

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (7): 1381.doi: 10.7503/cjcu20180814
• Analytical Chemistry • Previous Articles Next Articles
MA Yukun2, WANG Haijun1,*( ), GUO Mengyan3
), GUO Mengyan3
Received:2018-12-03
Online:2019-07-10
Published:2019-07-12
Contact:
WANG Haijun
E-mail:qmuhjwang@163.com
Supported by:CLC Number:
TrendMD:
MA Yukun, WANG Haijun, GUO Mengyan. Preparation of ECB Molecularly Imprinted Film Fluorescent Sensor for Detection of Traditional Chinese Herbal Medicine†[J]. Chem. J. Chinese Universities, 2019, 40(7): 1381.
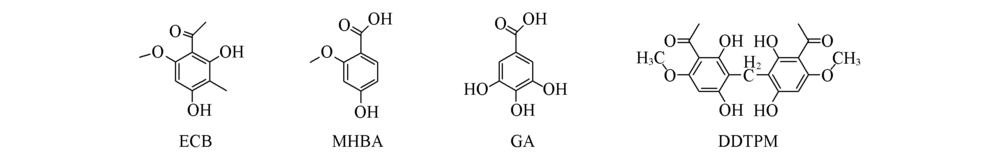
Scheme 2 Structures of ECB and three other similar compoundsECB: Ebracteolata Compound B; MHBA: 2-hydroxy-6-methoxyacetopheonone; GA: 3,5-trithydroxybenzoic acid; DDTPM: 3,3’-diacetyl-4,4’-dimethoxy-2,2’,6,6’-tetrahydroxydiphenylmethane.
| No. | Source | ECB content/(μmol·g-1) | |
|---|---|---|---|
| FL-MIF | HPLC | ||
| 1 | Medicine Garden of Qiqihar Medical University | 1.09 | 1.11 |
| 2 | Jiefang Township, Yi’an County, Qiqihar | 1.85 | 1.81 |
| 3 | Dajiazi Mountain, Nianzishan District, Qiqihar | 1.29 | 1.30 |
| 4 | Green grassland, Dulbert County, Daqing | 1.92 | 1.91 |
| 5 | Chabaqi Township, Arong Banner, Hulunbuir | 2.44 | 2.47 |
Table 1 Detection results of the ECB contents in Euphorbia fischeriana Steud from different habitats
| No. | Source | ECB content/(μmol·g-1) | |
|---|---|---|---|
| FL-MIF | HPLC | ||
| 1 | Medicine Garden of Qiqihar Medical University | 1.09 | 1.11 |
| 2 | Jiefang Township, Yi’an County, Qiqihar | 1.85 | 1.81 |
| 3 | Dajiazi Mountain, Nianzishan District, Qiqihar | 1.29 | 1.30 |
| 4 | Green grassland, Dulbert County, Daqing | 1.92 | 1.91 |
| 5 | Chabaqi Township, Arong Banner, Hulunbuir | 2.44 | 2.47 |
| [1] | Barrero R.A., Chapman B., Yang Y. F., Moolhuijzen P., Keeble-Gagnère G., Zhang N., Tang Q., Bellgard M. I., Qiu D. Y., BMC Genomics, 2011, 12, 600—613 |
| [2] | Jian B.Y., Zhang H., Liu J. C., Molecules, 2018, 23, 935—946 |
| [3] | Jian B.Y., Zhang H., Han C. C., Liu J. C., Molecules, 2018, 23, 387—397 |
| [4] | Shi Q. W., Su X. H.,Kiyota H., Chem. Rev., 2008, 108(10), 4295—4327 |
| [5] | Huang S.S., Li P., Zhang B. J., Deng S., Zhang H. L., Sun C. P., Huo X. K., Tian X. G., Ma X. C., Wang C., Phytochemistry Letters, 2017, 19, 151—155 |
| [6] | Wang H. B., Wang X. Y., Liu L. P., Qin G. W.,Kang T. G., Chem. Rev., 2011, 115, 2975—3011 |
| [7] | Wang C.J., Yan Q. L., Ma Y. F., Sun C. P., Chen C. M., Tian X. G., Han X. Y., Wang C., Deng S., Ma X. C., J. Nat. Prod., 2017, 80, 1248—1254 |
| [8] | Su X. L., Lin R. C., Wong S. K., Tsui S. K.,Kwan S. K., Phytochem. Anal., 2003, 14, 40—47 |
| [9] | Chen S., Hou P., Wang J., Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy, 2017, 173, 170—174 |
| [10] | Reja S.I., Gupta N., Bhalla V., Kaur D., Arora S., Kumar M., Sensors and Actuators B, 2016, 222, 923—929 |
| [11] | Xu Z.X., Gao H. J., Zhang L. M., Chen X. Q., Qiao X. G., J. Food Sci., 2011, 76, R69—R75 |
| [12] | Rai S., Singh B.K., Bhartiya P., Singh A., Kumar H., Dutta P. K., Mehrotra G. K., J. Lumin., 2017, 190, 492—503 |
