

Chem. J. Chinese Universities ›› 2017, Vol. 38 ›› Issue (1): 141.doi: 10.7503/cjcu20160596
• Polymer Chemistry • Previous Articles Next Articles
WANG Yifu, DONG Jinxin, WANG Jilin*( ), WANG Lulu, FENG Ruijiang
), WANG Lulu, FENG Ruijiang
Received:2016-08-23
Online:2017-01-10
Published:2016-12-06
Contact:
WANG Jilin
E-mail:wangjilin1978@163.com
Supported by:CLC Number:
TrendMD:
WANG Yifu, DONG Jinxin, WANG Jilin, WANG Lulu, FENG Ruijiang. Ordered Self-assembly of Gemini Molecules in Mesoporous Silica Channels Constructing Normal Micelles to Assist the Migration of OH-†[J]. Chem. J. Chinese Universities, 2017, 38(1): 141.
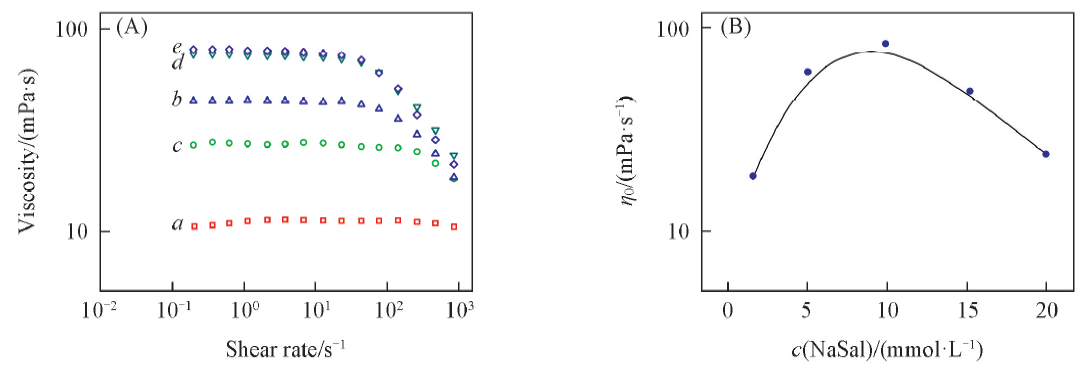
Fig.1 Effect of NaSal concentration on steady rheology(A) and zero-shear viscosity η0(B) of 100 mmol/L G8-2-8 solution c(NaSat)/(mmol·L-1): a. 2; b. 5; c. 10; d. 15; e. 20.
| Sample | BET surface area/(m2·g-1) | Pore volume/(cm3·g-1) | Small mean pore/nm | Large mean pore/nm |
|---|---|---|---|---|
| G8-2-8/NaSal-unloaded | 948.1 | 1.34 | 3.8 | 9.8 |
| G8-2-8/NaSal-loaded | 282.1 | 0.34 | 3.8 | 9.5 |
Table 1 N2 adsorption-desorption results of NaSal-loaded and NaSal-unloaded SiO2 nanoparticles
| Sample | BET surface area/(m2·g-1) | Pore volume/(cm3·g-1) | Small mean pore/nm | Large mean pore/nm |
|---|---|---|---|---|
| G8-2-8/NaSal-unloaded | 948.1 | 1.34 | 3.8 | 9.8 |
| G8-2-8/NaSal-loaded | 282.1 | 0.34 | 3.8 | 9.5 |
| Sample | 103 Conductivity/(S·cm-1) | IEC/(mmol·g-1) | ||
|---|---|---|---|---|
| 30 ℃ | 80 ℃ | Experimental | Theoretical | |
| NaSal(No NaSal) | NaSal(No NaSal) | NaSal(No NaSal) | ||
| PSF | 0(0) | 0(0) | 0(0) | 0 |
| Pw-5 | 3.29(2.21) | 5.20(2.91) | 0.14±0.02(0.15) | 0.16 |
| Pw-10 | 4.04(2.43) | 5.86(3.44) | 0.30±0.03(0.31) | 0.32 |
| Pw-15 | 4.52(2.60) | 6.37(4.19) | 0.46±0.04(0.46) | 0.48 |
| Pw-20 | 5.68(3.65) | 7.02(4.62) | 0.61±0.04(0.62) | 0.64 |
Table 2 Anionic conductivity and IEC of Pw-X anion exchange membranes before and after adding NaSal
| Sample | 103 Conductivity/(S·cm-1) | IEC/(mmol·g-1) | ||
|---|---|---|---|---|
| 30 ℃ | 80 ℃ | Experimental | Theoretical | |
| NaSal(No NaSal) | NaSal(No NaSal) | NaSal(No NaSal) | ||
| PSF | 0(0) | 0(0) | 0(0) | 0 |
| Pw-5 | 3.29(2.21) | 5.20(2.91) | 0.14±0.02(0.15) | 0.16 |
| Pw-10 | 4.04(2.43) | 5.86(3.44) | 0.30±0.03(0.31) | 0.32 |
