

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (12): 2446.doi: 10.7503/cjcu20150388
• Organic Chemistry • Previous Articles Next Articles
YU Qian1, HE Yingfang1, CHEN Yuan1, YANG Wenjiang2, ZHANG Chun3, LU Jie1,*( )
)
Received:2015-05-14
Online:2015-12-10
Published:2015-11-17
Contact:
LU Jie
E-mail:ljie74@bnu.edu.cn
Supported by:TrendMD:
YU Qian, HE Yingfang, CHEN Yuan, YANG Wenjiang, ZHANG Chun, LU Jie. Preparation and Biological Evaluation of 99mTc-labeled Pteroyl-lys Derivatives†[J]. Chem. J. Chinese Universities, 2015, 36(12): 2446.
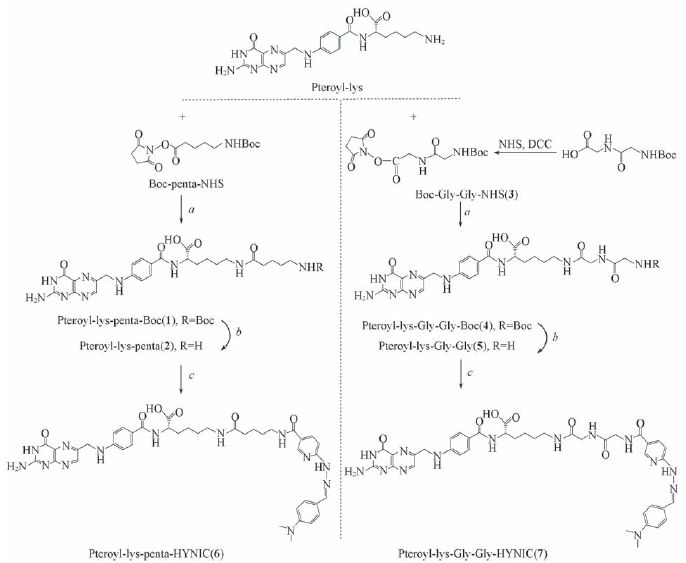
Scheme 1 Synthesis of conjugates Pteroyl-lys-penta-HYNIC(6) and Pteroyl-lys-Gly-Gly-HYNIC(7) a. DCC, DMSO/pyridine, r.t., overnight; b. TFA, 0 ℃, 8 h; c. NHS-HYNIC, DMSO/pyridine, r.t., 24 h.
| Compd. | Appearance | Yield(%) | ESI-MS,m/z[M+H]+ | HRMS,m/z[M+H]+ |
|---|---|---|---|---|
| 1 | Yellow powder | 55 | 640.9(640.3) | |
| 2 | Yellow powder | 50 | 540.5(540.3) | |
| 3 | Pale yellow powder | 70 | 329.9(329.1) | |
| 4 | Yellow powder | 49 | 655.5(655.3) | |
| 5 | Yellow powder | 51 | 555.3(555.2) | |
| 6 | Yellow powder | 54 | 806.4(806.4) | 806.3834(806.3856) |
| 7 | Yellow powder | 50 | 821.5(821.4) | 821.3597(821.3591) |
Table 1 Appearance, yields, ESI-MS and HRMS data for compounds 1—7
| Compd. | Appearance | Yield(%) | ESI-MS,m/z[M+H]+ | HRMS,m/z[M+H]+ |
|---|---|---|---|---|
| 1 | Yellow powder | 55 | 640.9(640.3) | |
| 2 | Yellow powder | 50 | 540.5(540.3) | |
| 3 | Pale yellow powder | 70 | 329.9(329.1) | |
| 4 | Yellow powder | 49 | 655.5(655.3) | |
| 5 | Yellow powder | 51 | 555.3(555.2) | |
| 6 | Yellow powder | 54 | 806.4(806.4) | 806.3834(806.3856) |
| 7 | Yellow powder | 50 | 821.5(821.4) | 821.3597(821.3591) |
| Compd. | 1H NMR(DMSO-d6, 400 MHz), δ | 13C NMR(DMSO-d6, 400 MHz), δ |
|---|---|---|
| 1 | 1.33—1.36(m, 2H), 1.39(s, 9H), 1.41—1.43(m, 2H), 1.74(s, 2H), 2.00(t, 2H), 2.54(s, 2H), 2.86—2.89(m, 4H), 2.99—3.00(m, 2H), 4.28(t, 1H), 4.48—4.49(m, 2H), 6.64(d, 2H,J=7.5 Hz), 7.66(d, 2H, J=8.6 Hz), 8.64(s, 1H) | |
