

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (3): 408.doi: 10.7503/cjcu20170667
• Articles: Inorganic Chemistry • Previous Articles Next Articles
Received:2017-10-09
Online:2018-03-10
Published:2018-01-13
Contact:
YANG Binsheng
E-mail:yangbs@sxu.edu.cn
Supported by:CLC Number:
TrendMD:
SONG Kaili, YANG Binsheng. Spectroscopic Study on Ternary Complexes of Pyoverdine-terbium(Ⅲ)-ciprofloxacin†[J]. Chem. J. Chinese Universities, 2018, 39(3): 408.
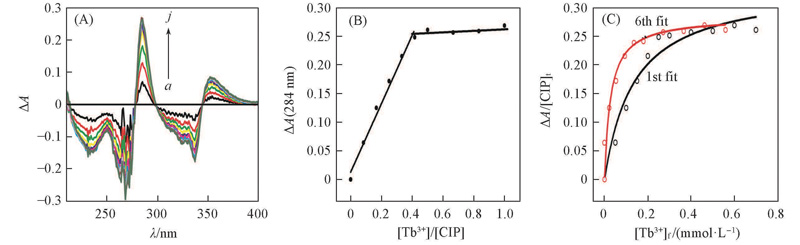
Fig.2 Difference UV spectra(A), titration curve(B) of 1.2 mL ciprofloxacin(0.05 mmol/L) with Tb3+(0.60 mmol/L) at pH=8.0, 50 mmol/L Tris-HCl, and fitting of ΔA/[CIP]t at 284 nm as a function of [Tb3+]f(C)V(Tb3+)/μL: a. 0; b. 10; c. 20; d. 30; e. 35; f. 40; g. 50; h. 60; i. 80; j. 100.
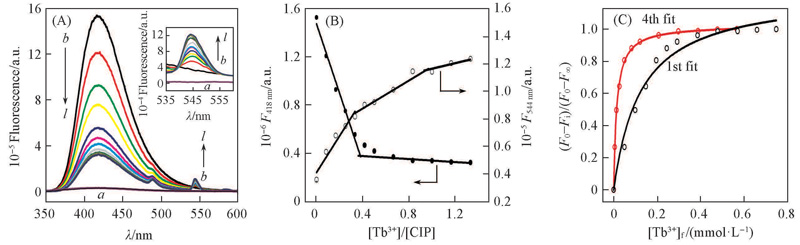
Fig.3 Fluorescence spectra(A), titration curve(B) of 1.2 mL ciprofloxacin(0.2 mmol/L) with Tb3+(2.4 mmol/L) at pH=8.0, 50 mmol/L Tris-HCl, and fitting of (F0-Fi)/(F0-F∞) as a function of [Tb3+]f(C)V(Tb3+)/μL: a. 0; b. 10; c. 20; d. 30; e. 40; f. 50; g. 60; h. 70; i. 80; j. 100; k. 120; l. 140.The inset in (A) is the enlarged part of the fluorescence spectra of ciprofloxacin with Tb3+.
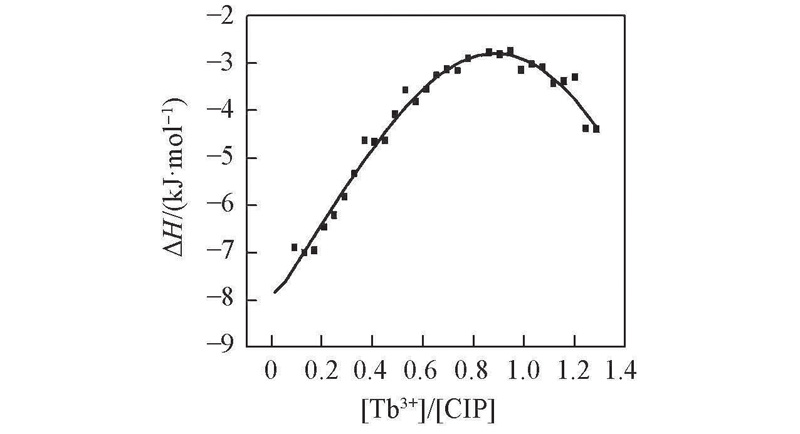
Fig.4 Isothermal titration calorimetry measurements produced by the titration of Tb3+(4 mmol/L) to 0.4 mmol/L CIP at pH=8.0 and 25 ℃The panel shows the integrated heat curve points normalized per mole of injectant as a function of molar ratio.