| Pw-15 | 4.52(2.60) | 6.37(4.19) | 0.46±0.04(0.46) | 0.48 |
| Pw-20 | 5.68(3.65) | 7.02(4.62) | 0.61±0.04(0.62) | 0.64 |
| Membrane material | Ionic group | IEC/(mmol·g-1) | 103 Conductivity/ (S·cm-1) | Con/IEC | Ref. |
|---|---|---|---|---|---|
| PSF/ G8-2-8 | QA | 0.15 | 3.29(30 ℃) | 21.93 | This research |
| PSF/G8-2-8 | QA | 0.15 | 5.20(80 ℃) | 34.66 | This research |
| Poly(tetrafluoroethene-co-hexafluoropropylene) | Q | 0.72 | 20(25 ℃) | 27.7 | [ |
| Poly(MMA-co-BA-co-VBC)a | QATMA | 0.66—1.25 | 5.2—13.5(80 ℃) | 7.8—10.8 | [ |
| PVBC grafted FEPb | QATMA | 0.92—1.10 | 11—16(25 ℃) | 11.9—14.5 | [ |
| ETFE grafted with VBC | QATMA | 1.03±0.11 | 27±5(20 ℃) | 26.2 | [ |
| Poly(arylene ether sulfone) | QATMA | 1.47—2.62 | 15-65(80 ℃) | 10.2—24.8 | [ |
| Cardo polyetherketone | QATMA | 0.11 | 1.6(20 ℃) | 14.5 | [ |
| SEBSc | QATMA | 0.3 | 5(30 ℃) | 16.6 | [ |
| Polyepichlorydrin | Q | 1.3 | 2.5(20 ℃) | 1.9 | [ |
| Silica/poly(2,6-dimethyl-1,4-phenylene oxide) | QATEA | 2.01 | 12(30 ℃) | 5.9 | [ |
| Polystyrene(ethylene utylenes) polystyrene | QATEA | 0.57 | 0.69(30 ℃) | 1.21 | [ |
| Poly(arylene ether sulfone) | Guanidinium | 0.79—1.89 | 5—45(20 ℃) | 6.3—23.8 | [ |
Table 3 IEC and conductivity values reported in the literature for different AEMs
| Membrane material | Ionic group | IEC/(mmol·g-1) | 103 Conductivity/ (S·cm-1) | Con/IEC | Ref. |
|---|---|---|---|---|---|
| PSF/ G8-2-8 | QA | 0.15 | 3.29(30 ℃) | 21.93 | This research |
| PSF/G8-2-8 | QA | 0.15 | 5.20(80 ℃) | 34.66 | This research |
| Poly(tetrafluoroethene-co-hexafluoropropylene) | Q | 0.72 | 20(25 ℃) | 27.7 | [ |
| Poly(MMA-co-BA-co-VBC)a | QATMA | 0.66—1.25 | 5.2—13.5(80 ℃) | 7.8—10.8 | [ |
| PVBC grafted FEPb | QATMA | 0.92—1.10 | 11—16(25 ℃) | 11.9—14.5 | [ |
| ETFE grafted with VBC | QATMA | 1.03±0.11 | 27±5(20 ℃) | 26.2 | [ |
| Poly(arylene ether sulfone) | QATMA | 1.47—2.62 | 15-65(80 ℃) | 10.2—24.8 | [ |
| Cardo polyetherketone | QATMA | 0.11 | 1.6(20 ℃) | 14.5 | [ |
| SEBSc | QATMA | 0.3 | 5(30 ℃) | 16.6 | [ |
| Polyepichlorydrin | Q | 1.3 | 2.5(20 ℃) | 1.9 | [ |
| Silica/poly(2,6-dimethyl-1,4-phenylene oxide) | QATEA | 2.01 | 12(30 ℃) | 5.9 | [ |
| Polystyrene(ethylene utylenes) polystyrene | QATEA | 0.57 | 0.69(30 ℃) | 1.21 | [ |
| Poly(arylene ether sulfone) | Guanidinium | 0.79—1.89 | 5—45(20 ℃) | 6.3—23.8 | [ |