| 2 | 1.38(m, 4H), 1.50(m, 4H), 1.74(m, 2H), 2.06(m, 2H), 2.76(m, 2H), 3.02(m, 2H), 4.30(m, 1H), 4.50(s, 2H), 6.64(d, 2H, J=8.3 Hz), 7.66(d, 2H, J=8.4 Hz), 8.66(s, 1H) | |
| 3* | 1.45(s, 9H), 2.85(s, 4H), 3.88(d, 2H), 4.43(d, 2H) | |
| 4 | 1.37(s, 9H), 1.74(m, 2H), 2.54(m, 4H), 3.03—3.04(m, 2H), 3.55(d, 2H), 3.64(d, 2H), 4.24—4.30(m, 1H), 4.48(d, 2H), 6.64(d, 2H, J=8.7 Hz), 7.66(d, 2H, J=8.7 Hz), 8.65(s, 1H) | |
| 5 | 1.21(m, 2H), 1.31(m, 2H), 1.64(m, 2H), 4.09(m, 5H), 4.41(m 6H), 6.58(d, 2H), 7.58(d, 2H), 8.57(s, 1H) | |
| 6 | 1.38(m, 3H), 1.49(m, 2H), 1.75(m, 2H), 1.90(m, 1H), 2.06(m, 1H), 2.50(m, 3H), 2.74(m, 1H), 2.93(m, 6H), 3.21(m, 3H), 4.29(s, 1H), 4.49(s, 2H), 6.65(d, 2H), 6.92(m, 3H), 7.49(s, 1H), 7.67(m, 3H), 8.04(m, 2H), 8.65(m, 2H) | 20.99, 22.89, 23.25, 28.75, 28.88, 30.37, 35.12, 38.18, 45.91, 52.38, 104.74, 111.17, 111.91, 120.32, 121.41, 127.46, 127.88, 128.94, 131.10, 136.76, 141.81, 148.52, 150.70, 153.81, 158.45, 161.07, 164.83, 166.37, 167.36, 171.97, 174.19 |
| 7 | 1.34(m, 4H), 1.72(m, 2H), 2.71(m, 2H), 2.93(m, 4H), 3.03(m, 2H), 3.84(m, 3H), 4.24(m, 2H), 4.46(s, 2H), 6.62(d, 2H), 6.94(m, 3H), 7.65(m, 3H), 8.04(m, 3H), 8.63(m, 2H) | 23.19, 28.67, 30.46, 38.35, 40.41, 42.06, 42.86, 45.91, 52.43, 104.76, 111.71, 177.78, 119.60, 121.47, 122.51, 127.51, 128.93, 131.07, 136.94, 142.03, 147.91, 148.51, 150.70, 151.91, 153.85, 156.21, 160.81, 165.61, 166.33, 167.34, 168.49, 169.44 |
Table 2 1H NMR and 13C NMR data for compounds 1—7
| Compd. | 1H NMR(DMSO-d6, 400 MHz), δ | 13C NMR(DMSO-d6, 400 MHz), δ |
|---|---|---|
| 1 | 1.33—1.36(m, 2H), 1.39(s, 9H), 1.41—1.43(m, 2H), 1.74(s, 2H), 2.00(t, 2H), 2.54(s, 2H), 2.86—2.89(m, 4H), 2.99—3.00(m, 2H), 4.28(t, 1H), 4.48—4.49(m, 2H), 6.64(d, 2H,J=7.5 Hz), 7.66(d, 2H, J=8.6 Hz), 8.64(s, 1H) | |
