

Chem. J. Chinese Universities ›› 2020, Vol. 41 ›› Issue (3): 388.doi: 10.7503/cjcu20190631
• Review • Previous Articles Next Articles
GAO Qilong1,LIANG Erjun1,XING Xianran2,CHEN Jun3,*
Received:2019-12-06
Online:2020-03-10
Published:2020-01-07
Contact:
Jun CHEN
Supported by:CLC Number:
TrendMD:
GAO Qilong, LIANG Erjun, XING Xianran, CHEN Jun. Negative Thermal Expansion in Prussian Blue Analogues †[J]. Chem. J. Chinese Universities, 2020, 41(3): 388.
| Compound | 106αa/K-1 | 106αc/K-1 | 106αl/K-1 | ΔT/K | Crystal | Ref. |
|---|---|---|---|---|---|---|
| Au(CN) | 61.6 | -8.9 | 95—490 | Hexagonal | [ | |
| Ag(CN) | 70.8 | -24.8 | 95—490 | Trigonal | [ | |
| HT-Cu(CN) | 84.1 | -30.7 | 95—490 | Trigonal | [ | |
| Ni(CN)2 | -6.5 | 61.8 | 28—300 | Trigonal | [ | |
| Hg(CN)2 | 22.8 | 71.3 | 100—395 | Tetragonal | [ | |
| HgCN(NO3) | 26.1 | -23.0 | 100—395 | Hexagonal | [ | |
| Zn[Au(CN)2]2 | 36.9 | -57.5 | 100—775 | Hexagonal | [ | |
| Zn[Au(CN)2]2-xAgCN | 4.07 | -21.7 | 100—375 | Hexagonal | [ | |
| Cd(CN)2 | -20.4 | 150—375 | Cubic | [ | ||
| Zn(CN)2 | -16.9 | 25—375 | Cubic | [ | ||
| Single-network Cd(CN)2 | -33.5 | 170—375 | Cubic | [ | ||
| Cd(CN)2-0.64CCl4 | -16.9 | 240—375 | Cubic | [ | ||
| Cd(CN)2-0.75CCl4 | -5.7 | 200—375 | Cubic | [ | ||
| Cd(CN)2-CCl4 | 10.0 | 100—240 | Cubic | [ | ||
| Me4NCuZn(CN)4 | 0.67 | 218—368 | Cubic | [ |
| Compound | 106αa/K-1 | 106αc/K-1 | 106αl/K-1 | ΔT/K | Crystal | Ref. |
|---|---|---|---|---|---|---|
| Au(CN) | 61.6 | -8.9 | 95—490 | Hexagonal | [ | |
| Ag(CN) | 70.8 | -24.8 | 95—490 | Trigonal | [ | |
| HT-Cu(CN) | 84.1 | -30.7 | 95—490 | Trigonal | [ | |
| Ni(CN)2 | -6.5 | 61.8 | 28—300 | Trigonal | [ | |
| Hg(CN)2 | 22.8 | 71.3 | 100—395 | Tetragonal | [ | |
| HgCN(NO3) | 26.1 | -23.0 | 100—395 | Hexagonal | [ | |
| Zn[Au(CN)2]2 | 36.9 | -57.5 | 100—775 | Hexagonal | [ | |
| Zn[Au(CN)2]2-xAgCN | 4.07 | -21.7 | 100—375 | Hexagonal | [ | |
| Cd(CN)2 | -20.4 | 150—375 | Cubic | [ | ||
| Zn(CN)2 | -16.9 | 25—375 | Cubic | [ | ||
| Single-network Cd(CN)2 | -33.5 | 170—375 | Cubic | [ | ||
| Cd(CN)2-0.64CCl4 | -16.9 | 240—375 | Cubic | [ | ||
| Cd(CN)2-0.75CCl4 | -5.7 | 200—375 | Cubic | [ | ||
| Cd(CN)2-CCl4 | 10.0 | 100—240 | Cubic | [ | ||
| Me4NCuZn(CN)4 | 0.67 | 218—368 | Cubic | [ |
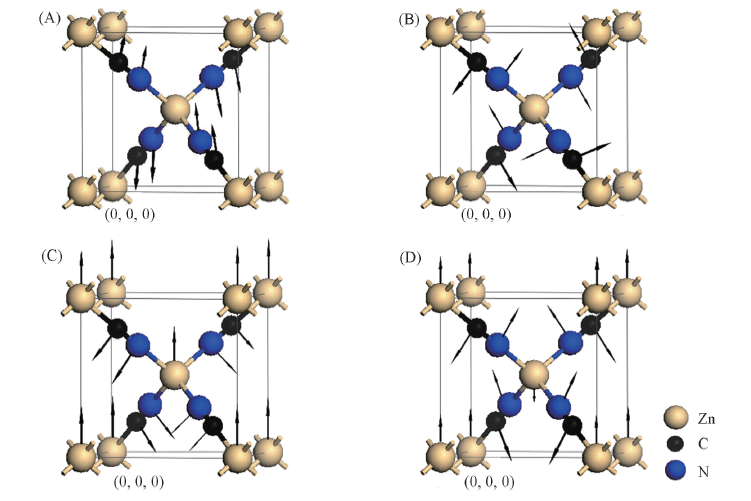
Fig.2 Typical vibrational manners of the phonon modes contributing to NTE in Zn(CN)2 of 47 cm-1(A), 263 cm-1(B), 189 cm-1(C) and 356 cm-1(D) [44] Copyright 2008, Chinese Physical Society.