| [13] | Xu J. G., Wang Z. B., Fluorescence Analysis, Science Press, Beijing, 2006, 3—17 |
| (许金钩, 王尊本. 荧光分析法, 北京: 科学出版社, 2006, 3—17) | |
| [14] | Vlatakis G., Andersson L. I., Müller R., Nature International Weekly Journal of Science, 1993, 361(6413), 645—647 |
| [15] | Chen X. J., Liu G. Q., Ren C. R., Gao M. J., Fan X. D., Chem. J. Chinese Universities, 2018, 39(2), 212—218 |
| (陈小娟, 刘根起, 任宸锐, 高敏君, 范晓东. 高等学校化学学报, 2018, 39(2), 212—218) | |
| [16] | Chen H. Y., Ding L., Liu M. L., Chem. J. Chinese Universities, 2015, 36(1), 67—73 |
| (陈海燕, 丁兰, 刘密兰. 高等学校化学学报, 2015, 36(1), 67—73) | |
| [17] | Zhao K.Y., Feng L. Z., Lin H. Q., Fu Y. F., Lin B. B., Cui W. K., Li S. D, Wei J. F., Catalysis Today, 2014, 3, 41—48 |
| [18] | Wu Y.L., Liu X. L., Cui J. Y., Dai J. D., Li C. X., Yan Y. S., Journal of Hazardous Materials, 2017, 323, 663—673 |
| [19] | Villar-Navarro M., Martín-Valero M.J., Fernández-Torres R. M., Callejón-Mochón M., Bello-López M. A., Journal of Chromatography B, 2017, 1044—1045 |
| [20] | Yu J.L., Wang X. Y. Kang Q., Shen D. Z., Chen L. X., Environ. Sci. Nano, 2017, 4, 493—502 |
| [21] | Ahmad R., Griffete N., Lamouri A., Felidj N., Chehimi M. M., Mangeney C., Chem. Mater, 2015, 27, 5464—5478 |
| [22] | Ji J., Zhou Z. H., Zhao X. L.,Sun J. D., Sun X. L., Biosens. Bioelectron., 2015, 66, 590—595 |
| [23] | Rao H. B., Chen M., Ge H. W., Lu Z. W.,Liu X., Zou P., Wang X. X., He H., Zeng X. Y., Wang Y. Y., Biosens. Bioelectron., 2017, 87, 1029—1035 |
| [24] | Lim G. W., Lim J. K.,Ahmad A. L., Anal. Bioanal. Chem., 2016, 408, 2083—2093 |
| [25] | Hou J., Li H.Y., Wang L., Zhang P., Zhou T. Y., Ding H., Ding L., Talanta, 2016, 146, 34—40 |
| [26] | Wang J. X., Qiu H., Shen H. Q., Pan J. M.,Dai X. H., Yan Y. S., Pan G. Q., Sellergren B., Biosens. Bioelectron., 2016, 85, 387—394 |
| [27] | Sergeyeva T., Yarynka D., Piletska E., Lynnik R., Zaporozhets O., Brovko O., Piletsky S., Elskaya A., Talanta, 2017, 175, 101—107 |
| [28] | Zhang X., Yang S., Jiang R., Sun L.Q., Pang S. P., Luo A. Q., Sensors and Actuators B: Chemical, 2018, 254, 1078—1086 |
| [29] | Hirano K., Kobayashi T., Ultrason. Sonochem., 2016, 30(18), 18—27 |
| [30] | Yang J., Huang L., Guo Z. Y., J. Lumin., 2016, 172, 290—296 |
| [31] | Ma Y., Study on Chemical Constituents and Antitumor Activities of Euphorbia Fischeriana, Qiqihar University, Qiqihar, 2017 |
| (马玉坤. 狼毒大戟化学成分及抗肿瘤活性研究, 齐齐哈尔: 齐齐哈尔大学, 2017) | |
| [32] | Song A., Wang X., LAM K. S., Tetrahedron Lett., 2003, 44(9), 1755—1758 |
| [33] | Barman N., Singha D., Sahu K., The Journal of Physical Chemistry A, 2013, 117(19), 3945—3953 |
| [1] | TANG Qian, DAN Feijun, GUO Tao, LAN Haichuang. Synthesis and Application of Quinolinone-coumarin-based Colorimetric Fluorescent Probe for Recognition of Hg2+ [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210660. |
| [2] | LI Lun, ZHANG Jingyan, LUO Jing, LIU Ren, ZHU Yi. Synthesis and Properties of UV/Vis-LED Excitable Photoinitiators Based on Coumarin Pyridinium Salt [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220178. |