| c(NaOH)/(mol·L-1) | Time/h | Tensile strength/MPa | Young modulus/GPa | Elongation at break(%) | IEC*/(mmol·g-1) |
|---|---|---|---|---|---|
| 0 | 0 | 15.52±0.45 | 2.91±0.08 | 3.59±0.26 | 0.65±0.02 |
| 3 | 24 | 14.45±0.39 | 2.51±0.13 | 3.18±0.13 | 0.61±0.04 |
| 3 | 48 | 13.63±0.31 | 2.23±0.11 | 3.43±0.12 | 0.64±0.03 |
| 3 | 72 | 12.07±0.67 | 2.09±0.07 | 4.11±0.34 | 0.58±0.05 |
| 3 | 96 | 7.84±0.98 | 1.82±0.09 | 4.57±0.24 | 0.65±0.02 |
| 3 | 120 | 6.98±0.61 | 1.11±0.15 | 5.21±0.36 | 0.68±0.03 |
| 3 | 240 | 6.29±0.36 | 0.98±0.05 | 5.64±0.22 | 0.61±0.02 |
| 3 | 400 | 5.79±0.32 | 1.04±0.03 | 5.53±0.23 | 0.63±0.04 |
| 10 | 48 | 6.88±0.45 | 2.15±0.12 | 2.16±0.19 | 0.59±0.05 |
| 10 | 72 | 6.67±1.14 | 2.10±0.14 | 3.51±0.21 | 0.62±0.05 |
Table 4 Mechanical properties and IEC values of Pw-20 membranes after immersion in 3 and 10 mol/L NaOH solution at 60 ℃
| c(NaOH)/(mol·L-1) | Time/h | Tensile strength/MPa | Young modulus/GPa | Elongation at break(%) | IEC*/(mmol·g-1) |
|---|---|---|---|---|---|
| 0 | 0 | 15.52±0.45 | 2.91±0.08 | 3.59±0.26 | 0.65±0.02 |
| 3 | 24 | 14.45±0.39 | 2.51±0.13 | 3.18±0.13 | 0.61±0.04 |
| 3 | 48 | 13.63±0.31 | 2.23±0.11 | 3.43±0.12 | 0.64±0.03 |
| 3 | 72 | 12.07±0.67 | 2.09±0.07 | 4.11±0.34 | 0.58±0.05 |
| 3 | 96 | 7.84±0.98 | 1.82±0.09 | 4.57±0.24 | 0.65±0.02 |
| 3 | 120 | 6.98±0.61 | 1.11±0.15 | 5.21±0.36 | 0.68±0.03 |
| 3 | 240 | 6.29±0.36 | 0.98±0.05 | 5.64±0.22 | 0.61±0.02 |
| 3 | 400 | 5.79±0.32 | 1.04±0.03 | 5.53±0.23 | 0.63±0.04 |
| 10 | 48 | 6.88±0.45 | 2.15±0.12 | 2.16±0.19 | 0.59±0.05 |
| 10 | 72 | 6.67±1.14 | 2.10±0.14 | 3.51±0.21 | 0.62±0.05 |
| [1] | Gu S., Cai R., Yan Y., Chem. Commun., 2011, 47(10), 2856—2858 |
| [2] | Zhang F., Zhang H., Qu C., J. Mater. Chem., 2011, 21(34), 12744—12752 |
| [3] | Yan X. M., He G. H., Gu S., Wu X. M., Du L. G., Wang Y. D., Int. J. Hydrogen Energy,2012, 37(6), 5216—5224 |
| [4] | Merle G., Wessling M., Nijmeijer K., J. Membr. Sci., 2011, 377(1/2), 1—35 |
| [5] | Chempath S., Einsla B. R., Pratt L. R., J. Phys. Chem C,2008, 112(9), 3179—3182 |
| [6] | Hibbs M. R., Fujimoto C. H., Cornelius C. J., Macromolecules,2009, 42(21), 8316—8321 |
| [7] | Wu X., Scott K., J. Power Sources,2012, 214(4), 124—129 |
| [8] | Elabd Y. A., Hickner M. A., J. Macromolecules,2010, 44(1), 1—11 |