| Compound | 106αV/K-1 | ΔT/K | Ref. | Compound | 106αV/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|---|---|---|
| FeCo(CN)6 | -4.41 | 4.2—300 | [ | YFe(CN)6 | -33.7 | 300—525 | [ |
| GaFe(CN)6 | -12 | 100—475 | [ | Mn3[Co(CN)6]2 | -87.6 | 123—300 | [ |
| FeFe(CN)6 | -12.8 | 100—475 | [ | Fe3[Co(CN)6]2 | -58.8 | 123—300 | [ |
| CdPt(CN)6 | -30.06 | 100—240 | [ | Co3[Co(CN)6]2 | -119.1 | 123—300 | [ |
| MnPt(CN)6 | -19.74 | 100—300 | [ | Ni3[Co(CN)6]2 | -90.0 | 123—300 | [ |
| FePt(CN)6 | -12 | 100—315 | [ | Cu3[Co(CN)6]2 | -60.0 | 123—300 | [ |
| ZnPt(CN)6 | -10.59 | 100—400 | [ | Zn3[Co(CN)6]2 | -89.1 | 123—300 | [ |
| CoPt(CN)6 | -4.8 | 100—350 | [ | Fe3[Fe(CN)6]2 | -29.7 | 123—300 | [ |
| NiPt(CN)6 | -3.06 | 100—330 | [ | Cu3[Fe(CN)6]2 | -59.7 | 123—300 | [ |
| CuPt(CN)6 | -4.71 | 100—400 | [ | Zn3[Fe(CN)6]2 | -118.8 | 123—300 | [ |
| ErCo(CN)6 | -27 | 100—500 | [ | Cs0.7Ni[Fe(CN)6]0.9·2.9H2O | -1.2 | 100—300 | [ |
| LaCo(CN)6 | -43.86 | 100—500 | [ | Cs0.97Cu[Fe(CN)6]0.99·1.1H2O | -6.3 | 100—300 | [ |
| SmCo(CN)6 | -37.38 | 100—500 | [ | Cs0.91Zn[Fe(CN)6]0.97·0.4H2O | -12.3 | 100—300 | [ |
| HoCo(CN)6 | -30.15 | 100—500 | [ | CsCd[Fe(CN)6]·0.5H2O | -26.4 | 100—300 | [ |
| LuCo(CN)6 | -27.15 | 100—500 | [ | Rb0.78Fe[Fe(CN)6]0.83·2.8H2O | -6.3 | 100—300 | [ |
| YCo(CN)6 | -31.59 | 100—500 | [ | Rb0.64Zn[Fe(CN)6]0.88·2.3H2O | -17.7 | 100—300 | [ |
| Compound | 106αV/K-1 | ΔT/K | Ref. | Compound | 106αV/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|---|---|---|
| FeCo(CN)6 | -4.41 | 4.2—300 | [ | YFe(CN)6 | -33.7 | 300—525 | [ |
| GaFe(CN)6 | -12 | 100—475 | [ | Mn3[Co(CN)6]2 | -87.6 | 123—300 | [ |
| FeFe(CN)6 | -12.8 | 100—475 | [ | Fe3[Co(CN)6]2 | -58.8 | 123—300 | [ |
| CdPt(CN)6 | -30.06 | 100—240 | [ | Co3[Co(CN)6]2 | -119.1 | 123—300 | [ |
| MnPt(CN)6 | -19.74 | 100—300 | [ | Ni3[Co(CN)6]2 | -90.0 | 123—300 | [ |