| [3] | ZHAO Huijun, WU Tong, SUN Yue, DUAN Lian, MA Yanyu. A Coumarin-based Ratiometric Fluorescent Probe for BF3 Detection in Solution and Air [J]. Chem. J. Chinese Universities, 2021, 42(8): 2422. |
| [4] | KUANG Xiaojun, YI Jingwei, FANG Xiaoxia, LAI Dongmei, XU Hong. Preparation of Water-soluble Coumarin Fluorescent Substrate and Its Application in Droplet Based Digital Detection [J]. Chem. J. Chinese Universities, 2021, 42(11): 3537. |
| [5] | HAN Tao,CAI Xiaoxia,LI Cong,QIAO Congde,ZHAO Hui. Preparation and Properties Characterization of Biobased Dihydrocoumarin Toughened Epoxy Resin† [J]. Chem. J. Chinese Universities, 2019, 40(5): 1043. |
| [6] | HAN Ruixia,LÜ Jitao,ZHANG Shuzhen. Molecular Probe for the Determination of Hydroxyl Radicals in Heterogeneous Systems: Coumarin† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2658. |
| [7] | YANG Shu-Ping, HAN Li-Jun, PAN Yan, WANG Da-Qi, WANG Nan-Nan, WANG Ting. Synthesis, Characterization, Biological Activity and Interaction with Bovine Serum Albumin of 8- or 6-(3-Chlorobenzoyl)coumarin Derivatives [J]. Chem. J. Chinese Universities, 2013, 34(2): 364. |
| [8] | WEI Qing-Quan, WEI Tian-Xin*, PEI Xiao-Fei. Novel Method for in situ Monitoring the Polymerization Process of Molecularly Imprinted Films by Surface Plasmon Resonance [J]. Chem. J. Chinese Universities, 2011, 32(2): 204. |
| [9] | DONG She-Ying*, LI Jing, HUANG Ting-Lin. Microwave-induced Synthesis and Spectral Properties of New 4-Hydroxycoumarin Derivatives [J]. Chem. J. Chinese Universities, 2009, 30(8): 1516. |
| [10] | MA Xiang-Xia1; HE Xi-Wen1; ZHANG Mo1; LI Wen-You1*; ZHANG Yu-Kui1,2*. Studies on Binding of Molecularly Imprinted Polymer Composite Membranes to Coumarin-3-carboxylic Acid as Template and Permeable Selectivity [J]. Chem. J. Chinese Universities, 2006, 27(7): 1237. |
| [11] | LI Jun1, LIU Xiang-Jun1, ZHAO Rui1, XIONG Shao-Xiang1, MA Hui-Min1, L<SPAN lang=EN-US style=. Labeling Analysis of Neuropeptides with 7-Methoxycoumarin-3-carboxylic Acid N-Succinimidyl Ester [J]. Chem. J. Chinese Universities, 2006, 27(12): 2297. |
| [12] | LIU Xue-Feng, XIA Yong-Mei, CAO Yu-Hua, FANG Yun, ZOU Zhu-Yan, MAO Ben-Gang, DING Yi. Structure-performance Relationship of some Chinese Herb Components Containing Structural Unit of Coumarin During Binding to Bovine Serum Albumin [J]. Chem. J. Chinese Universities, 2006, 27(1): 150. |
| [13] | PANG Mei-Li, WANG Yong-Mei, MENG Ji-Ben, WANG Ji-Tao . Synthesis and Characteristics of Compounds Having Both Photochromic and Fluorescent Character [J]. Chem. J. Chinese Universities, 2000, 21(6): 903. |
| [14] | LI Long-Di, WU Ying-Guang, ZHANG Zhong-Xiao, TONG Ai-Jun. Studies on Cation Recognition Properties of 7-Methoxy-4-methene-coumarin-monoaza-18-crown-6 [J]. Chem. J. Chinese Universities, 2000, 21(12): 1824. |
| [15] | DU Da-Ming, WANG Yong-Mei, MENG Ji-Ben . Microenvironmental Effects on the Photochemical and Photophysical Processes of Long Chain Coumarin Esters [J]. Chem. J. Chinese Universities, 1998, 19(10): 1611. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||