| 2 | 1.38(m, 4H), 1.50(m, 4H), 1.74(m, 2H), 2.06(m, 2H), 2.76(m, 2H), 3.02(m, 2H), 4.30(m, 1H), 4.50(s, 2H), 6.64(d, 2H, J=8.3 Hz), 7.66(d, 2H, J=8.4 Hz), 8.66(s, 1H) | |
| 3* | 1.45(s, 9H), 2.85(s, 4H), 3.88(d, 2H), 4.43(d, 2H) | |
| 4 | 1.37(s, 9H), 1.74(m, 2H), 2.54(m, 4H), 3.03—3.04(m, 2H), 3.55(d, 2H), 3.64(d, 2H), 4.24—4.30(m, 1H), 4.48(d, 2H), 6.64(d, 2H, J=8.7 Hz), 7.66(d, 2H, J=8.7 Hz), 8.65(s, 1H) | |
| 5 | 1.21(m, 2H), 1.31(m, 2H), 1.64(m, 2H), 4.09(m, 5H), 4.41(m 6H), 6.58(d, 2H), 7.58(d, 2H), 8.57(s, 1H) | |
| 6 | 1.38(m, 3H), 1.49(m, 2H), 1.75(m, 2H), 1.90(m, 1H), 2.06(m, 1H), 2.50(m, 3H), 2.74(m, 1H), 2.93(m, 6H), 3.21(m, 3H), 4.29(s, 1H), 4.49(s, 2H), 6.65(d, 2H), 6.92(m, 3H), 7.49(s, 1H), 7.67(m, 3H), 8.04(m, 2H), 8.65(m, 2H) | 20.99, 22.89, 23.25, 28.75, 28.88, 30.37, 35.12, 38.18, 45.91, 52.38, 104.74, 111.17, 111.91, 120.32, 121.41, 127.46, 127.88, 128.94, 131.10, 136.76, 141.81, 148.52, 150.70, 153.81, 158.45, 161.07, 164.83, 166.37, 167.36, 171.97, 174.19 |
| 7 | 1.34(m, 4H), 1.72(m, 2H), 2.71(m, 2H), 2.93(m, 4H), 3.03(m, 2H), 3.84(m, 3H), 4.24(m, 2H), 4.46(s, 2H), 6.62(d, 2H), 6.94(m, 3H), 7.65(m, 3H), 8.04(m, 3H), 8.63(m, 2H) | 23.19, 28.67, 30.46, 38.35, 40.41, 42.06, 42.86, 45.91, 52.43, 104.76, 111.71, 177.78, 119.60, 121.47, 122.51, 127.51, 128.93, 131.07, 136.94, 142.03, 147.91, 148.51, 150.70, 151.91, 153.85, 156.21, 160.81, 165.61, 166.33, 167.34, 168.49, 169.44 |
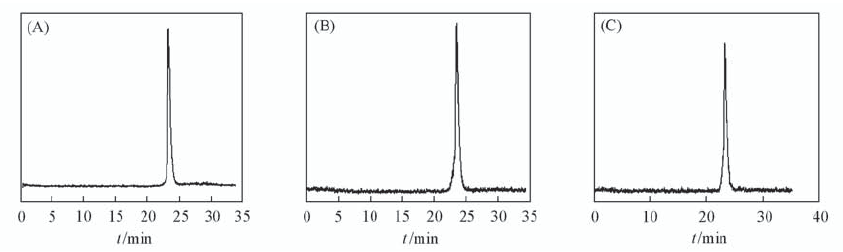
Fig.1 Radioactive HPLC profiles of 99mTc(HYNIC-penta-lys-Pteroyl)(Tricine/TPPTS) in saline at 0 h(A) and 4 h(B), and mouse plasma stability after 4 h incubation at 37 ℃(C) The retention time of 99mTc(HYNIC-penta-lys-Pteroyl)(Tricine/TPPTS) was 23.3 min.
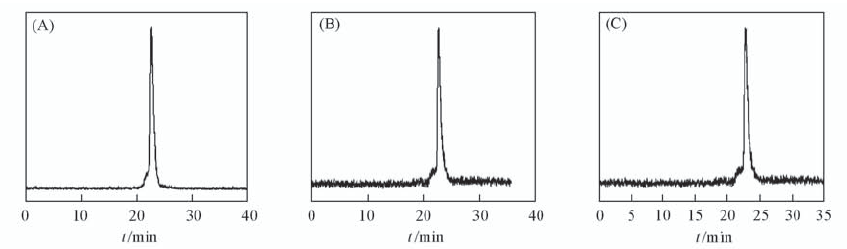
Fig.2 Radioactive HPLC profiles of 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS) in saline at 0 h(A) and 4 h(B), and mouse plasma stability after 4 h incubation at 37 ℃(C) The retention time of 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS) was 22.6 min.