| FePt(CN)6 | -12 | 100—315 | [ | Cu3[Co(CN)6]2 | -60.0 | 123—300 | [ |
| ZnPt(CN)6 | -10.59 | 100—400 | [ | Zn3[Co(CN)6]2 | -89.1 | 123—300 | [ |
| CoPt(CN)6 | -4.8 | 100—350 | [ | Fe3[Fe(CN)6]2 | -29.7 | 123—300 | [ |
| NiPt(CN)6 | -3.06 | 100—330 | [ | Cu3[Fe(CN)6]2 | -59.7 | 123—300 | [ |
| CuPt(CN)6 | -4.71 | 100—400 | [ | Zn3[Fe(CN)6]2 | -118.8 | 123—300 | [ |
| ErCo(CN)6 | -27 | 100—500 | [ | Cs0.7Ni[Fe(CN)6]0.9·2.9H2O | -1.2 | 100—300 | [ |
| LaCo(CN)6 | -43.86 | 100—500 | [ | Cs0.97Cu[Fe(CN)6]0.99·1.1H2O | -6.3 | 100—300 | [ |
| SmCo(CN)6 | -37.38 | 100—500 | [ | Cs0.91Zn[Fe(CN)6]0.97·0.4H2O | -12.3 | 100—300 | [ |
| HoCo(CN)6 | -30.15 | 100—500 | [ | CsCd[Fe(CN)6]·0.5H2O | -26.4 | 100—300 | [ |
| LuCo(CN)6 | -27.15 | 100—500 | [ | Rb0.78Fe[Fe(CN)6]0.83·2.8H2O | -6.3 | 100—300 | [ |
| YCo(CN)6 | -31.59 | 100—500 | [ | Rb0.64Zn[Fe(CN)6]0.88·2.3H2O | -17.7 | 100—300 | [ |
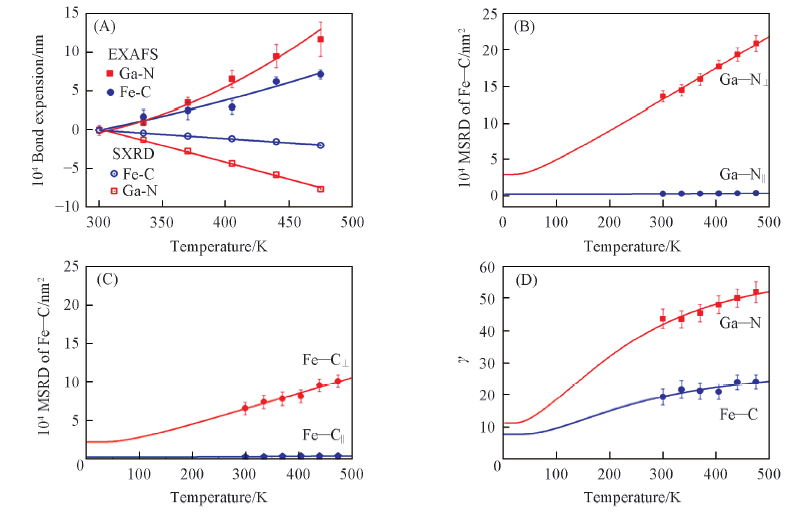
Fig.5 Comparison between the “apparent” and the “true” Ga—N and Fe—C bond lengths in GaFe(CN)6 as a function of temperature(A), Ga—N(B) and Fe—C(C) perpendicular(⊥) and parallel(‖) MSRDs of Ga—N atomic pairs of GaFe(CN)6 as a function of temperature, anisotropy of the relative thermal vibrations of the Ga—N and Fe—C atomic pairs of GaFe(CN)6 determined by EXAFS(D)[48] Copyright 2018, American Chemical Society.
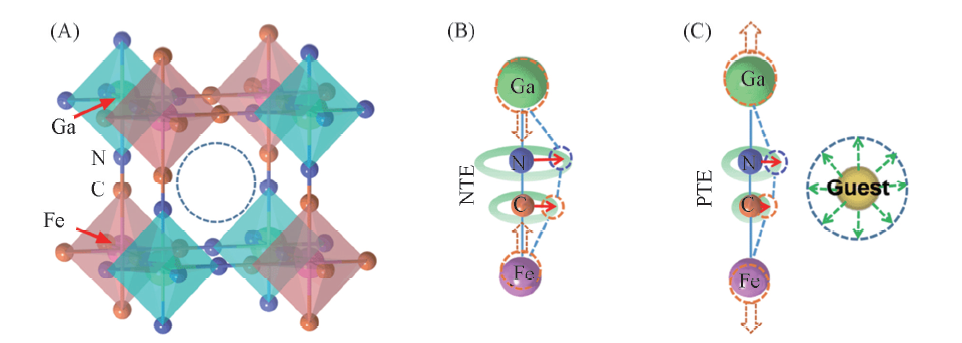
Fig.6 Sketch of the cubic structure of GaFe(CN)6 consisting of alternating GaN6 and FeC6 octahedra(A), the negative thermal expansion of GaFe(CN)6 derived from the transverse vibrations of CN cyanogen ions(B) and the transverse vibrations of CN cyanogen ions hindered by guest ions or molecules(C)[57] (A) The empty space indicated by the dash circle can host guest ions or molecules. Copyright 2018, American Chemical Society.
