

Chem. J. Chinese Universities ›› 2020, Vol. 41 ›› Issue (2): 228.doi: 10.7503/cjcu20190605
Previous Articles Next Articles
PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian( )
)
Received:2019-11-25
Online:2020-02-10
Published:2019-12-31
Contact:
Daqian SONG
E-mail:songdq@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
PIAO Huilan,MA Pinyi,QIN Zucheng,JIANG Yanxiao,SUN Ying,WANG Xinghua,SONG Daqian. Determination of Triazine Herbicides from Fruit Juice Samples Using Effervescence Assisted Microextraction Method Based on Acidic Ionic Liquid Packed Syringe[J]. Chem. J. Chinese Universities, 2020, 41(2): 228.
| Analyte | Liner range/ (ng·mL-1) | Regression equation | Correlation coefficient, r | LOD/ (ng·mL-1) | LOQ/ (ng·mL-1) | RSD(%) | |
|---|---|---|---|---|---|---|---|
| Inter-day | Intra-day | ||||||
| Atraton | 1—200 | A=32273c-45035 | 0.9993 | 0.06 | 0.21 | 1.6 | 7.6 |
| Desmetryn | 1—200 | A=22833c-32310 | 0.9991 | 0.09 | 0.31 | 6.8 | 7.2 |
| Secbumeton | 2—200 | A=18533c-30871 | 0.9984 | 0.16 | 0.54 | 5.8 | 7.8 |
| Terbumeton | 2—200 | A=16835c-24740 | 0.9990 | 0.18 | 0.61 | 5.4 | 6.9 |
| Prometryn | 1—200 | A=26847c-34238 | 0.9990 | 0.06 | 0.18 | 2.9 | 6.3 |
| Analyte | Liner range/ (ng·mL-1) | Regression equation | Correlation coefficient, r | LOD/ (ng·mL-1) | LOQ/ (ng·mL-1) | RSD(%) | |
|---|---|---|---|---|---|---|---|
| Inter-day | Intra-day | ||||||
| Atraton | 1—200 | A=32273c-45035 | 0.9993 | 0.06 | 0.21 | 1.6 | 7.6 |
| Desmetryn | 1—200 | A=22833c-32310 | 0.9991 | 0.09 | 0.31 | 6.8 | 7.2 |
| Secbumeton | 2—200 | A=18533c-30871 | 0.9984 | 0.16 | 0.54 | 5.8 | 7.8 |
| Terbumeton | 2—200 | A=16835c-24740 | 0.9990 | 0.18 | 0.61 | 5.4 | 6.9 |
| Prometryn | 1—200 | A=26847c-34238 | 0.9990 | 0.06 | 0.18 | 2.9 | 6.3 |
| Matrix | Spiked/ (ng·mL-1) | Atraton | Desmetryn | Secbumeton | Terbumeton | Prometryn | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | ||