| Compound | Binding number, n | 10-4 Ki/(L·mol-1 ) | ΔH/(kJ·mol-1) | TΔS/(kJ·mol-1) | ΔG/(kJ·mol-1) |
|---|---|---|---|---|---|
| CIP-Tb3+ | 1 | 15.39±2.60 | -7.99±0.24 | 21.65 | -29.64 |
| 1 | 3.21±0.11 | 12.15±0.19 | 37.92 | -25.77 | |
| 1 | 1.20±0.50 | -50.19±0.11 | -26.78 | -23.41 | |
| Pvd-Tb3+ | 0.90±0.03 | 2.97±0.03 | -9.50±0.20 | 16.15 | -25.65 |
| CIP-Tb3+-Pvd | 1 | 0.31±0.04 | 1.77±0.58 | 21.78 | -20.01 |
| 1 | 0.27±0.04 | -222.55±10.82 | -202.79 | -19.79 |
Table 1 Binding parameters of Tb3+ complexes from ITC at pH=8.0 and 25 ℃
| Compound | Binding number, n | 10-4 Ki/(L·mol-1 ) | ΔH/(kJ·mol-1) | TΔS/(kJ·mol-1) | ΔG/(kJ·mol-1) |
|---|---|---|---|---|---|
| CIP-Tb3+ | 1 | 15.39±2.60 | -7.99±0.24 | 21.65 | -29.64 |
| 1 | 3.21±0.11 | 12.15±0.19 | 37.92 | -25.77 | |
| 1 | 1.20±0.50 | -50.19±0.11 | -26.78 | -23.41 | |
| Pvd-Tb3+ | 0.90±0.03 | 2.97±0.03 | -9.50±0.20 | 16.15 | -25.65 |
| CIP-Tb3+-Pvd | 1 | 0.31±0.04 | 1.77±0.58 | 21.78 | -20.01 |
| 1 | 0.27±0.04 | -222.55±10.82 | -202.79 | -19.79 |
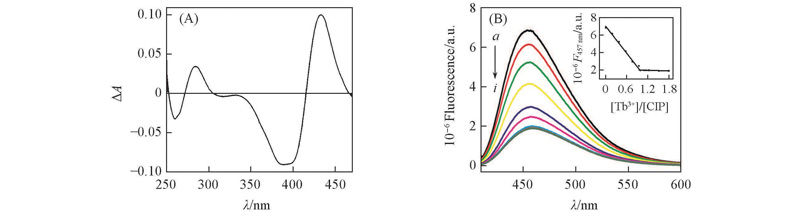
Fig.5 Difference UV-Vis spectrum of Pvd-Tb3+ at 1∶1(A) and fluorescence spectra generated by the titration approximately of 1 mL of Pvd(5 μmol/L) with Tb3+(100 μmol/L)(B) at pH=8.0 in 50 mmol/L Tris-HClV(Tb3+)/μL: a. 0; b. 10; c. 20; d. 30; e. 40; f. 50; g. 60; h. 70; i. 90. Inset in (B) is the fluorescence titration curve of Pvd with Tb3+.
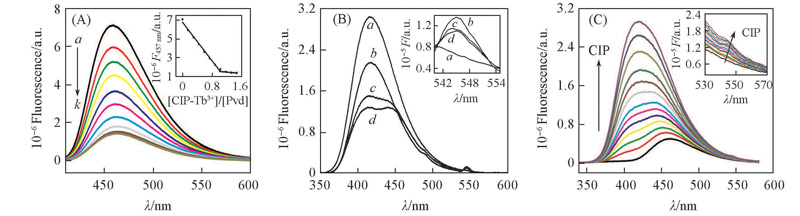
Fig.7 Fluorescence spectra for the addition of CIP-Tb3+(0.18 mmol/L) to the solution of 1.2 mL Pvd(0.01 mmol/L)(A), fluorescence spectra of CIP, CIP-Tb3+, Pvd-Tb3+-CIP, and 2Pvd-Tb3+-CIP(B), and fluorescence spectra for the addition of CIP(0.2 mmol/L) to Pvd-Tb3+(0.01 mmol/L) at pH=8.0 in 50 mmol/L Tris-HCl(C)(A) V(Tb3+)/μL: a. 0; b. 10; c. 20; d. 30; e. 40; f. 50; g. 60; h. 70; i. 80; j. 90; k. 100. Inset in (A) is the fluorescence titration curve of CIP-Tb3+ with Pvd. (B) a. CIP; b. CIP-Tb3+; c. Pvd-Tb3+-CIP; d. 2Pvd-Tb3+-CIP.