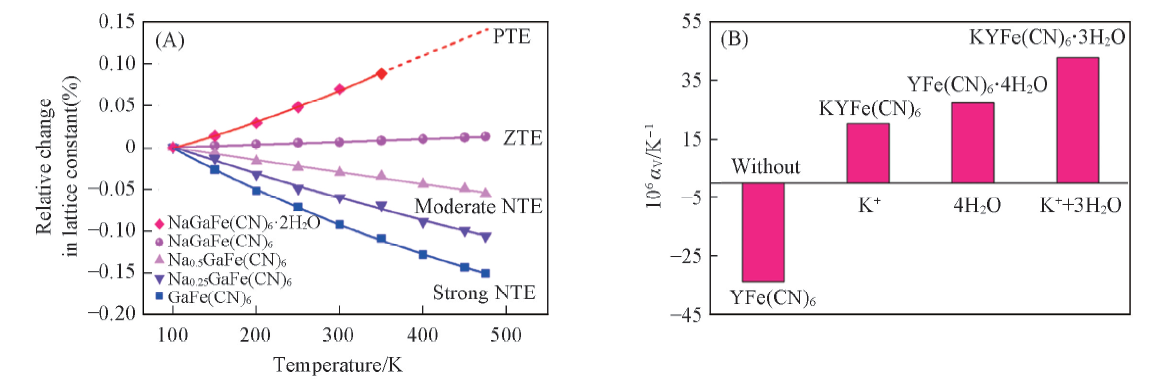
Fig.7 Temperature dependence of the relative change in lattice constant of GaFe(CN)6-based compounds(A)[57] and the coefficient of thermal expansion for YFe(CN)6-based materials within different guests(B)[28] (A) Copyright 2018, American Chemical Society; (B) copyright 2017, Wiley.
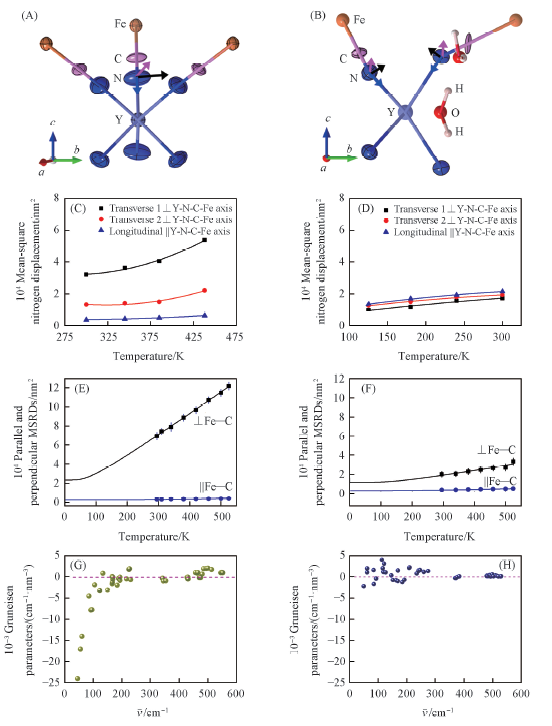
Fig.8 ADPs depiction and its temperature dependence for N atoms in YFe(CN)6(A,C) and YFe(CN)6·4H2O(B,D), respectively, the temperature dependence of MSRDs of the Fe—C atomic and the frequency dependence of mode Grüneisen parameters in YFe(CN)6(E,G) and KYFe(CN)6(F,H), respectively[28] Copyright 2017, Wiley.
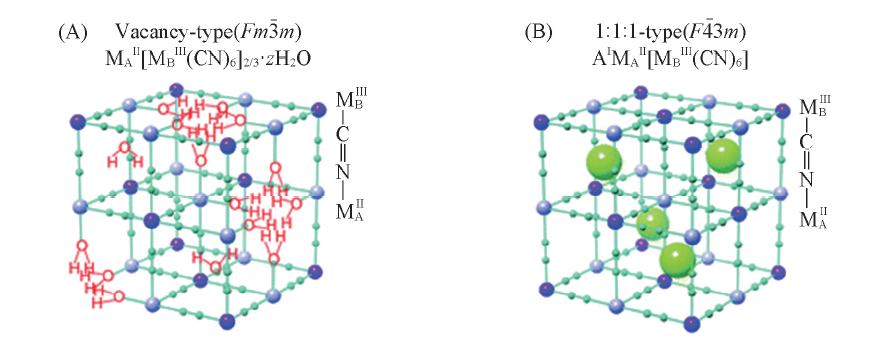
Fig.9 Schematic crystal structures of Prussian blue analogues MAⅡ[MBⅢ(CN)6]2/3·zH2O(A) and AⅠMⅡ[MⅢ(CN)6](B) [59] Copyright 2011, Royal Society of Chemistry.
