

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (4): 665.doi: 10.7503/cjcu20140991
• Organic Chemistry • Previous Articles Next Articles
LI Hongyu*( ), WANG Bo, LIU Shuming
), WANG Bo, LIU Shuming
Received:2014-11-10
Online:2015-04-10
Published:2015-03-18
Contact:
LI Hongyu
E-mail:lihy2006@126.com
Supported by:CLC Number:
TrendMD:
LI Hongyu, WANG Bo, LIU Shuming. Synthesis and Characterization of Task-specific Ionic Liquids with Alkyl Phosphate Cations and for U(Ⅵ) Extraction†[J]. Chem. J. Chinese Universities, 2015, 36(4): 665.
| Compd. | 1H NMR(300 MHz), δ | 13C NMR(75 MHz), δ |
|---|---|---|
| 2a | 8.63(s, 1H), 7.38(s, 1H), 7.33(s, 1H), 4.21—4.24(t, J=6.9 Hz, 2H), 3.96—3.98(m, 6H), 3.75(s, 3H), 2.13—2.16(m, 2H), 1.51—1.54(m, 4H), 1.24—1.25(m, 4H), 0.75—0.79(t, J=6.9 Hz, 6H) | 12.76, 18.02, 25.65, 31.22, 36.35, 48.78, 61.34, 65.89, 121.76, 122.12, 136.23 |
| 3a | 8.82—8.83(d, J=8.5 Hz, 2H), 8.49—8.52(t, J=1H), 8.01—8.04(t, J=8.5 Hz, 2H), 4.70—4.73(t, J=6.9 Hz, 2H), 4.13—4.15(m, 2H), 3.95—4.03(m, 4H), 2.35—2.41(m, 2H), 1.52—1.59(m, 4H), 1.29—1.33(m, 4H), 0.80—0.83(t, J=6.9 Hz, 6H) | 12.21, 18.60, 26.34, 31. 56, 53.13, 61,76, 65.87, 129, 11, 146.01, 147.31 |
| 4b | 8.57(s, 1H), 7.28(s, 1H), 7.22(s, 1H), 4.24—4.27(t, J=6.9 Hz, 2H), 3.96—4.00(m, 6H), 3.83(s, 3H), 2.10—2.20(m, 2H), 1.56—1.63(m, 4H), 1.29—1.38(m, 4H), 0.85—0.88(t, J=6.9 Hz, 6H) | 13.23, 17.81, 27.96, 31.12, 37.21, 54.62, 62.58, 65.72, 127.15, 132.18, 147.20 |
| 5b | 8.85(s, 1H), 7.44(s, 1H), 7.21(s, 1H), 4.29—4.32(m, 2H), 3.96—4.01(m, 6H), 3.88(s, 3H), 2.18—2.19(m, 2H), 1.58—1.61(m, 4H), 1.31—1.36(m, 4H), 0.87—0.89(t, J=6.9 Hz, 6H) | 13.43, 17.92, 28.20, 31.36, 37.45, 54.87, 62.94, 66.05, 127.31,132.24, 147.45, 159.47 |
| 6b | 8.89—8.90(d, J=8.5 Hz, 2H), 8.41—8.44(t, J=8.5 Hz, 1H), 7.96—8.00(t, J=8.5 Hz, 2H), 4.71—4.75(t, J=6.9 Hz, 2H), 4.01—4.03(m, 2H), 3.94—3.99(m, 4H), 2.35—2.39(m, 2H), 1.55—1.62(m, 4H), 1.28—1.37(m, 4H), 0.85—0.88(t, J=6.9 Hz, 6H) | 12.68, 17,79, 26.22, 30.96, 52.66, 61.74, 65.83, 127.67, 145.68, 146.00, 152.89 |