| Fruit Juice | 3.0 | 103.4 | 7.2 | 101.6 | 7.3 | 114.9 | 4.7 | 102.5 | 6.0 | 94.3 | 4.7 |
| 10.0 | 102.4 | 4.6 | 104.0 | 5.8 | 107.7 | 6.5 | 83.0 | 5.3 | 123.2 | 8.2 | |
| 20.0 | 106.0 | 2.0 | 105.0 | 2.2 | 96.6 | 1.5 | 89.6 | 3.6 | 116.5 | 5.8 | |
| Tea beverage | 3.0 | 90.4 | 6.9 | 123.5 | 4.4 | 107.6 | 8.7 | 103.2 | 4.0 | 94.4 | 4.6 |
| 10.0 | 91.5 | 2.6 | 101.0 | 5.3 | 100.6 | 2.7 | 85.2 | 3.4 | 100.3 | 6.1 | |
| 20.0 | 96.9 | 5.7 | 119.0 | 6.0 | 112.5 | 6.0 | 93.1 | 5.7 | 100.1 | 6.2 | |
| Water | 3.0 | 104.9 | 8.3 | 118.3 | 5.3 | 112.5 | 8.3 | 83.9 | 2.3 | 109.1 | 4.3 |
| 10.0 | 96.5 | 2.2 | 102.1 | 7.1 | 99.7 | 7.9 | 80.8 | 7.2 | 104.0 | 5.3 | |
| 20.0 | 92.1 | 0.4 | 112.3 | 1.5 | 101.4 | 1.5 | 94.9 | 3.6 | 113.8 | 3.0 | |
| Matrix | Spiked/ (ng·mL-1) | Atraton | Desmetryn | Secbumeton | Terbumeton | Prometryn | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | Recovery (%) | RSD (%) | ||
| Fruit Juice | 3.0 | 103.4 | 7.2 | 101.6 | 7.3 | 114.9 | 4.7 | 102.5 | 6.0 | 94.3 | 4.7 |
| 10.0 | 102.4 | 4.6 | 104.0 | 5.8 | 107.7 | 6.5 | 83.0 | 5.3 | 123.2 | 8.2 | |
| 20.0 | 106.0 | 2.0 | 105.0 | 2.2 | 96.6 | 1.5 | 89.6 | 3.6 | 116.5 | 5.8 | |
| Tea beverage | 3.0 | 90.4 | 6.9 | 123.5 | 4.4 | 107.6 | 8.7 | 103.2 | 4.0 | 94.4 | 4.6 |
| 10.0 | 91.5 | 2.6 | 101.0 | 5.3 | 100.6 | 2.7 | 85.2 | 3.4 | 100.3 | 6.1 | |
| 20.0 | 96.9 | 5.7 | 119.0 | 6.0 | 112.5 | 6.0 | 93.1 | 5.7 | 100.1 | 6.2 | |
| Water | 3.0 | 104.9 | 8.3 | 118.3 | 5.3 | 112.5 | 8.3 | 83.9 | 2.3 | 109.1 | 4.3 |
| 10.0 | 96.5 | 2.2 | 102.1 | 7.1 | 99.7 | 7.9 | 80.8 | 7.2 | 104.0 | 5.3 | |
| 20.0 | 92.1 | 0.4 | 112.3 | 1.5 | 101.4 | 1.5 | 94.9 | 3.6 | 113.8 | 3.0 | |
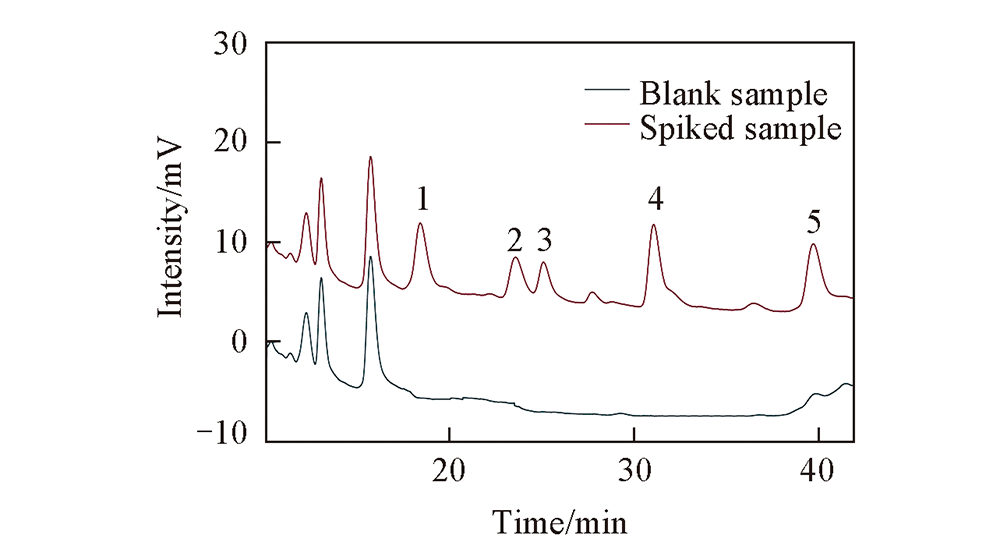
Fig.6 HPLC chromatograms of blank and spiked fruit juice samples Peak 1: atraton; peak 2: desmetryn; peak 3: secbumeton; peak 4: terbumeton; peak 5: prometryn.