| [9] | Mamlouk M., Scott K., J. Power Sources,2012, 211(4), 140—146 |
| [10] | Tsai L. D., Chien H. C., Wang C. H., Lai C. M., Lin J. N., Zhu C. Y., Int. J. Hydrogen Energy,2013, 38(26), 11331—11339 |
| [11] | Kumar M., Singh S., Shahi V. K., J. Phys. Chem. B,2010, 114(1), 198—206 |
| [12] | Pan J., Chen C., Zhuang L., J. Acc. Chem. Res., 2012, 45, 473—481 |
| [13] | Pan J., Wang L., Zhuang L., J. Energy Environ. Sci., 2014, 7, 354—360 |
| [14] | Huang B. B., Xu X. M., Li D. H., Lan Q., Han G. B., Chem. J. Chinese Universities,2008, 29(10), 2029—2034 |
| (黄贝蓓, 徐晓明, 李东华, 蓝琴, 韩国彬. 高等学校化学学报, 2008, 29(10), 2029—2034) | |
| [15] | Menger F. M., Littau C. A., J. Am. Chem. Soc., 2002, 113(4), 1451—1452 |
| [16] | Li R. Q., Xu C. F., Wei L. B., Wang J. B., Chem. J. Chinese Universities,2010, 31(10), 2024—2029 |
| (李荣强, 徐承凤, 韦鲁滨, 王金本. 高等学校化学学报, 2010, 31(10), 2024—2029) | |
| [17] | Zana R., J. Colloid Interf. Sci., 2002, 248(2), 203—220 |
| [18] | Lu T., Huang J. B., Li Z. H., Jia S. K., Fu H. L., J. Phys. Chem. B,2008, 112, 2909 |
| [19] | Zhao J., Prog. Chem., 2015, 27(2/3), 168—173 |
| [20] | Zhao J., Prog. Chem., 2014, 26(8), 1339—1351 |
| [21] | Wang Y. Z., Sun L. Z., Song A. H., J. Shenyang Pharm. Univ., 2012, 29(4), 258—263 |
| [22] | Wang J. L., He R. H., Che Q. T., J. Colloid Interf. Sci., 2011, 361, 219—225 |
| [23] | Hibbs M. R., Hickner M. A., Alam T. M.,Mcintyre S. K., Fujimoto C. H., Cornelius C. J., Chem. Mater., 2008, 20(7), 2566—2573 |
| [24] | Han K. F., Yang Q. T., Yu S. P., Yu J. H., Zhu H., Wang Z. M., Chem. J. Chinese Universities,2013, 34(10), 2437—2444 |
| (韩克飞, 杨起涛, 于书平, 于景华, 朱红, 汪中明. 高等学校化学学报, 2013, 34(10), 2437—2444) | |
| [25] | Kalur G. C., Raghavan S. R., J. Phys. Chem. B,2005, 109(18), 8599—8604 |
| [26] | Zhang Y., Han Y., Chu Z., He S., Zhang J., Feng Y., J. Colloid Interf. Sci., 2013, 394(394), 319—328 |
| [27] | Bhadani A., Shrestha R. G., Koura S., Endo T., Sakai K., Abe M., Sakai H., Colloid Surface A,2014, 461, 258—266 |
| [28] | Li H., Hao J., J. Phys. Chem. B,2008, 112(34), 10497—10508 |
| [29] | Varcoe J. R., Slade R. C. T., Danks T. N., J. Mater. Chem., 2003, 4(4), 712—721 |
| [30] | Fan H. M., Yan Y., Li Z. C., Xu Y., Jiang L. X., Xu L. M., Zhang B., Huang J. B., J. Colloid Interf. Sci., 2010, 348(2), 491—497 |