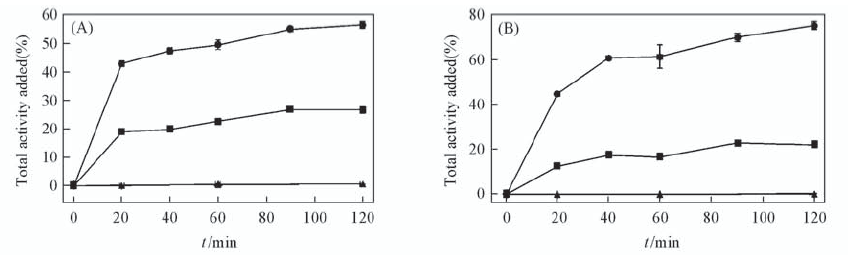
Fig.3 Cellular binding of 99mTc(HYNIC-penta-lys-Pteroyl)(Tricine/TPPTS)(A) and 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS)(B) in KB tumor cells at 37 ℃ The cell total binding fractions(●), cell internalized fractions(■) and blocked by excess folic acid(▲) were expressed in relation to the total added activity(%, of total activity added).
| Sample | Biodistribution/(%ID·g-1) | ||||
|---|---|---|---|---|---|
| 1 h | 2 h | 2 h(Blockade) | 2 h(PMX) | 4 h | |
| Heart | 1.37±0.06 | 1.79±0.29 | 0.36±0.13 | 1.22±0.15 | 1.32±0.52 |
| Liver | 2.09±0.60 | 2.00±0.64 | 0.97±0.05 | 2.10±0.26 | 1.78±0.17 |
| Spleen | 0.80±0.28 | 0.44±0.14 | 0.34±0.04 | 0.65±0.11 | 0.41±0.20 |
| Lung | 1.80±0.28 | 1.38±0.41 | 1.05±0.57 | 1.18±0.51 | 1.04±0.44 |
| Kidney | 105.91±12.73 | 112.80±7.00 | 18.52±0.69a | 44.11±6.89b | 127.93±18.34 |
| Bone | 0.60±0.21 | 0.34±0.27 | 0.65±0.88 | 1.55±0.82 | 0.36±0.24 |
| Muscle | 1.44±0.19 | 1.93±0.30 | 0.97±1.34 | 1.73±0.72 | 1.92±0.43 |
| Stomach | 1.25±0.61 | 1.40±0.50 | 1.23±0.40 | 0.58±0.09 | 1.03±0.40 |
| Intestine | 1.28±0.09 | 1.31±0.54 | 2.17±0.43 | 1.41±0.28 | 1.28±0.04 |
| Tumor | 8.55±2.78 | 8.83±0.42 | 1.64±0.49a | 10.02±2.88 | 7.99±2.28 |
| Blood | 0.83±0.12 | 0.29±0.14 | 0.73±0.19 | 0.43±0.13 | 0.15±0.05 |
| Ratio of tumor/muscle | 5.94 | 4.58 | 5.80 | 4.15 | |
| Ratio of tumor/blood | 10.26 | 30.07 | 23.14 | 54.90 | |
| Ratio of tumor/kidney | 0.08 | 0.08 | 0.23 | 0.06 | |
| Ratio of tumor/liver | 4.09 | 4.41 | 4.76 | 4.48 | |
Table 3 Biodistribution of 99mTc(HYNIC-penta-lys-Pteroyl)(Tricine/TPPTS) in KB tumor-bearing mice(n=5)
| Sample | Biodistribution/(%ID·g-1) | ||||
|---|---|---|---|---|---|
| 1 h | 2 h | 2 h(Blockade) | 2 h(PMX) | 4 h | |
| Heart | 1.37±0.06 | 1.79±0.29 | 0.36±0.13 | 1.22±0.15 | 1.32±0.52 |
| Liver | 2.09±0.60 | 2.00±0.64 | 0.97±0.05 | 2.10±0.26 | 1.78±0.17 |
| Spleen | 0.80±0.28 | 0.44±0.14 | 0.34±0.04 | 0.65±0.11 | 0.41±0.20 |
| Lung | 1.80±0.28 | 1.38±0.41 | 1.05±0.57 | 1.18±0.51 | 1.04±0.44 |
| Kidney | 105.91±12.73 | 112.80±7.00 | 18.52±0.69a | 44.11±6.89b | 127.93±18.34 |