| Matrix | Analytes | Extraction method | Detection | LOD/(ng·mL-1) | Recovery(%) | Ref. |
|---|---|---|---|---|---|---|
| Water | Triazine herbicides | SPME | HPLC-DAD | 0.05—0.2 | 86.0—94.6 | [ |
| Water | Triazine herbicides | HF-LLLME | HPLC-DAD | 0.07—0.69 | 85.2—113.0 | [ |
| Fruit juice | Triazine herbicides | CSDF-ME | HPLC-UV | 0.5—1.0 | 71.0—90.0 | [ |
| Milk | Triazine herbicides | MSPE | HPLC-DAD | 0.134—0.176 | 81.0—109.0 | [ |
| Milk | Triazine herbicides | SPE | HPLC-MS | 0.03—0.12 | 82.5—97.5 | [ |
| Water | Pesticide residues | UA-DLLME | GC-FID | 0.09—0.57 | 90.5—107.7 | [ |
| Fruit juice | Triazine herbicides | EA-DLLME | HPLC-UV | 0.06—0.18 | 80.8—123.5 | This work |
| Matrix | Analytes | Extraction method | Detection | LOD/(ng·mL-1) | Recovery(%) | Ref. |
|---|---|---|---|---|---|---|
| Water | Triazine herbicides | SPME | HPLC-DAD | 0.05—0.2 | 86.0—94.6 | [ |
| Water | Triazine herbicides | HF-LLLME | HPLC-DAD | 0.07—0.69 | 85.2—113.0 | [ |
| Fruit juice | Triazine herbicides | CSDF-ME | HPLC-UV | 0.5—1.0 | 71.0—90.0 | [ |
| Milk | Triazine herbicides | MSPE | HPLC-DAD | 0.134—0.176 | 81.0—109.0 | [ |
| Milk | Triazine herbicides | SPE | HPLC-MS | 0.03—0.12 | 82.5—97.5 | [ |
| Water | Pesticide residues | UA-DLLME | GC-FID | 0.09—0.57 | 90.5—107.7 | [ |
| Fruit juice | Triazine herbicides | EA-DLLME | HPLC-UV | 0.06—0.18 | 80.8—123.5 | This work |
| [1] |
Chen P. S., Haung W. Y., Huang S. D., J. Chromatogr. B, 2014,955/956, 116— 123
doi: 10.1016/j.jchromb.2014.02.032 URL |
| [2] |
Zhang W., Ruan G., Li X., Jiang X., Huang Y., Du F., Li J., Anal. Chim. Acta, 2019,1071, 17— 24
doi: 10.1016/j.aca.2019.04.041 URL |
| [3] |
Fang R., Chen G. H., Yi L. X., Shao Y. X., Zhang L., Cai Q. H., Xiao J., Food Chem., 2014,145, 41— 48
doi: 10.1016/j.foodchem.2013.08.028 URL |
| [4] | Chen L., Song D., Tian Y., Ding L., Yu A., Zhang H ., Tr AC-Trend. Anal. Chem., 2008,27, 151— 159 |
| [5] |
Qiao C., Bi S., Sun Y., Song D., Zhang H., Zhou W ., Spectrochim. Acta A, 2008,70, 136— 143
doi: 10.1016/j.saa.2007.07.038 URL |
| [6] |
Jiang Y., Piao H., Qin Z., Li X., Ma P., Sun Y., Wang X., Song D ., J. Sep. Sci., 2019,42, 2900— 2908
doi: 10.1002/jssc.v42.18 URL |
| [7] |
Sun T., Wang M., Wang D., Du Z ., Talanta, 2020,207, 120244
doi: 10.1016/j.talanta.2019.120244 URL |
| [8] |
Torbati M., Farajzadeh M. A., Mogaddam M. R. A., Torbati M., J. Sep. Sci., 2019,42, 1768— 1776
doi: 10.1002/jssc.v42.9 URL |
| [9] |
Guinez M., Canales R., Talio C., Gomez D., Smichowski P ., Talanta, 2020,206, 120182
doi: 10.1016/j.talanta.2019.120182 URL |
| [10] |
Rodríguez-González N., González-Castro M. J., Beceiro-González E., Muniategui-Lorenzo S., Microchem. J., 2017,133, 137— 143
doi: 10.1016/j.microc.2017.03.022 URL |
| [11] |
Yuan J., Cui Z., Cheng C., Wang X., Wang S., Song X., Li F., Acta Chromatogr., 2017,29, 487— 492
doi: 10.1556/1326.2016.00122 URL |
| [12] |
Camino-Sanchez F. J., Rodriguez-Gomez R., Zafra-Gomez A., Santos-Fandila A., Vilchez J. L., Talanta, 2014,130, 388— 399