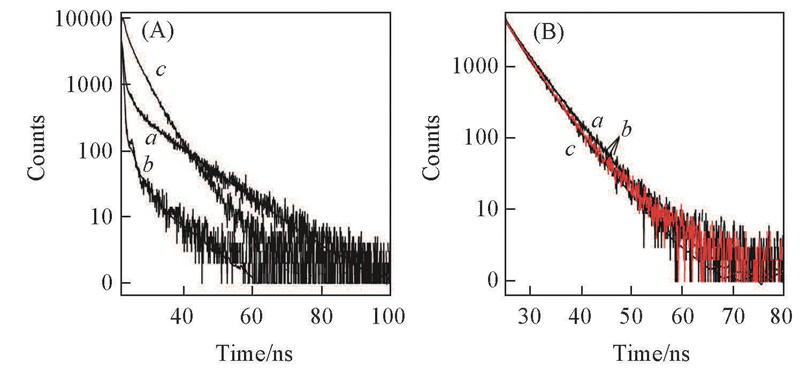
Fig.9 Fluorescence decay curves for CIP(a), CIP-Tb3+(1∶1)(b), CIP-Tb3+-Pvd(1∶1∶1)(c)(A) and Pvd(a), Pvd-Tb3+(1∶1)(b) and Pvd-Tb3+-CIP(1∶1∶1)(c)(B)(A) λex=405 nm, λem=418 nm; (B) λex=405 nm, λem=457 nm.
| System | <τ>/ns | χ2 | System | <τ>/ns | χ2 |
|---|---|---|---|---|---|
| CIP | 10.74±0.05 | 1.01 | Pvd-Tb3+ | 4.27±0.02 | 1.05 |
| Pvd | 4.48±0.02 | 1.06 | Pvd-Tb3+-CIP(418 nm) | 4.64±0.03 | 1.09 |
| CIP-Tb3+ | 5.17±0.04 | 1.10 | Pvd-Tb3+-CIP(457 nm) | 4.15±0.02 | 1.02 |
Table 2 Fluorescence decay parameters for CIP, Tb3+ and Pvd ternary system*
| System | <τ>/ns | χ2 | System | <τ>/ns | χ2 |
|---|---|---|---|---|---|
| CIP | 10.74±0.05 | 1.01 | Pvd-Tb3+ | 4.27±0.02 | 1.05 |
| Pvd | 4.48±0.02 | 1.06 | Pvd-Tb3+-CIP(418 nm) | 4.64±0.03 | 1.09 |
| CIP-Tb3+ | 5.17±0.04 | 1.10 | Pvd-Tb3+-CIP(457 nm) | 4.15±0.02 | 1.02 |
| [1] | Gasser V., Guillon L., Cunrath O., Schalk I. J., J. Inorg. Biochem., 2015, 148, 27—34 |
| [2] | Mashiach R., Meijler M. M., Org. Lett., 2013, 15(7), 1702—1705 |
| [3] | Hijazi S., Visca P., Frangipani E., Front. Cell. Infect. Microbiol., 2017, 7, 12 |
| [4] | Cézard C., Farvacques N., Sonnet P., Curr. Med. Chem., 2014, 22(2), 165—186 |
| [5] | Hannauer M., SchäferM., Hoegy F., Gizzi P., Wehrung P., Mislin G. L., Budzikiewicz H., Schalk I. J., FEBS Lett., 2012, 586(1), 96—101 |
| [6] | Wurst J. M., Drake E. J., Theriault J. R., Jewett I. T., VerPlank L., Perez J. R., Dandapani S., Palmer M., Moskowitz S. M., Schreiber S. L., Munoz B., Gulick A. M., ACS Chem. Biol., 2014, 9(7), 1536—1544 |
| [7] | Albrecht-Gary A. M., Blanc S., Rochel N., Ocacktan A. Z., Abdallah M. A., Inorg. Chem., 1994, 33, 6391—6402 |
| [8] | Dane P. R., Pawar S. P., Kankariya R. A., Chaudhari B. L., RSC Adv., 2016, 6(10), 8503—8510 |
| [9] | Braun V., Int. J. Med. Microbiol., 2001, 291(2), 67—79 |
| [10] | De Vos D., de Chial M., Cochez C., Jansen S., Tummler B., Meyer J. M., Cornelis P., Arch. Microbiol., 2001, 175(5), 384—388 |
| [11] | Meyer J. M., Geoffroy V. A., Baysse C., Cornelis P., Barelmann I., Taraz K., Budzikiewicz H., Arch. Biochem. Biophys., 2002, 397(2), 179—183 |
| [12] | Schalk I. J., J. Inorg. Biochem., 2008, 102(5/6), 1159—1169 |
| [13] | Yang B. S., Hoegy F., Mislin G. L. A., Mesini P. J., Schalk I. J., J. Inorg. Biochem., 2011, 105(10), 1293—1298 |
| [14] | Braud A., Hoegy F., Jezequel K., Lebeau T., Schalk I. J., Environ. Microbiol., 2009, 11(5), 1079—1091 |
| [15] | Zhang W. L., Xu C. H., Yang B. S., Chinese J. Inorg. Chem., 2014, 30(1), 106—112 |
