

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (2): 292.doi: 10.7503/cjcu20130465
• Organic Chemistry • Previous Articles Next Articles
ZHANG Yongpo, ZHANG Yang, SUN Huailin*( )
)
Received:2013-05-16
Online:2014-02-10
Published:2014-01-03
Contact:
SUN Huailin
E-mail:sunhl@nankai.edu.cn
Supported by:CLC Number:
TrendMD:
ZHANG Yongpo, ZHANG Yang, SUN Huailin. Stereo Selective Synthesis and Absolute Configuration Determination of (2E,4S)-4-(t-butoxycarbonylamino)-5-[(3S)-2'-oxo-3'-pyrrolidinyl]-2-pentenoic Acid Ethyl Ester†[J]. Chem. J. Chinese Universities, 2014, 35(2): 292.
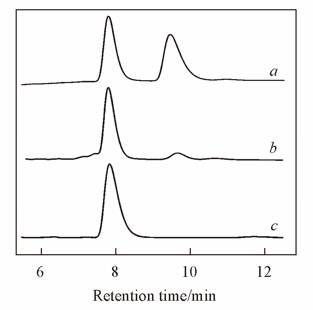
Fig.2 Comparison of chiral HPLC analyses results of compound 4 and the racemic samplea. Racemic sample; b. compound 4 obtained in refluxing THF; c. optically pure compound 4 obtained at room temperature.
| Empirical formula | C12H22N2O4 | F(000) | 560 |
|---|---|---|---|
| Formula weight | 258.32 | θ range | 5.82°—72.56° |
| Temperature/K | 113(2) | Limiting indices | -9≤h≤9, -10≤k≤9, -26≤l≤25 |
| Wavelength/nm | 0.154187 | Reflections collected/unique | 16207/2747[R(int)=0.0432] |
| Crystal system | Orthorhombic | Completeness to θ=72.56(%) | 98.9 |
| Space group | P212121 | Absorption correction | Semi-empirical from equivalents |
| a/nm | 0.80190(10) | Max. and min. transmission | 0.8768 and 0.8523 |
| b/nm | 0.81220(10) | Refinement method | Full-matrix least-squares on F2 |
| c/nm | 2.1735(3) | Data/restraints/parameters | 2747/0/179 |
| α/(°) | 90 | Goodness-of-fit on F2 | 1.089 |
| β/(°) | 90 | Final R indices[I>2σ(I)] | R1=0.0293, wR2=0.0769 |
| γ/(°) | 90 | R indices(all data) | R1=0.0303, wR2=0.0779 |
| Volume/nm3, Z | 1.4156(3), 4 | Absolute structure parameter | 0.19(15) |
| Calculated density/(g·cm-3) | 1.212 | Extinction coefficient | 0.0057(8) |
| Absorption coefficient/mm-1 | 0.750 | Largest diff. peak and hole/(e·nm-3) | 154 and -192 |
Table 1 X-Ray diffraction data of compound 5
| Empirical formula | C12H22N2O4 | F(000) | 560 |
|---|---|---|---|
| Formula weight | 258.32 | θ range | 5.82°—72.56° |
| Temperature/K | 113(2) | Limiting indices | -9≤h≤9, -10≤k≤9, -26≤l≤25 |
| Wavelength/nm | 0.154187 | Reflections collected/unique | 16207/2747[R(int)=0.0432] |
| Crystal system | Orthorhombic | Completeness to θ=72.56(%) | 98.9 |
| Space group | P212121 | Absorption correction | Semi-empirical from equivalents |
| a/nm | 0.80190(10) | Max. and min. transmission | 0.8768 and 0.8523 |
| b/nm | 0.81220(10) | Refinement method | Full-matrix least-squares on F2 |
| c/nm | 2.1735(3) | Data/restraints/parameters | 2747/0/179 |
| α/(°) | 90 | Goodness-of-fit on F2 | 1.089 |
| β/(°) | 90 | Final R indices[I>2σ(I)] | R1=0.0293, wR2=0.0769 |
| γ/(°) | 90 | R indices(all data) | R1=0.0303, wR2=0.0779 |
| Volume/nm3, Z | 1.4156(3), 4 | Absolute structure parameter | 0.19(15) |
| Calculated density/(g·cm-3) | 1.212 | Extinction coefficient | 0.0057(8) |