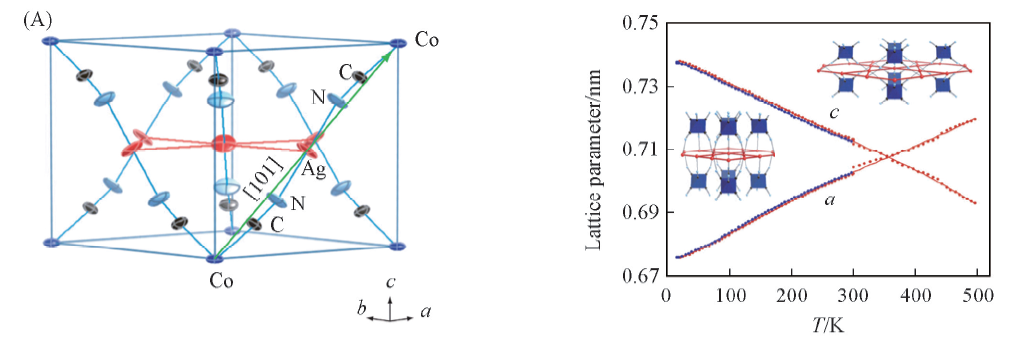
Fig.10 Structure of Ag3Co(CN)6, the strongest bonding interactions, which occur within Co—CN—Ag—NC—Co linkages, all lie parallel to the crystallographic [101] directions(A) and the lattice parameters for Ag3Co(CN)6 as a function of temperature(B)[20] (B) Inset show the sketch of large changes for Ag…Ag and [Co(CN)6]…[Co(CN)6] unit. Copyright 2008, Science.
| Compound | 106a/K-1 | 106c/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|
| H3Co(CN)6 | 12.0(4) | -8.8(3) | 4—300 | [ |
| D3Co(CN)6 | 17.4(6) | -5.04(24) | 8—304 | [ |
| Cu3Co(CN)6 | 25.4(5) | -43.5(8) | 100—596 | [ |
| Ag3Co(CN)6 | 145.9(6) | -122.1(3) | 16—500 | [ |
| Ag3Fe(CN)6 | 124.0(10) | -113(3) | 8—310 | [ |
| In[Au(CN)2]3 | 86.1—83.9 | -62.2—-63.3 | 100—395 | [ |
| In[Ag(CN)2]3 | 104—106 | -85—-83 | 100—395 | [ |
| KCd[Au(CN)2]3 | 72.9—71.4 | -56.1—-57.1 | 100—395 | [ |
| KCd[Ag(CN)2]3 | 76.6—74.9 | -64.3—-65.6 | 100—395 | [ |
| KNi[Au(CN)2]3 | 59.7—58.7 | -42.2—-42.7 | 100—395 | [ |
| KMn[Ag(CN)2]3 | 61(2) | -60(3) | 100—395 | [ |
| Compound | 106a/K-1 | 106c/K-1 | ΔT/K | Ref. |
|---|---|---|---|---|
| H3Co(CN)6 | 12.0(4) | -8.8(3) | 4—300 | [ |
| D3Co(CN)6 | 17.4(6) | -5.04(24) | 8—304 | [ |
| Cu3Co(CN)6 | 25.4(5) | -43.5(8) | 100—596 | [ |
| Ag3Co(CN)6 | 145.9(6) | -122.1(3) | 16—500 | [ |
| Ag3Fe(CN)6 | 124.0(10) | -113(3) | 8—310 | [ |
| In[Au(CN)2]3 | 86.1—83.9 | -62.2—-63.3 | 100—395 | [ |
| In[Ag(CN)2]3 | 104—106 | -85—-83 | 100—395 | [ |
| KCd[Au(CN)2]3 | 72.9—71.4 | -56.1—-57.1 | 100—395 | [ |
| KCd[Ag(CN)2]3 | 76.6—74.9 | -64.3—-65.6 | 100—395 | [ |
| KNi[Au(CN)2]3 | 59.7—58.7 | -42.2—-42.7 | 100—395 | [ |
| KMn[Ag(CN)2]3 | 61(2) | -60(3) | 100—395 | [ |
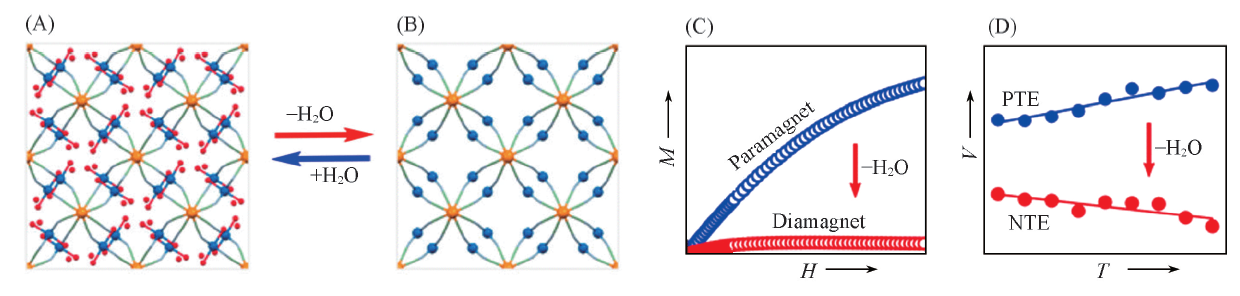
Fig.11 Structure schematic of Ni2Ⅱ[WⅣ(CN)8]·6H2O(A) and Ni2Ⅱ[WⅣ(CN)8](B), the comparison physical property of Ni2Ⅱ[WⅣ(CN)8]·6H2O and Ni2Ⅱ[WⅣ(CN)8] in magnetism(C) and thermal expansion(D) [32] Copyright 2016, American Chemical Society.