| (张文龙, 许彩虹, 杨斌盛.无机化学学报,2014, 30(1), 106—112) | |
| [16] | Song Z., Dong J. L., Ren Y. H., Yuan W., Zhang C. F., Yang B. S., Chem. J. Chinese. Universities, 2016, 37(7), 1245—1249 |
| (宋珍, 董金龙, 任跃红, 袁雯, 张彩凤, 杨斌盛.高等学校化学学报,2016, 37(7), 1245—1249) | |
| [17] | Ferrer M., Méndezgarcía C., Rojo D., Barbas C., Moya A., Biochem. Pharmacol., 2017, 134, 114—126 |
| [18] | Phiboonchaiyanan P. P., Kiratipaiboon C., Chanvorachote P., Chem. Biol. Interact., 2016, 250, 1—11 |
| [19] | Attia K. A., Nassar M. W., Elzeiny M. B., SeragA., Spectrochim. Acta Part A, 2015, 154, 232—236 |
| [20] | Li J., Huang W. Y., Qian S. S., Li Q. Y., Zhu H. L., Inorg. Chim. Acta, 2015, 435, 16—24 |
| [21] | Chen H., Ma L. Q., Gao B., Gu C., J. Hazard. Mater., 2013, 262, 805—811 |
| [22] | Wu S. Q., Cao Q. E., Zhao Y. K., Zhang W. J., Chen X. G., Hu Z. D., Chinese J. Analyt. Chem., 2000, 28(12), 1462—1466 |
| (吴淑清, 曹秋娥, 赵云昆, 张五卷, 陈兴国, 胡之德.分析化学,2000, 28(12), 1462—1466) | |
| [23] | Yang J., Ren Y., Xu Y. Z., Su Y. L., Zhang L., Liu Z., Wang F., Yang L. M., Wang J., Gao H. C., Ye F., Wu J. G., Chem. J. Chinese Universities, 2004, 25(2), 243—246 |
| (杨军, 任宇, 徐怡庄, 苏允兰, 张莉, 刘智, 王凡, 杨丽敏, 王晶, 高宏成, 叶放, 吴瑾光.高等学校化学学报,2004, 25(2), 243—246) | |
| [24] | Song Z., Wang J. L., Yang B. S., Spectrochim. Acta Part A, 2014, 118, 454—460 |
| [25] | Karla A., Robinson S. G., Lyngaas S. S., Cherala S. S., Hartzell M., Mei S .,Vilic A., Girel J. K., Kuemmell A., Vrettos J. S., Zielinski J., Liechti K., Jin L., Inorg. Chim. Acta, 2016, 441, 181—191 |
| [26] | Shi E. X., Zhang W. L., Zhao Y. Q., Yang B. S., RSC Adv., 2017, 7, 27139—27149 |
| [27] | Yang P., Yang B.S., Introduction to Ion Probe Methods, Science Press, Beijing, 1994, 167—168 |
| (杨频, 杨斌盛. 离子探针方法导论, 北京: 科学出版社, 1994, 167—168) |
| [1] | YANG Mingxuan, MA Jie, SUN Yiran, XIONG Xinzhu, LI Chenlu, LI Qiang, CHEN Junhong. Synthesis of Carbon Nanotubes/FeS Fenton-like Catalyst and Its Catalytic Properties† [J]. Chem. J. Chinese Universities, 2014, 35(3): 570. |
| [2] | LIU Jun-Bo, TANG Shan-Shan, SUN Jia-Ni, JIN Rui-Fa. Theoretical Research on Self-assembly System of Molecular Imprinted Polymers Formed by Ciprofloxacin and Trifluoromethacrylic Acid [J]. Chem. J. Chinese Universities, 2013, 34(11): 2566. |
| [3] | FU Shi-Tao, ZHANG Zhi, YU Kai-Chao, WONG Wai-Kwok, LI Zao-Ying*. Synthesis, Characterization and Near-infrared Photoluminescence of Ytterbium(Ⅲ) Monoporphyrinate Complexes [J]. Chem. J. Chinese Universities, 2009, 30(11): 2131. |