doi: 10.1016/j.talanta.2014.07.022 URL |
| [13] |
Abujaber F., Guzman Bernardo F. J., Rodriguez Martin-Doimeadios R. C., Talanta, 2019,201, 266— 270
doi: 10.1016/j.talanta.2019.04.005 URL |
| [14] | Lasarte-Aragones G., Lucena R., Cardenas S., Valcarcel M., Anal. Chim. Acta, 2014,807, 61— 66 |
| [15] |
Wei Q., Song Z., Nie J., Xia H., Chen F., Li Z., Lee M ., J. Sep. Sci., 2016,39, 4603— 4609
doi: 10.1002/jssc.201600619 URL |
| [16] |
Ghoochani Moghadam A., Rajabi M., Hemmati M., Asghari A ., J. Mol. Liq., 2017,242, 1176— 1183
doi: 10.1016/j.molliq.2017.07.038 URL |
| [17] |
Yao L., Liu H., Wang X., Xu W., Zhu Y., Wang H., Pang L., Lin C., Food Chem., 2018,256, 212— 218
doi: 10.1016/j.foodchem.2018.02.132 URL |
| [18] |
Paduszyński K., Królikowski M., Orzeł P ., J. Mol. Liq., 2019,279, 733— 739
doi: 10.1016/j.molliq.2019.01.149 URL |
| [19] |
Wu X., Li X., Yang M., Zeng H., Zhang S., Lu R., Gao H., Xu D ., J. Chromatogr. A, 2017,1497, 1— 8
doi: 10.1016/j.chroma.2017.03.005 URL |
| [20] |
Pan H., Li H., Zhang H., Wang A., Yang S ., Fuel, 2019,239, 886— 895
doi: 10.1016/j.fuel.2018.11.093 URL |
| [21] | Guan L., Luo Q., Liang N., Yu W., Chem. J. Chinese Universities, 2018,34(6), 887— 892 |
| [22] |
Mehrdad A., Noorani N., Sep. Purif. Technol., 2019,226, 138— 145
doi: 10.1016/j.seppur.2019.05.086 URL |
| [23] |
Kong J., Zhu F., Huang W., He H., Hu J., Sun C., Xian Q., Yang S ., J. Chromatogr. A, 2019,1603, 92— 101
doi: 10.1016/j.chroma.2019.06.063 URL |
| [24] |
Farajzadeh M. A., Abbaspour M., Kazemian R., J. Chromatogr. A, 2019,1603, 51— 60
doi: 10.1016/j.chroma.2019.06.051 URL |
| [25] |
Wu Q., Feng C., Zhao G., Wang C., Wang Z ., J. Sep. Sci., 2012,35, 193— 199
doi: 10.1002/jssc.v35.2 URL |
| [26] |
Yang Q., Chen B., He M., Hu B ., Talanta, 2018,186, 88— 96
doi: 10.1016/j.talanta.2018.04.012 URL |
| [27] |
Ahmadi-Jouibari T., Pasdar Y., Pirsaheb M., Fattahi N ., Anal. Methods, 2017,9, 980— 985
doi: 10.1039/C6AY02839J URL |
| [28] |
Mohd N. I., Gopal K., Raoov M., Mohamad S., Yahaya N., Lim V., Zain N. N. M., Talanta, 2019,196, 217— 225
doi: 10.1016/j.talanta.2018.12.043 URL |
| [29] |
Zhang F., Zhao Q., Yan X., Li H., Zhang P., Wang L., Zhou T., Li Y., Ding L., Food Chem., 2016,197, 943— 949
doi: 10.1016/j.foodchem.2015.11.056 URL |
| [30] |
Cui S., Chen Q., Wang W., Miao J., Wang A., Chen J ., Chromatogr., 2013,76, 671— 678
doi: 10.1007/s10337-013-2441-7 URL |
| [1] | PAN Liya, LI Zhifeng, NI Yuxiang, YAO Zhengang, YU Zhiping, WU Wenkang, YING Anguo. Knoevenagel Condensation Catalyzed by Novel Acidic Ionic Liquid [J]. Chem. J. Chinese Universities, 2015, 36(1): 81. |
| [2] | YU Chuan-Ji, LIU Chen-Jiang*. Synthesis of β-Indolylketones Catalyzed by BrΦnsted Acidic Ionic Liquid [HSO3-bpy][HSO4] [J]. Chem. J. Chinese Universities, 2010, 31(6): 1158. |
| [3] | ZHI Hui-Zhen, LUO Jun, MA Wei, LÜ Chun-Xu*. Esterification of Aromatic Acids and Alcohols in Acidic PEG Thermoregulated Ionic Liquid [J]. Chem. J. Chinese Universities, 2008, 29(4): 772. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||