| [1] | Chen J., Hu L., Deng J., Xing X ., Chem. Soc. Rev., 2015, 44, 3522— 3567 |
| [2] | Pryde A. K. A., Hammonds K. D., Dove M. T., Heine V., Gale J. D., Warren M. C ., J. Phys. Condens. Matter., 1996, 8, 10973— 10982 |
| [3] | Ernst G., Broholm C., Kowach G. R., Ramirez A. P ., Nature, 1998, 396, 56— 57 |
| [4] | Ramirez A. P., Kowach G. R ., Phys. Rev. Lett., 1998, 80, 4903— 4906 |
| [5] | Yuan B., Liu X., Song W., Cheng Y., Liang E., Chao M ., Phys. Lett. A, 2014, 378, 3397— 3401 |
| [6] | Chen D., Yuan B., Cheng Y., Ge X., Jia Y., Liang E., Chao M ., Phys. Lett. A, 2016, 380, 4070— 4074 |
| [7] | Huang L. P., Kieffer J ., Phys. Rev. Lett., 2005, 95, 215901 |
| [8] | Yamamura Y., Ikeuchi S., Saito K ., Chem. Mater., 2009, 21, 3008— 3016 |
| [9] | Qiu L., Bao Y., Wen S., Liang E. J ., Chinese Phys. B, 2012, 21, 046501 |
| [10] | Liu Y., Zhang N., Jia Y., Sun Q., Chao M ., Phys. Lett. A, 2015, 379, 2756— 2760 |
| [11] | Liang E. J ., Chinese Phys. Lett., 2013, 30, 126502 |
| [12] | Chang D., Yu W., Sun Q., Jia Y ., Phys. Chem. Chem. Phys., 2017, 19,2067—2072 |
| [13] | Woodcock D. A., Lightfoot P., Villaescusa L. A., Diaz-Cabanas M. J ., Chem. Mater., 1999, 11, 2508— 2514 |
| [14] | Ge X., Liu X., Cheng Y., Yuan B., Chen D., Chao M., Liang E. J ., J. Appl. Phys., 2016, 120, 205101 |
| [15] | Carey T., Corma A., Rey F., Tang C. C., Hriljac J. A., Anderson P. A ., Chem. Commun., 2012, 48, 5829— 5831 |
| [16] | Goodwin A. L., Chapman K. W., Kepert C. J ., J. Am. Chem. Soc., 2005, 127, 17980— 17981 |
| [17] | Hibble S. J., Chippindale A. M., Pohl A. H., Hannon A. C ., Angew. Chem. Int. Ed., 2007, 46, 7116— 7118 |
| [18] | Margadonna S., Prassides K., Fitch A. N ., J. Am. Chem. Soc., 2005, 126, 15390— 15391 |
| [19] | Duyker S. G., Peterson V. K., Kearley G. J., Anibal J. R., Kepert C. J ., Angew. Chem., 2013, 125, 5374— 5378 |
| [20] | Goodwin A. L., Calleja M., Conterio M. J., Dove M. T., Evans J. S. O., Keen D. A., Peters L., Tucker M. G ., Science, 2008, 319, 794— 797 |
| [21] | Li M., Li Y., Wang C. Y., Sun Q ., Chinese Phys. Lett., 2019, 36, 066301 |
| [22] | Liu Z., Liu C., Li Q., Chen J., Xing X ., Phys. Chem. Chem. Phys., 2017, 19, 24436— 24439 |
| [23] | Dubbeldam D., Walton K. S., Ellis D. E., Snurr R. Q ., Angew. Chem. Int. Ed., 2007, 119, 4580— 4583 |
| [24] | Wang L., Yuan P. F., Wang F., Sun Q., Liang E. J., Jia Y., Guo Z. X ., Phys. Lett. A, 2014, 378, 2906— 2909 |
| [25] | Hu L., Chen J., Fan L., Ren Y., Xing X ., J. Am. Chem. Soc., 2014, 136, 13566— 13569 |
| [26] | Hu L., Chen J., Sanson A Wu H., Rodriguez C. G., Olivi L., Ren Y., Fan L., Deng J., Xing X ., J. Am. Chem. Soc., 2016, 138, 8320— 8323 |
| [27] | Chang D., Niu C. Y., Huang X., Sun Q., Cho J. H., Jia Y ., Phys. Rev. B, 2017, 95, 104101 |