Table 1 1H NMR, 13C NMR data for compounds 2—6
| Compd. | 1H NMR(300 MHz), δ | 13C NMR(75 MHz), δ |
|---|---|---|
| 2a | 8.63(s, 1H), 7.38(s, 1H), 7.33(s, 1H), 4.21—4.24(t, J=6.9 Hz, 2H), 3.96—3.98(m, 6H), 3.75(s, 3H), 2.13—2.16(m, 2H), 1.51—1.54(m, 4H), 1.24—1.25(m, 4H), 0.75—0.79(t, J=6.9 Hz, 6H) | 12.76, 18.02, 25.65, 31.22, 36.35, 48.78, 61.34, 65.89, 121.76, 122.12, 136.23 |
| 3a | 8.82—8.83(d, J=8.5 Hz, 2H), 8.49—8.52(t, J=1H), 8.01—8.04(t, J=8.5 Hz, 2H), 4.70—4.73(t, J=6.9 Hz, 2H), 4.13—4.15(m, 2H), 3.95—4.03(m, 4H), 2.35—2.41(m, 2H), 1.52—1.59(m, 4H), 1.29—1.33(m, 4H), 0.80—0.83(t, J=6.9 Hz, 6H) | 12.21, 18.60, 26.34, 31. 56, 53.13, 61,76, 65.87, 129, 11, 146.01, 147.31 |
| 4b | 8.57(s, 1H), 7.28(s, 1H), 7.22(s, 1H), 4.24—4.27(t, J=6.9 Hz, 2H), 3.96—4.00(m, 6H), 3.83(s, 3H), 2.10—2.20(m, 2H), 1.56—1.63(m, 4H), 1.29—1.38(m, 4H), 0.85—0.88(t, J=6.9 Hz, 6H) | 13.23, 17.81, 27.96, 31.12, 37.21, 54.62, 62.58, 65.72, 127.15, 132.18, 147.20 |
| 5b | 8.85(s, 1H), 7.44(s, 1H), 7.21(s, 1H), 4.29—4.32(m, 2H), 3.96—4.01(m, 6H), 3.88(s, 3H), 2.18—2.19(m, 2H), 1.58—1.61(m, 4H), 1.31—1.36(m, 4H), 0.87—0.89(t, J=6.9 Hz, 6H) | 13.43, 17.92, 28.20, 31.36, 37.45, 54.87, 62.94, 66.05, 127.31,132.24, 147.45, 159.47 |
| 6b | 8.89—8.90(d, J=8.5 Hz, 2H), 8.41—8.44(t, J=8.5 Hz, 1H), 7.96—8.00(t, J=8.5 Hz, 2H), 4.71—4.75(t, J=6.9 Hz, 2H), 4.01—4.03(m, 2H), 3.94—3.99(m, 4H), 2.35—2.39(m, 2H), 1.55—1.62(m, 4H), 1.28—1.37(m, 4H), 0.85—0.88(t, J=6.9 Hz, 6H) | 12.68, 17,79, 26.22, 30.96, 52.66, 61.74, 65.83, 127.67, 145.68, 146.00, 152.89 |
| Compd. | FTIR(KBr), | Elemental analysis(calcd., %) | ||
|---|---|---|---|---|
| C | H | N | ||
| 2 | 3142, 3061, 2960, 2873, 1736, 1572, 1465, 1373, 1267, | 41.44(43.59) | 7.34(7.32) | 7.43(6.78) |
| 1170, 1028, 906, 862, 756, 621 | ||||
| 3 | 3035, 2961, 2874, 1737, 1634, 1488, 1372, 1265, 1170, | 46.12(47.07) | 7.32(7.17) | 3.78(3.43) |
| 1028, 868, 776, 684 | ||||