| Absorption coefficient/mm-1 | 0.750 | Largest diff. peak and hole/(e·nm-3) | 154 and -192 |
| O1—C1 | 0.12349(15) | O4—C8 | 0.13519(13) | N2—C8 | 0.13423(14) |
|---|---|---|---|---|---|
| O2—C7 | 0.14192(16) | O4—C9 | 0.14592(13) | N2—C6 | 0.14547(14) |
| O2—H2A | 0.085(2) | N1—C1 | 0.13377(16) | N2—H2B | 0.0898(16) |
| O3—C8 | 0.12269(13) | N1—C4 | 0.14600(16) | C1—C2 | 0.15205(17) |
| C1—N1—C4 | 11.358(11) | C1—C2—C3 | 10.300(10) | N2—C6—C5 | 11.042(9) |
| C8—N2—C6 | 12.351(9) | C5—C2—C3 | 11.559(10) | C7—C6—C5 | 11.038(9) |
| O1—C1—N1 | 12.492(12) | C2—C3—C4 | 10.419(10) | O2—C7—C6 | 11.350(11) |
| O1—C1—C2 | 12.610(11) | N1—C4—C3 | 10.244(10) | O3—C8—N2 | 12.589(10) |
| N1—C1—C2 | 10.896(10) | C2—C5—C6 | 11.257(9) | O3—C8—O4 | 12.498(10) |
| C1—C2—C5 | 11.315(10) | N2—C6—C7 | 10.907(10) | N2—C8—O4 | 10.913(9) |
Table 2 Selected bond lengths(nm) and bond angles(°) of compound 5
| O1—C1 | 0.12349(15) | O4—C8 | 0.13519(13) | N2—C8 | 0.13423(14) |
|---|---|---|---|---|---|
| O2—C7 | 0.14192(16) | O4—C9 | 0.14592(13) | N2—C6 | 0.14547(14) |
| O2—H2A | 0.085(2) | N1—C1 | 0.13377(16) | N2—H2B | 0.0898(16) |
| O3—C8 | 0.12269(13) | N1—C4 | 0.14600(16) | C1—C2 | 0.15205(17) |
| C1—N1—C4 | 11.358(11) | C1—C2—C3 | 10.300(10) | N2—C6—C5 | 11.042(9) |
| C8—N2—C6 | 12.351(9) | C5—C2—C3 | 11.559(10) | C7—C6—C5 | 11.038(9) |
| O1—C1—N1 | 12.492(12) | C2—C3—C4 | 10.419(10) | O2—C7—C6 | 11.350(11) |
| O1—C1—C2 | 12.610(11) | N1—C4—C3 | 10.244(10) | O3—C8—N2 | 12.589(10) |
| N1—C1—C2 | 10.896(10) | C2—C5—C6 | 11.257(9) | O3—C8—O4 | 12.498(10) |
| C1—C2—C5 | 11.315(10) | N2—C6—C7 | 10.907(10) | N2—C8—O4 | 10.913(9) |
| [1] | Tian Q., Nayyar N. K., Babu S., Chen L., Tao J., Lee S., Tibbetts A., Moran T., Liou J., Guo M., Tetrahedron Lett., 2001, 42, 6807—6809 |
| [2] | Dragovich P. S., Prins T. J., Zhou R., Webber S. E., Marakovits J. T., Fuhrman S. A., Patick A. K., Matthews D. A., Lee C. A., Ford C. E., J. Med. Chem., 1999, 42, 1213—1224 |
| [3] | Dragovich P. S., Prins T. J., Zhou R., Brown E. L., Maldonado F. C., Fuhrman S. A., Zalman L. S., Tuntland T., Lee C. A., Patick A. K., Matthews D. A., Hendrickson T. F., Kosa M. B., Liu B., Batugo M. R., Gleeson J. P. R., Sakata S. K., Chen L., Guzman M. C., Meador J. W., Ferre R. A., Worland S. T., J. Med. Chem., 2002, 45, 1607—1623 |
| [4] | Dragovich P. S., Prins T. J., Zhou R., Johnson T. O., Hua Y., Luu H. T., Sakata S. K., Brown E. L., Maldonado F. C., Tuntland T., Lee C. A., Fuhrman S. A., Zalman L. S., Patick A. K., Matthews D. A., Wu E. Y., Guo M., Borer B. C., Nayyar N. K., Moran T., Chen L., Rejto P. A., Rose P. W., Guzman M. C., Dovalsantos E. Z., Lee S., McGee K., Mohajeri M., Liese A., Tao J., Kosa M. B., Liu B., Batugo M. R., Gleeson J. P. R., Wu Z. P., Liu J., Meador J. W., Ferre R. A., J. Med. Chem., 2003, 46, 4572—4585 |
| [5] | Yang H., Xie W., Xue X., Yang K., Ma J., Liang W., Zhao Q., Zhou Z., Pei D., Ziebuhr J., Hilgenfeld R., Yuen K., Wong L., Gao G., Chen S., Chen Z., Ma D., Bartlam M., Rao Z., PLoS Biology, 2005, 3, 1742—1752 |