| [28] | Gao Q. L., Chen J., Sun Q., Chang D. H., Huang Q. Z., Wu H., Sanson A., Milazzo R., Zhu H., Li Q., Liu Z. N., Deng J. X., Xing X. R ., Angew. Chem. Int. Ed., 2017, 56, 9023— 9028 |
| [29] | Mary T. A., Evans J. S. O., Vogt T., Sleight A. W ., Science, 1996, 272, 90— 92 |
| [30] | Liu Y., Jia Y., Sun Q., Liang E. J ., Phys. Lett. A, 2015, 379, 54— 59 |
| [31] | Buser H. J., Schwarzenbach D., Petter W., Ludi A ., Inorg. Chem., 1977, 16, 2704— 2710 |
| [32] | Reczyński M., Chorazy S., Nowicka B., Sieklucka B., Ohkoshi S ., Inorg. Chem., 2016, 56, 179— 185 |
| [33] | Tokoro H., Nakagawa K., Imoto K., Hakoe F., Ohkoshi S ., Chem. Mater., 2012, 24, 1324— 1330 |
| [34] | Chippindale A. M., Hibble S. J., Bilbé E. J., Marelli E., Hannon A. C., Allain C., Pansu R., Hartl F ., J. Am. Chem. Soc., 2012, 134, 16387— 16400 |
| [35] | Goodwin A. L., Kepert C. J ., Phys. Rev. B, 2005, 71, 140301 |
| [36] | Williams D. J., Partin D. E., Lincoln F. J., Kouvetakis J., O'Keeffe M ., J. Solid State Chem., 1997, 134, 164— 169 |
| [37] | Hibble S. J., Hannon A. C., Cheyne S. M ., Inorg. Chem., 2003, 34, 4724— 4730 |
| [38] | Korčok J. L., Leznoff D. B ., Polyhedron, 2013, 52, 72— 77 |
| [39] | Goodwin A. L., Kennedy B. J., Kepert C. J ., J. Am. Chem. Soc., 2009, 131, 6334— 6335 |
| [40] | Phillips A. E., Goodwin A. L., Halder G. J., Southon P. D., Kepert C. J ., Angew. Chem. Int. Ed., 2008, 47, 1396— 1399 |
| [41] | Phillips A. E., Halder G. J., Chapman K. W., Goodwin A. L., Kepert C. J ., J. Am. Chem. Soc., 2009, 132, 10— 11 |
| [42] | Zwanziger J. W ., Phys. Rev. B, 2007, 76, 052102 |
| [43] | Ding P., Liang E. J., Jia Y., Du Z ., J. Phys. Condens. Matter, 2008, 20, 275224 |
| [44] | Du Z. Y., Liang E. J., Ding P., Wang J. P., Xu E. M ., J. Light Scattering, 2008, 20( 2), 145— 150 |
| ( 杜志勇, 梁二军, 丁佩, 王俊平, 徐二明 . 光散射学报, 2008, 20( 2), 145— 150) | |
| [45] | Hibble S. J., Chippindale A. M., Marelli E., Kroeker S., Michaelis V. K., Greer B. J., Aguiar P. M., Bilbé E. J., Barney E. R., Hannon A. C ., J. Am. Chem. Soc., 2013, 135, 16478— 16489 |
| [46] | Chapman K. W., Peter J. C ., J. Am. Chem. Soc., 2007, 129, 10090— 10091 |
| [47] | Ovens J. S., Leznoff D. B ., Inorg. Chem., 2017, 56, 7332— 7343 |
| [48] | Gao Q., Shi N., Sun Q., Sanson A., Milazzo R., Carnera A., Zhu H., Lapidus S. H., Ren Y., Huang Q., Chen J., Xing X ., Inorg. Chem., 2018, 57, 10918— 10924 |
| [49] | Shi N., Gao Q., Sanson A., Li Q., Fan L., Ren Y., Olivi L., Chen J., Xing X ., Dalton T., 2019, 48, 3658— 3663 |
| [50] | Chapman K. W., Chupas P. J., Kepert C. J ., J. Am. Chem. Soc., 2010, 37, 7009— 7014 |
| [51] | Pretsch T., Chapman K. W., Halder G. J., Kepert C. J ., Chem. Commun., 2006, ( 17), 1857— 1859 |
| [52] | Adak S., Daemen L. L., Hartl M., Williams D., Summerhill J., Nakotte H ., J. Solid State Chem., 2011, 184, 2854— 2861 |