| 4 | 3168, 3122, 2963, 2875, 1577, 1466, 1263, 1171, 1029, | 37.48(37.66) | 5.06(6.32) | 5.80(5.86) |
| 840 | ||||
| 5 | 3157, 3119, 2965, 2877, 1576, 1467, 1431, 1353, 1264, | 33.32(33.28) | 5.01(4.93) | 6.69(6.85) |
| 1196, 1138, 1058, 1031, 803, 789, 740, 653, 617, 570 | ||||
| 6 | 3138, 3071, 2964, 2877, 1637, 1491, 1468, 1353, 1266, | 35.03(35.41) | 4.96(4.79) | 4.37(4.59) |
| 1194, 1137, 1058, 1030, 788, 740, 684, 617, 570 | ||||
Table 2 FTIR and elemental analysis data for compounds 2—6
| Compd. | FTIR(KBr), | Elemental analysis(calcd., %) | ||
|---|---|---|---|---|
| C | H | N | ||
| 2 | 3142, 3061, 2960, 2873, 1736, 1572, 1465, 1373, 1267, | 41.44(43.59) | 7.34(7.32) | 7.43(6.78) |
| 1170, 1028, 906, 862, 756, 621 | ||||
| 3 | 3035, 2961, 2874, 1737, 1634, 1488, 1372, 1265, 1170, | 46.12(47.07) | 7.32(7.17) | 3.78(3.43) |
| 1028, 868, 776, 684 | ||||
| 4 | 3168, 3122, 2963, 2875, 1577, 1466, 1263, 1171, 1029, | 37.48(37.66) | 5.06(6.32) | 5.80(5.86) |
| 840 | ||||
| 5 | 3157, 3119, 2965, 2877, 1576, 1467, 1431, 1353, 1264, | 33.32(33.28) | 5.01(4.93) | 6.69(6.85) |
| 1196, 1138, 1058, 1031, 803, 789, 740, 653, 617, 570 | ||||
| 6 | 3138, 3071, 2964, 2877, 1637, 1491, 1468, 1353, 1266, | 35.03(35.41) | 4.96(4.79) | 4.37(4.59) |
| 1194, 1137, 1058, 1030, 788, 740, 684, 617, 570 | ||||
| Compd. | Td/℃ | Tmax/℃ | Mass loss(%) |
|---|---|---|---|
| [Phos-C3-MIM][PF6](4) | 260, 330 | 510 | 80 |
| [Phos-C3-MIM][NTf2](5) | 230, 300, 420 | 470 | 80 |
| [Phos-C3-Pyr][NTf2](6) | 210, 280, 310 | 510 | 80 |
Table 3 TGA data of ionic liquids*
| Compd. | Td/℃ | Tmax/℃ | Mass loss(%) |
|---|---|---|---|
| [Phos-C3-MIM][PF6](4) | 260, 330 | 510 | 80 |
| [Phos-C3-MIM][NTf2](5) | 230, 300, 420 | 470 | 80 |
| [Phos-C3-Pyr][NTf2](6) | 210, 280, 310 | 510 | 80 |
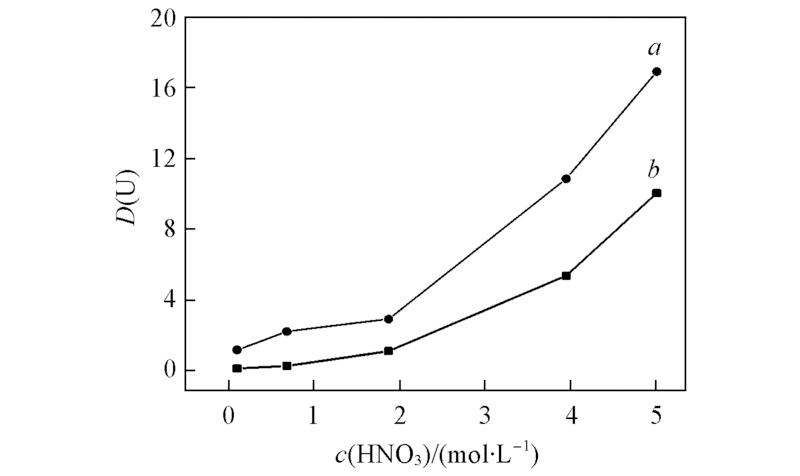
Fig.6 Distribution ratio D(U) with equilibrium concentration of nitric acid for the extraction of U(Ⅵ) in different concentrations of nitric acid a. 1.1 mol/L TBP-compound 4; b. compound 4;c(U)=58 mg/L.