| Bone | 0.60±0.21 | 0.34±0.27 | 0.65±0.88 | 1.55±0.82 | 0.36±0.24 |
| Muscle | 1.44±0.19 | 1.93±0.30 | 0.97±1.34 | 1.73±0.72 | 1.92±0.43 |
| Stomach | 1.25±0.61 | 1.40±0.50 | 1.23±0.40 | 0.58±0.09 | 1.03±0.40 |
| Intestine | 1.28±0.09 | 1.31±0.54 | 2.17±0.43 | 1.41±0.28 | 1.28±0.04 |
| Tumor | 8.55±2.78 | 8.83±0.42 | 1.64±0.49a | 10.02±2.88 | 7.99±2.28 |
| Blood | 0.83±0.12 | 0.29±0.14 | 0.73±0.19 | 0.43±0.13 | 0.15±0.05 |
| Ratio of tumor/muscle | 5.94 | 4.58 | 5.80 | 4.15 | |
| Ratio of tumor/blood | 10.26 | 30.07 | 23.14 | 54.90 | |
| Ratio of tumor/kidney | 0.08 | 0.08 | 0.23 | 0.06 | |
| Ratio of tumor/liver | 4.09 | 4.41 | 4.76 | 4.48 | |
| Sample | Biodistribution/(%ID·g-1) | ||||
|---|---|---|---|---|---|
| 1 h | 2 h | 2 h(Blockade) | 2 h(PMX) | 4 h | |
| Heart | 3.06±0.90 | 3.45±1.04 | 0.44±0.26 | 2.52±0.48 | 2.89±0.68 |
| Liver | 3.73±0.55 | 3.87±1.57 | 0.62±0.38 | 3.06±0.64 | 3.12±1.08 |
| Spleen | 0.63±0.28 | 0.37±0.12 | 0.47±0.22 | 0.27±0.06 | 0.21±0.05 |
| Lung | 2.60±0.44 | 2.23±0.41 | 0.82±0.27 | 1.65±0.56 | 2.01±0.45 |
| Kidney | 136.65±10.01 | 113.43±17.42 | 15.38±1.71a | 69.31±7.55b | 112.04±13.03 |
| Bone | 0.83±0.88 | 0.94±1.21 | 1.24±0.90 | 0.93±0.74 | 0.42±0.33 |
| Muscle | 2.81±0.84 | 2.40±1.35 | 1.23±0.79 | 2.64±0.15 | 2.73±0.94 |
| Stomach | 2.94±0.29 | 1.62±0.64 | 1.04±0.35 | 1.93±0.29 | 1.30±0.41 |
| Intestine | 1.79±0.38 | 1.71±0.22 | 1.42±0.17 | 1.29±0.25 | 1.06±0.62 |
| Tumor | 9.77±1.54 | 9.68±1.24 | 2.00±0.97a | 12.97±1.07 | 11.93±1.98 |
| Blood | 0.65±0.09 | 0.25±0.05 | 0.62±0.19 | 0.59±0.47 | 0.13±0.06 |
| Ratio of tumor/muscle | 3.47 | 4.04 | 4.91 | 4.38 | |
| Ratio of tumor/blood | 14.99 | 38.51 | 21.98 | 91.17 | |
| Ratio of tumor/kidney | 0.07 | 0.09 | 0.19 | 0.11 | |
| Ratio of tumor/liver | 2.62 | 2.50 | 4.24 | 3.82 | |
Table 4 Biodistribution of 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS) in KB tumor-bearing mice(n=5)
| Sample | Biodistribution/(%ID·g-1) | ||||
|---|---|---|---|---|---|
| 1 h | 2 h | 2 h(Blockade) | 2 h(PMX) | 4 h | |
| Heart | 3.06±0.90 | 3.45±1.04 | 0.44±0.26 | 2.52±0.48 | 2.89±0.68 |
| Liver | 3.73±0.55 | 3.87±1.57 | 0.62±0.38 | 3.06±0.64 | 3.12±1.08 |