| [53] | Matsuda T., Kim J. E., Ohoyama K., Moritomo Y ., Phys. Rev. B, 2009, 79, 172302 |
| [54] | Sanson A ., Chem. Mater., 2014, 26, 3716— 3720 |
| [55] | Chapman K. W., Chupas P. J., Kepert C. J ., J. Am. Chem. Soc., 2005, 127, 15630— 15636 |
| [56] | Vaccari M., Grisenti R., Fornasini P., Rocca F., Sanson A ., Phys. Rev. B, 2007, 75, 184307 |
| [57] | Gao Q., Shi N., Sanson A., Sun Y., Milazzo R., Olivi L., Chen J., Xing X ., Inorg. Chem., 2018, 57, 14027— 14030 |
| [58] | Chen J., Gao Q. L., Sanson A., Jiang X. X., Huang Q. Z., Carnera A., Rodriguez C. G., Olivi L., Wang L., Hu L., Lin K., Ren Y., Lin Z. S., Wang C., Gu L., Deng J. X., Attfield J. P., Xing X. R ., Nat. Commun. 2017, 8, 14441 |
| [59] | Tokoro H., Ohkoshi S. I ., Dalton T., 2011, 40, 6825— 6833 |
| [60] | Adak S., Daemen L. L., Nakotte H ., J. Phys Conf. Ser., 2010, 251, 012007 |
| [61] | Matsuda T., Tokoro H., Hashimoto K., Ohkoshi S. I ., Dalton T., 2006, 42, 5046— 5050 |
| [62] | Korcok J. L., Katz M. J., Leznoff D. B ., J. Am. Chem. Soc., 2009, 131, 4866— 4871 |
| [63] | Wang L., Wang C., Luo H., Sun Y ., J. Phys. Chem. C, 2017, 121, 333— 341 |
| [64] | Cairns A. B., Thompson A. L., Tucker M. G., Haines J., Goodwin A. L ., J. Am. Chem. Soc., 2012, 134, 4454— 4456 |
| [65] | Keen D. A., Dove M. T., Evans J. S., Goodwin A. L., Peters L., Tucker M. G ., J. Phys-Condens Mat., 2010, 22, 404202 |
| [66] | Sapnik A. F., Liu X., Boström H. L. B., Coates C. S., Overy A. R., Reynolds E. M., Tkatchenko A., Goodwin A. L ., J. Solid State Chem., 2018, 258, 298— 306 |
| [1] | SHI Naike, ZHANG Ya, SANSON Andrea, WANG Lei, CHEN Jun. Uniaxial Negative Thermal Expansion and Mechanism in Zn(NCN) [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220124. |
| [2] | REN Yu-Ying ZHOU Ding GUO Jing SHI Ying XIE Jian-Jun XIE Zhi HE Bo. Synthesis, Structure and Gas Adsorption Property of a Novel 2D Layer Nickel Organo\|diphosphonate Compound [J]. Chem. J. Chinese Universities, 2011, 32(1): 32. |
| [3] | LIU Qin-Qin, YANG Juan, SUN Xiu-Juan, CHENG Xiao-Nong. Preparation, Characterization and Negative Thermal Expansion Property of ZrWMoO8 Powders with Different Morphologies [J]. Chem. J. Chinese Universities, 2007, 28(3): 397. |
| [4] | LIU Xiao-Hua, KANG Mao-Qing, WANG Xin-Kui . Studies on the Active Species of Propylene Oxide Polymerization Initiated by Double Metal Cyanide Complex Catalyst [J]. Chem. J. Chinese Universities, 2000, 21(11): 1748. |
| [5] | Guo Zhenya, Wang Jingzhong, Wang Kaixuan, Zhou Daming, Xiang Shouhe, Li Hexuan . Physico-Chemical Characterization of ZSM-5 Zeolite Synthesized Without Template [J]. Chem. J. Chinese Universities, 1990, 11(8): 800. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||