| [1] | Welton T., Chem. Rev., 1999, 99, 2071—2083 |
| [2] | Shen X. H., Xu C., Liu X. Q., Chu T. W., Journal of Nuclear and Radiochemistry,2006, 28(3), 129—138 |
| (沈兴海, 徐超, 刘新起, 褚泰伟. 核化学与放射化学, 2006, 28(3), 129—138) | |
| [3] | Li H. Y., Liu Z. P., Uranium Mining and Metallurgy,2008, 27(2), 86—91 |
| (李宏宇, 刘正平. 铀矿冶, 2008, 27(2), 86—91) | |
| [4] | Zhang H., Tang B. K., Row K., Chem. Res. Chinese Universities,2014, 31(1), 37—41 |
| [5] | Feng X. D., Liang F. H., Su R., Wu L. J., Li X. Y., Wang X. H., Zhang H. Q., Yu A. M., Chem. Res. Chinese Universities,2013, 29(4), 647—652 |
| [6] | Giridhal P., Venkatesan K. P., Srinivasan T. G., J. Radioanal. Nucl. Chem., 2004, 5(1), 21—26 |
| [7] | Chu T. W., Qin L, Liu X. Q., J. Nucl. Radiochem., 2007, 29(3), 146—150 |
| (褚泰伟, 秦丽, 刘新起. 核化学与放射化学, 2007, 29(3), 146—150) | |
| [8] | Giridhar P., Venkatesan K. A., Srinivasan T. G., Vasudeva Rao P. R., J. Radioanal. Nucl. Chem., 2005, 265(1), 31—38 |
| [9] | Dietz M. L., Stepinski D. C., Talanta,2008, 75(2), 598—603 |
| [10] | Dietz M. L., Dzielawa J. A., Laszak I., Young B. A., Jensen M. P., Green Chem., 2003, 5, 682—685 |
| [11] | Stepinski D. C., Jensen M. P., Dzielawa J. A., Dietz M. L., Green Chem., 2005, 7, 151—158 |
| [12] | Shen Y., Tan X., Wang L., Wu W., Sep. Purif. Technol., 2011, 78(3), 298—302 |
| [13] | Turanov A. N., Karandashev V. K., Yarkevich A. N., Radiochemistry,2013, 55(4), 382—387 |
| [14] | Sengupta A., Murali M. S., Mohapatra P. K., J. Radioanal. Nucl. Chem., 2013, 298(1), 209—217 |
| [15] | Nockemann P., Thijs B., Pittois S., Thoen J., Glorieux C., Hecks K. V., Meervelt L. V., Kirchner B., Binnemans K., J. Phys. Chem. B,2006, 110(42), 20978—20992 |
| [16] | Gao L. X., Yu J., Chem. J. Chinese Universities,2013, 34(1), 108—114 |
| (高丽霞, 余江. 高等学校化学学报, 2013, 34(1), 108—104) | |
| [17] | Huddleston J. G., Visser A. G., Reichert W. M., Willauer H. D., Broker G. A., Rogers R. D., Green Chem., 2001, 3, 156—164 |
| [18] | Visser A., Swatsolty R., Matthewreicher W., Mayton R., Sheff S., Wierzbicki A., Davis J. H., Rogers R. D., Environ. Sci. Technol., 2002, 36(11), 2523—2529 |
| [19] | Rout A., Venkatesan K. A., Srinivasan T. G., Vasudeva Rao P. R., J. Hazardous. Mater., 2012, 221/222, 62—67 |
| [20] | Biswas S., Rupawate V. H., Roy S. B., Sahu M., J. Radioanal. Nucl. Chem., 2014, 300(2), 853—858 |
| [21] | Ilia G., Popa A., Iliescu S., Bora A., Dehelean G., Pascariu A., Phosphorus Sulfur, and Silicon and the Related Elements,2003, 178(7), 1513—1519 |
| [22] | Huddleston J.G., Rogers R. D.,Chem. Commun., 1998, 1765—1766 |