| [6] | Ghosh A. K., Xi K., Grum-Tokars V., Xu X., Ratia K., Fu W., Houser K. V., Baker S. C., Johnson M. E., Mesecar A. D., Bioorg. Med. Chem. Lett., 2007, 17, 5876—5880 |
| [7] | Shie J. J., Fang J. M., Kuo T. H., Kuo C. J., Liang P. H., Huang H. J., Wu Y. T., Jan J. T., Cheng Y. S., Wong C. H., Bioorg. Med. Chem., 2005, 13, 5240—5252 |
| [8] | Mou K., Xu B., Ma C., Yang X., Zou X., Lü Y., Xu P., Bioorg. Med. Chem. Lett., 2008, 18, 2198—2202 |
| [9] | Kuo C. J., Shie J. J., Fang J. M., Yen G. R., Hsu J. T., Liu H. G., Tseng S. N., Chang S. C., Lee C. Y., Shih S. R., Liang P. H., Bioorg. Med. Chem., 2008, 16, 7388—7398 |
| [10] | Ghosh A. K., Xi K., Ratia K., Santarsiero B. D., Fu W., Harcourt B. H., Rota P. A., Baker S. C., Johnson M. E., Mesecar A. D., J. Med. Chem., 2005, 48, 6767—6771 |
| [11] | Johnson T.O., Hua Y., Luu H. T., Brown E. L., Chan F., Chu S. S., Dragovich P. S., Eastman B. W., Ferre R. A., Fuhrman S. A., Hendrickson T. F., Maldonado F. C., Matthews D. A., Meador J. W., Patick A. K., Reich S. H., Skalitzky D. J., Worland S. T., Yang M., Zalman L. S., J. Med. Chem., 2002, 45, 2016—2023 |
| [12] | Lin D., Qian W., Hilgenfeld R., Jiang H., Chen K., Liu H., Sci. China Chem., 2012, 55, 1101—1107 |
| [13] | Hanessian S., Margarita R., Tetrahedron Lett., 1998, 39, 5887—5890 |
| [14] | McCarthy T. J., Nuzzo R. G., Whitesides G. M., J. Am. Chem. Soc., 1981, 103, 3396—3403 |
| [15] | Kokotos G., Padron J. M., Martin T., Gibbons W. A., Martin V. S., J. Org. Chem., 1998, 63, 3741—3744 |
| [16] | Wu Y.C., Bernadat G., Masson G. R., Couturier C. D., Schlama T., Zhu J., J. Org. Chem., 2009, 74, 2046—2052 |
| [17] | Arda A., G. Soengas R., Nieto M. I., Jiménez C., Rodriguez J., Org. Lett., 2008, 10, 2175—2178 |
| [18] | Padrón J. M., Kokotos G., Martín T., Markidis T., Gibbons W. A., Martín V. S., Tetrahedron: Asymmetry, 1998, 9, 3381—3394 |
| [19] | Gu Z.Q., Hesson D. P., Tetrahedron: Asymmetry, 1995, 6, 2101—2104 |
| [20] | Chen L., Lee S., Renner M., Tian Q., Nayyar N., Org. Proc. Res. Dev., 2006, 10, 163—164 |
| [21] | Zhou D. J., Toyooka N., Chem. J. Chinese Universities, 2012, 33(3), 511—515 |
| (周德军, 丰岗尚树.高等学校化学学报,2012, 33(3), 511—515) |
| [1] | LÜ Haoting, LIU Tingting, ZHANG Mingtao, LIU Jie, SUN Huailin. Synthesis of η6-[Tris(trimethylsilyl)phenylsilane]tricarbonylchromium Complexes and Theoretical Studies on the Possible Interaction of Intermolecular Si—Si Bonds and Chromium Centers† [J]. Chem. J. Chinese Universities, 2014, 35(11): 2341. |
| [2] | LIU Hao, XU Fen, SUN Li-Xian, CAO Zhong, ZHOU Huai-Ying. Preparation and Hydrogen Generation for Al-LiBH4 Composite Materials [J]. Chem. J. Chinese Universities, 2013, 34(8): 1953. |