| Spleen | 0.63±0.28 | 0.37±0.12 | 0.47±0.22 | 0.27±0.06 | 0.21±0.05 |
| Lung | 2.60±0.44 | 2.23±0.41 | 0.82±0.27 | 1.65±0.56 | 2.01±0.45 |
| Kidney | 136.65±10.01 | 113.43±17.42 | 15.38±1.71a | 69.31±7.55b | 112.04±13.03 |
| Bone | 0.83±0.88 | 0.94±1.21 | 1.24±0.90 | 0.93±0.74 | 0.42±0.33 |
| Muscle | 2.81±0.84 | 2.40±1.35 | 1.23±0.79 | 2.64±0.15 | 2.73±0.94 |
| Stomach | 2.94±0.29 | 1.62±0.64 | 1.04±0.35 | 1.93±0.29 | 1.30±0.41 |
| Intestine | 1.79±0.38 | 1.71±0.22 | 1.42±0.17 | 1.29±0.25 | 1.06±0.62 |
| Tumor | 9.77±1.54 | 9.68±1.24 | 2.00±0.97a | 12.97±1.07 | 11.93±1.98 |
| Blood | 0.65±0.09 | 0.25±0.05 | 0.62±0.19 | 0.59±0.47 | 0.13±0.06 |
| Ratio of tumor/muscle | 3.47 | 4.04 | 4.91 | 4.38 | |
| Ratio of tumor/blood | 14.99 | 38.51 | 21.98 | 91.17 | |
| Ratio of tumor/kidney | 0.07 | 0.09 | 0.19 | 0.11 | |
| Ratio of tumor/liver | 2.62 | 2.50 | 4.24 | 3.82 | |
| Complex | Biodistribution/(%ID·g-1) | |||
|---|---|---|---|---|
| Tumor | Tumor/Muscle | Tumor/Blood | Tumor/Liver | |
| 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS) | 11.93±1.98 | 4.38 | 91.17 | 3.82 |
| 99mTc-EC20 | 12.26±1.05 | — | 59.56 | 3.40 |
| 111In-DTPA-folate(γ) | 6.8± 1.2 | 2.5 | 106 | 3.3 |
Table 5 Comparison of biodistribution data between 99mTc(HYNIC-Gly-Gly-lys-Pteroyl) and other FR imaging agents in KB tumor-bearing mice at 4 h post-injection
| Complex | Biodistribution/(%ID·g-1) | |||
|---|---|---|---|---|
| Tumor | Tumor/Muscle | Tumor/Blood | Tumor/Liver | |
| 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS) | 11.93±1.98 | 4.38 | 91.17 | 3.82 |
| 99mTc-EC20 | 12.26±1.05 | — | 59.56 | 3.40 |
| 111In-DTPA-folate(γ) | 6.8± 1.2 | 2.5 | 106 | 3.3 |
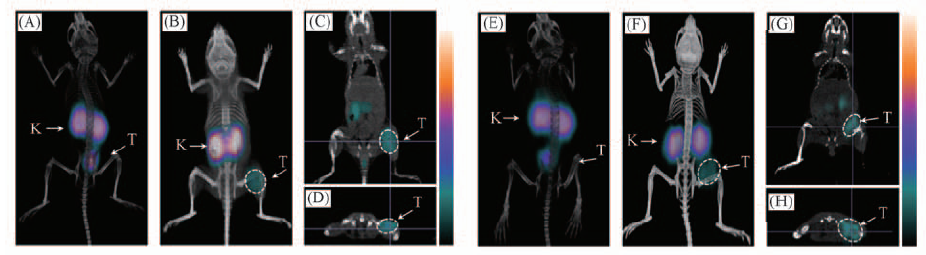
Fig.4 Whole-body, coronal and transaxial images 99mTc(HYNIC-penta-lys-Pteroyl)(Tricine/TPPTS)(A―D) and 99mTc(HYNIC-Gly-Gly-lys-Pteroyl)(Tricine/TPPTS)(E―H) in the athymic nude mice bearing KB tumors(female, 18—20 g) at 120 min p.i.; tumors(T) and kidneys(K) were highlighted with arrows on the images. The uptakes of radiotracer in FR-positive tumors were significantly blocked by co-injection of excess folic acid(A and E).
