi . Microwave Assisted Photocatalytic Oxidation and Its Applications [J]. Chem. J. Chinese Universities, 2002, 23(12): 2351. |
| [7] | ZHENG Yi, LI Dan-Zhen, FU Xian-Zhi . Microwave-assisted Heterogeneous Photocatalytic Oxidation of Ethylene [J]. Chem. J. Chinese Universities, 2001, 22(3): 443. |
| [8] | JING Li-Qiang, ZHENG Ying-Guang, XU Zi-Li, DONG Feng-Xia, SUN Xiao-Jun, CAI Wei-Min, XU Ying-Kai . Electronic Paramagnetic Resonance Characteristics of ZnO Ultrafine Particles and Their Photocatalytic Performance [J]. Chem. J. Chinese Universities, 2001, 22(11): 1885. |
| [9] | SHANG Jing, XU Zi-Li, DU Yao-Guo, GUO Hai-Chen . Studies on Photocatalytic Oxidation Reaction of SO2 over TiO2 [J]. Chem. J. Chinese Universities, 2000, 21(8): 1299. |
| [10] | ZHANG Yuan-Qin, HU Qi-Shan, ZENG Xian-Cheng, TIAN An-Min. Analog Thermogram Method(Ⅱ)──Studies on Thermokinetics of Irreversible Consecutive First-order Reactions [J]. Chem. J. Chinese Universities, 1997, 18(3): 463. |
| [11] | FANG Jing-huai, ZHANG Xiang-yang, LU Tong, LU Zu-hong. Sensitized Photocatalytic Oxidation of Dyes [J]. Chem. J. Chinese Universities, 1995, 16(S1): 226. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||