| [3] | SHAO Chun-Guang, ZHUO Ran-Ran, LI Qian, CAO Wei, ZHANG Yang, ZHANG Rui-Jing, LIU Cheng-Gang, SHEN Chang-Yu. Relationship Between Crystal Orientation and Stress-induced Crystalline Melting of Syndiotactic Polypropylene Under Uniaxial Deformation via in situ Wide-angle X-ray Scattering [J]. Chem. J. Chinese Universities, 2013, 34(2): 485. |
| [4] | ZHOU Jing, LI Liang, HUANG Feng-Xian, SHEN Hong-Zhi, YANG Hang, ZHOU Qiang, WANG Wen-Quan, XU Da-Peng. In-situ Raman Spectra and X-ray Diffraction Study on Pressure-induced Phase Transition in Columbite ZnNb2O6 [J]. Chem. J. Chinese Universities, 2013, 34(10): 2383. |
| [5] | LIU Qiu-Hua, SUN Huai-Lin. Synthesis, Structure and Si—Si Bond Stability of η6-[Tris(trimethylsilyl)phenylsilane]tricarbonylmolybdenum Complexes [J]. Chem. J. Chinese Universities, 2011, 32(7): 1537. |
| [6] | GUO Xing-Yuan*, DING Zhan-Hui*, ZHAO Xu-Dong, QIU Li-Xia, XUE Yan-Feng, .... Influence of Excitation Intensities on Photoluminescence of ZnO(Wurtzite) Whiskers at Room Temperature [J]. Chem. J. Chinese Universities, 2010, 31(2): 243. |
| [7] | ZHENG Yi-Fan*, LIU Hua-Zhang*, LI Xiao-Nian. In situ X-ray Diffraction Investigation on Reduction Process of Ammonia-synthesis Fused-iron Catalysts and the Formation Mechanism of Its Active Phase [J]. Chem. J. Chinese Universities, 2009, 30(6): 1177. |
| [8] | SHI Shu-Yun1,2*, ZHAO Yu2, ZHANG Yu-Ping1, HUANG Ke-Long1, LIU Su-Qin1. Separatian and Identification of Constituents from Ligularia atroviolacea [J]. Chem. J. Chinese Universities, 2008, 29(5): 941. |
| [9] | SUN Feng-Mei, SHI De-Qing, TIAN Man-Man, TAN Xiao-Song. Synthesis and Biological Activities of 2-oxo-2-[1-(3-pyridylmethylamino)-1'-aryl]methyl-4-aryl-5,5-dimethyl-1,3,2-dioxaphosphinane [J]. Chem. J. Chinese Universities, 2006, 27(11): 2092. |
| [10] | GUO Xing-Yuan1, YU Ying-Ning2, XU Da-Peng1, DING Zhan-Hui1, SU Wen-Hui1,3,4. Growth of ZnO(Wurtzite) Whiskers Using the Floating Zone Method at High Oxygen Pressure [J]. Chem. J. Chinese Universities, 2006, 27(10): 1811. |
| [11] | LIU Lei-Jing, GENG Jian-Xin, ZHOU Yun-Chun, YIN Li, LI Gao, ZHOU En-Le, Jacky Lam Wing Yip, Tang Ben-Zhong. Crystal Structure of 5[(4'-Heptoxy-biphenylyl-4-yl)oxy]carbonyl-1-pentyne [J]. Chem. J. Chinese Universities, 2005, 26(7): 1366. |
| [12] | WANG Qiu-An, LUO Jun-Xia, LIAO Tou-Gen. A Novel Synthesis of Optically Pure trans-(+)-Sobrerol [J]. Chem. J. Chinese Universities, 2004, 25(6): 1069. |
| [13] | ZHANG Sheng-Mao, ZHANG Zhi-Jun, DANG Hong-Xin, LIU Wei-Min, XUE Qun-Ji . Preparation and Characterization of Orderly Alternating TiO2/CTAB Multilayer Film [J]. Chem. J. Chinese Universities, 2003, 24(6): 1136. |
| [14] | LU Yi, ZHU Zhen-Ping, WU Wei-Ze, LIU Zhen-Yu . Preparation of Carbon Nanotubes by Catalysis-assisted Detonation [J]. Chem. J. Chinese Universities, 2003, 24(6): 1063. |
| [15] | LI Zhao-Yang, ZHANG Zhi-Chao, LIU Yi-Shan, ZHOU Qi-Lin, GAO Ru-Yu, WANG Qin-Sun. Determination of Uniconazole in Soil with an Enantioselective Way [J]. Chem. J. Chinese Universities, 2003, 24(5): 840. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||