| [4] | YANG Feng-Zhen, YI Guang-Shun, CHEN De-Pu, CHENG Jing . Synthesis and Up-conversion Luminescence Properties of Nanocrystal Yb, Ho Co-doped Sodium Yttrium Fluoride [J]. Chem. J. Chinese Universities, 2004, 25(9): 1589. |
| [5] | YANG Jun, REN Yu, XU Yi-Zhuang, SU Yun-Lan, ZHANG Li, LIU Zhi, WANG Fan, YANG Li-Min, WANG Jing, GAO Hong-Cheng, YE Fang, WU Jin-Guang . Interaction Between DNA and Terbium(Ⅲ)-Ciprofloxacin Fluorescence Complex and New Tissue Staining Method [J]. Chem. J. Chinese Universities, 2004, 25(2): 243. |
| [6] | ZHANG Ning, ZHAO Bang-Tun, LIU Yu. Luminescence Behavior on the Formation Complex of Water Soluble Calixarene Derivative with Earth Terbium(Ⅲ) Ion [J]. Chem. J. Chinese Universities, 2002, 23(8): 1533. |
| [7] | LIAN Ning, ZHAO Hui-Chun, SUN Chun-Yan, JIN Lin-Pei, ZHANG Zhong-Lun, ZHENG Yan-Zhen. Terbium Sensitized Chemiluminescence of Gatifloxacin and Its Application [J]. Chem. J. Chinese Universities, 2002, 23(4): 564. |
| [8] | YANG Bin-Sheng, LI Ying-Qi . Effect of Chelate Reagents on Terbium-apotransferrin (Ⅱ)——The Properties of Anions Binding to Terbium-apotransferrin [J]. Chem. J. Chinese Universities, 2001, 22(7): 1086. |
| [9] | LIU De-Long, SUN Da-Ye, YANG Yan-Sheng, ZHANG Hong-Jie, WANG Shu-Bin, GONG Meng-Lian . Studies on Plant Extracellular Calmodulin by Lanthanide Luminescence Probe [J]. Chem. J. Chinese Universities, 2000, 21(6): 860. |
| [10] | ZHANG Xiao-Wei, ZHAO Feng-Lin, LI Ke-An. Studies on the Reaction Between Ciprofloxacin and Bovine Serum Albumin [J]. Chem. J. Chinese Universities, 1999, 20(7): 1063. |
| [11] | ZHANG Ai-Ping, YANG Bin-Sheng . Effect of Chelate Reagents on Terbium-apotransferrin [J]. Chem. J. Chinese Universities, 1999, 20(7): 1106. |
| [12] | GUO Dong-Sheng, YUAN Xiao-Ying . The Regulation of Mg(Ⅱ) to the Combination of Ciprofloxacin and Calf Thymus DNA [J]. Chem. J. Chinese Universities, 1999, 20(4): 584. |
| [13] | CUI Bin, CHEN Kai-Xun, TANG Zong-Xun, GUO Zhi-Zhen . A Study on the Phase Equilibrium of the Ternary System of Yb(ClO4)3-BAPHDCA H2O3BAPHDCAH2O(30℃) and the Preparation of Ternary Complex [J]. Chem. J. Chinese Universities, 1999, 20(2): 181. |
| [14] | FU Jie, ZHUO Ren-Xi, FAN Chang-Lie . Studies on the Syntheses and Properties of Poly(ester anhydride)s for DDS [J]. Chem. J. Chinese Universities, 1998, 19(5): 813. |
| [15] | JING Hai-Qiang, WU Guo-Qing, DU Bao-Shi . Preparation of Phosphors MBPO5: Eu2+ and MBPO5: Yb2+ in Air [J]. Chem. J. Chinese Universities, 1997, 18(9): 1425. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