| [1] | ZHANG Yongpo, SUN Huailin. Phenoxyacetylcobalt Tetracarbonyl as Catalyst for Copolymerization of Imines and CO to Produce Polypeptides† [J]. Chem. J. Chinese Universities, 2014, 35(1): 54. |
| [2] | LI Zhi-Zhang1, GU Zheng2, ZHANG Rong2, WU Yun-Dong2, WANG Xiao-Yong1, JIANG Hai-Ming1, XIANG Jian-Nan2*. New Method for the Preparation of Acetylenic Retinoids [J]. Chem. J. Chinese Universities, 2009, 30(3): 502. |
| [3] | LIU Jian, LIU Yu, LUO Cui-Ping, LIU En-Hui, YANG Yu-Ping, GAN Quan, ZHU Mei-Xiang, ZHU Wei-Guo. Synthesis of Cyclometalated Platinum Complexes Containing Triarylamino Group and Their Photophysical and Electrochemical Properties [J]. Chem. J. Chinese Universities, 2006, 27(10): 1873. |
| [4] | CHAI Jian-Fang, TANG Liang-Fu, JIA Wen-Li, WANG Zhi-Hong, WANG Ji-Tao . Studies on the Substituent Effect and Electrochemical Behavior of ⅥB Metal Carbonyl Complexes Containing Bis(pyrazol-1-yl)alkanes [J]. Chem. J. Chinese Universities, 2001, 22(6): 943. |
| [5] | SONG Xin-Yi, CHEN Ying, CHEN Hui-Lan. Spin Trapping EPR Investigation on Rearrangement Reactions of Radicals Generated by Cleavage of Co——C Bonds of Three Aqua(butyl)cobaloximes Under Photolysis [J]. Chem. J. Chinese Universities, 2000, 21(2): 169. |
| [6] | LIU Wan-Yi, YUAN Yao-Feng, ZHANG Ling-Yun, WANG Ji-Tao. The Relationship Between the Bridging Alkyl Group and Electrochemical Behavior of Some Biferrocenyl Alkanes [J]. Chem. J. Chinese Universities, 1998, 19(8): 1251. |
| [7] | YAN Peng-Fei, GAO Jin-Sheng, LE Zheng-Yu, MAO Gui-jie . Studies on the Formation and Reactivity of(Na[Fe(CO)2C5H5]2)n [J]. Chem. J. Chinese Universities, 1998, 19(1): 21. |
| [8] | XIANG Ping, LUO Lai-Bin, CHEN Ying, CHEN Hut-Lan. Studies on Synthesis and Properties of Supermolecular Compound Involving Cobaltoximes with a Long Chain Alkyl and β-CD [J]. Chem. J. Chinese Universities, 1997, 18(11): 1753. |
| [9] | DU Hong-Guang, SHI Xin-Xu, YE Ji-Dong, SHI Shu-Jian. Synthesis of Organosilylferrocene and 2-(Tributyltin)-ferrocene Derivatives [J]. Chem. J. Chinese Universities, 1997, 18(10): 1642. |
| [10] | SONG Li-Cheng, YAN Chao-Guo, MAO Xi-An. Studies on 1H and 13C NMR Spectra of Ditertiary Phosphine Bis-substituted Derivatives of Butterfly Fe/SeCluster Complex (μ-p-MeC6H4Se)2Fe2 (CO)6 [J]. Chem. J. Chinese Universities, 1997, 18(7): 1093. |
| [11] | GAO Li-Xia, YU Jiang. Synthesis and Characterization of Task-specific Schiff-base Ionic Liquid [J]. Chem. J. Chinese Universities, 2013, 34(1): 108. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||