| [31] | Lisy V., Tothova J., Zatovsky A. V., J. Chem. Phys., 2004, 121(21), 699—706 |
| [32] | Luo Y., Guo J., Wang C., Chu D., J. Power Sources,2010, 195(12), 3765—3771 |
| [33] | Poynton S. D., Kizewski J. P., Slade R. C. T., Varcoe J. R., Solid State Ionics,2010, 181(3/4), 219—222 |
| [34] | Varcoe J. R., Slade R. C. T., Simon D. P., Daniel J. D., David C., Chem. Mater., 2007, 19(10), 2686—2693 |
| [35] | Wang J., Zhao Z., Gong F., Li S., Zhang S., Macromolecules,2009, 42(22), 8711—8717 |
| [36] | Xiong Y., Fang J., Zeng Q. H., Liu Q. L., J. Membr. Sci., 2007, 311(1), 319—325 |
| [37] | Zeng Q. H., Liu Q. L., Broadwell I., Zhu A. M., Xiong Y., Tu X. P., J. Membr. Sci., 2010, 349(1/2), 237—243 |
| [38] | Stoica D., Ogier L., Akrour L., Alloin F., Fauvarque J. F., Electrochim. Acta,2007, 53(4), 1596—1603 |
| [39] | Wu Y., Wu C., Varcoe J. R., Poynton S. D., Xu T., Fu Y., J. Power. Sources,2010, 195(10), 3069—3076 |
| [40] | Vinodh R., Ilakkiya A., Elamathi S., Sangeetha D., Mat. Sci. Eng. B,2010, 167(1), 43—50 |
| [41] | Wang J., Li S., Zhang S., Macromolecules,2010, 43(8), 3890—3896 |
| [42] | Zhang Q., Li S., Zhang S., Chem. Commun., 2010, 46(40), 7495—7497 |
| [1] | HAN Yixiu, WU Dianguo, LI Hongpu, YIN Hongyao, MEI Yongjun, FENG Yujun, ZHONG Zuqin. Interactions Between Hydrophobic Associating Poly(sodium acrylate) and a Zwitterionic Surfactant in Non-aqueous Media and Low Temperature Environment [J]. Chem. J. Chinese Universities, 2021, 42(6): 2056. |
| [2] | YANG Jiarui, WANG Yifu, WANG Jilin, WANG Lulu, FENG Ruijiang. In situ Initiation, Polymerization and Construction of Cationic Active Sites of Gemini Molecules in Polysulfone Substrates† [J]. Chem. J. Chinese Universities, 2018, 39(8): 1829. |
| [3] | LONG Shijun,CHEN Mingmin,ZHAO Youjiao,WANG Xiaotao,LI Xuefeng,LIAO Yonggui. Regulation of Gemini Surfactant on Photochromic Behavior of PVA Dispersed Spiropyran Organogel Thin Film† [J]. Chem. J. Chinese Universities, 2018, 39(5): 1078. |
| [4] | GUO Zhenhao, GUI Qifeng, ZHANG Bo, REN Shuaizhen, ZHANG Shupeng, LI Xinzhong, REN Tianrui. Application of Polycarboxylate and Naphthalenesulfonate Dispersants in High Concentration Suspension Concentrate† [J]. Chem. J. Chinese Universities, 2017, 38(7): 1278. |