| [1] | Elnakat H., Ratnam M., Adv. Drug Deliv. Rev., 2004, 56, 1067—1084 |
| [2] | Ross J. F., Chaudhuri P. K., Ratnam M., Cancer, 1994, 73, 2432—2443 |
| [3] | Weitman S. D., Lark R. H., Coney L. R., Fort D. W., Frasca V., Zurawski V. R., Kamen B. A., Cancer Res., 1992, 52, 3396—3401 |
| [4] | Armstrong D. K., White A. J., Weil S. C., Phillips M., Coleman R. L., Gynecol. Oncol., 2013, 129, 452—458 |
| [5] | Naumann R. W., Coleman R. L., Burger R. A., Sausville E. A., Kutarska E., Ghamande S. A., Gabrail N. Y., DePasquale S. E., Nowara E., Gilbert L., Gersh R. H., Teneriello M. G., Harb W. A., Konstantinopoulos P. A., Penson R. T., Symanowski J. T., Lovejoy C. D., Leaman C. P., Morgenstern D. E., Messmann R. A., J. Clin. Oncol., 2013, 31, 4400—4406 |
| [6] | Low P. S., Henne W. A., Doorneweerd D. D., Acc Chem. Res., 2008, 41, 120—129 |
| [7] | Ke C.Y., Mathias C. J., Green M. A., Adv. Drug Delivery Rev., 2004, 56, 1143—1160 |
| [8] | Müller C., Schibli R., J. Nucl. Med., 2011, 52, 1—4 |
| [9] | Maurer A. H., Elsinga P., Fanti S., Nguyen B., Oyen W. J. G., Weber W. A., J. Nucl. Med., 2014, 55, 701—704 |
| [10] | Müller C., Molecules, 2013, 18, 5005—5031 |
| [11] | Siegel B. A., Dehdashti F., Mutch D. G., Podoloff D. A., Wendt R., Sutton G. P., Burt R. W., Ellis P. R., Mathias C. J., Green M. A., Gershenson D. M., J. Nucl. Med., 2003, 44, 700—707 |
| [12] | Leamon C. P., Parker M. A., Vlahov I. R., Xu L. C., Reddy J. A., Vetzel M., Douglas N., Bioconjugate Chem., 2002, 13, 1200—1210 |
| [13] | Lu J., Pang Y., Xie F., Guo H. J., Li Y., Yang Z., Wang X. B., Nucl. Med. Biol., 2011, 38, 557—565 |
| [14] | Guo H.J., Xie F., Zhu M. L., Li Y., Yang Z., Wang X. B., Lu J., Bioorg. Med. Chem. Lett., 2011, 21, 2025—2029 |
| [15] | Chen Y., Guo H. J., Xie F., Lu J., J. Labelled Comp. Radiopharm., 2014, 57, 12—17 |
| [16] | Yan M. D., Cai S. X., Wybourne M. N., Keana J. F. W., Bioconjugate Chem., 1994, 5, 151—157 |
| [17] | Plumb K., Kraatz H. B., Bioconjugate Chem., 2003, 14, 601—606 |
| [18] | Xie F., Zhang C., Yu Q., Pang Y., Chen Y., Yang W., Xue J., Liu Y., Lu J., RSC Adv., 2014, 4, 32197—32206 |
| [19] | Liang K., Chu T. W., Chem. J. Chinese Universities,2013, 34(4), 870—874 |
| (梁坤, 褚泰伟. 高等学校化学学报,2013, 34(4), 870—874) | |
| [20] | Müller C., Reddy J. A., Leamon C. P., Schibli R., Mol. Pharm., 2010, 7, 597—604 |
| [21] | Ke C. Y., Mathias C. J., Green M. A., J. Am. Chem. Soc., 2005, 127, 7421—7426 |
| [1] | Lü Feng, LI Yan-Zhou, WU Li, LIU Tian-Jun*. Galactose Substituted Phthalocyanines for Tumor Imaging as Near-infrared Fluorescence Probe [J]. Chem. J. Chinese Universities, 2011, 32(5): 1010. |
| [2] | ZHANG Ya-Dong, PANG Yan, LU Jie*, WEI Xiao-Yan, YANG Wen-Jiang, ZHU Mei-Lin . Preparation, Labeling and Biological Evaluation of Human Serum Albumin-folate Conjugate for Targeting Folate Receptor [J]. Chem. J. Chinese Universities, 2010, 31(10): 1999. |
| [3] | ZHANG Hui-Zhu*, ZHANG Qi-Qing. Preparation of Folate Targeted Pullulan Acetate Nanoparticles and Cell Uptake in vitro [J]. Chem. J. Chinese Universities, 2009, 30(6): 1146. |
| [4] | ZHANG Xian-Zhong, TIAN Yan-Cong, WANG Xue-Bin, LUO Qian . Uptake of 99mTcN-MIBI in Tumor Cells in vitro and the Comparison of it with 99mTc-MIBI [J]. Chem. J. Chinese Universities, 2003, 24(10): 1742. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||