| [5] | LI Dan, WU Qian, LIU Li, SUN Xiaori, GUO Meng, FENG Yimin. Preparation and Stability of Gemini Surfactant Modified Gold Nanofluids† [J]. Chem. J. Chinese Universities, 2017, 38(10): 1829. |
| [6] | ZHI Zijian, ZOU Mingming, LI Shan, CHEN Jianle, YE Xingqian, CHEN Shiguo. Rheological and Structural Characterization of Pectin Polysaccharides from Citrus Pulp† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1175. |
| [7] | BU Honghong, JIAO Yanni, LAN Wenjun, ZHUANG Xiaoli, DAI Xiaonan, LI Tianduo. Rheological Properties of GuHCl/Type Ⅰ Collagen Dispersions† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1189. |
| [8] | LIU Yangjian, RUAN Yingbo, ZHANG Baoqing, QIAO Xin, LIU Chenyang. Tuning of Ionic Interaction and Rheological Properties of Nanoscale Ionic Materials† [J]. Chem. J. Chinese Universities, 2016, 37(4): 767. |
| [9] | ZHANG Huanhuan, XU Donghua, GUAN Dongbo, YAO Weiguo, SHI Tongfei. Rheological Properties of Two-component Silicon Rubber During Cross-linking by Addition Reaction† [J]. Chem. J. Chinese Universities, 2015, 36(4): 788. |
| [10] | LI Dan, CHI Hai, FANG Wen-Jun. Preparation and Thermal Stability of Oil-based Au Nanofluids [J]. Chem. J. Chinese Universities, 2013, 34(2): 414. |
| [11] | LIANG Ya-Qin, HU Zhi-Yong, CAO Duan-Lin, LIANG Dong. Surface Activity Properties of Chiral L-Lysine Based Gemini Surfactants [J]. Chem. J. Chinese Universities, 2013, 34(12): 2783. |
| [12] | FAN Hai-Ming, ZHU Xiao-Ming, YAN Yun, HUANG Jian-Bin, KANG Wan-Li. Photodimerization of Cinnamoyl Moiety in the Hydrophobic Chains of Gemini Surfactant [J]. Chem. J. Chinese Universities, 2013, 34(11): 2551. |
| [13] | LIU Mei-Tang, PU Min-Feng, MA Hong-Wen. Molecular Dynamics Simulation on Adsorbing Behavior of Anionic Gemini Surfactants at Decane/Water Interface [J]. Chem. J. Chinese Universities, 2012, 33(06): 1319. |
| [14] | XU Hai-Tao, LI Jing, LIANG Hong-Bo, XIONG Lei, GAO Ge. Synthesis and Properties of Fluorine-Containing Electron Beam Curing Polyurethane Acrylate [J]. Chem. J. Chinese Universities, 2012, 33(04): 823. |
| [15] | LÜ Shao-Yi, SHAO Zi-Qiang, ZHANG Zhen-Ling, WANG Fei-Jun, WANG Wen-Jun. Rheological Properties of New Stimuli-responsive Energetic Gels [J]. Chem. J. Chinese Universities, 2012, 33(02): 409. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||