

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (7): 1447.doi: 10.7503/cjcu20190091
• Physical Chemistry • Previous Articles Next Articles
LI Chunxiao, LI Jian*( ), LIANG Wenjun, LIANG Quanming
), LIANG Wenjun, LIANG Quanming
Received:2019-02-01
Online:2019-07-10
Published:2019-07-12
Contact:
LI Jian
E-mail:ljian@bjut.edu.cn
Supported by:CLC Number:
TrendMD:
LI Chunxiao, LI Jian, LIANG Wenjun, LIANG Quanming. Low Temperature NH3-SCR Activity of Cr Doped V2O5-WO3/TiO2 Catalyst†[J]. Chem. J. Chinese Universities, 2019, 40(7): 1447.
| Catalyst | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) | Average pore size/nm |
|---|---|---|---|
| 3V6W/TiO2 | 78.857 | 0.380 | 17.533 |
| 1Cr3V6W/TiO2 | 66.756 | 0.325 | 17.944 |
| 3Cr3V6W/TiO2 | 66.182 | 0.312 | 18.028 |
| 5Cr3V6W/TiO2 | 65.287 | 0.310 | 18.523 |
| 7Cr3V6W/TiO2 | 63.937 | 0.301 | 18.729 |
| 9Cr3V6W/TiO2 | 62.172 | 0.293 | 19.239 |
Table 1 Specific surface area, pore volume and average pore size of the catalysts
| Catalyst | Specific surface area/(m2·g-1) | Pore volume/(cm3·g-1) | Average pore size/nm |
|---|---|---|---|
| 3V6W/TiO2 | 78.857 | 0.380 | 17.533 |
| 1Cr3V6W/TiO2 | 66.756 | 0.325 | 17.944 |
| 3Cr3V6W/TiO2 | 66.182 | 0.312 | 18.028 |
| 5Cr3V6W/TiO2 | 65.287 | 0.310 | 18.523 |
| 7Cr3V6W/TiO2 | 63.937 | 0.301 | 18.729 |
| 9Cr3V6W/TiO2 | 62.172 | 0.293 | 19.239 |
| Catalyst | Eb/eV | Surface atomic concentration | V5+/V4+ | Oβ/Oα | Cr6+/Cr3+ | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| V2p | Cr2p | O1s | O | V | Cr | |||||||
| 3V6W/TiO2 | 516.4 | 517.1 | — | — | 530.0 | 531.6 | 13.52 | 1.00 | — | 1.401 | 0.127 | |
| 3Cr3V6W/TiO2 | 516.4 | 517.1 | 575.8 | 579.5 | 529.9 | 531.4 | 16.06 | 0.91 | 1.09 | 0.312 | 0.425 | 0.230 |
Table 2 Binding energies and surface atomic concentrations of O, Cr and V for the prepared catalysts determined from deconvoluted XPS spectra*
| Catalyst | Eb/eV | Surface atomic concentration | V5+/V4+ | Oβ/Oα | Cr6+/Cr3+ | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| V2p | Cr2p | O1s | O | V | Cr | |||||||
| 3V6W/TiO2 | 516.4 | 517.1 | — | — | 530.0 | 531.6 | 13.52 | 1.00 | — | 1.401 | 0.127 | |
| 3Cr3V6W/TiO2 | 516.4 | 517.1 | 575.8 | 579.5 | 529.9 | 531.4 | 16.06 | 0.91 | 1.09 | 0.312 | 0.425 | 0.230 |
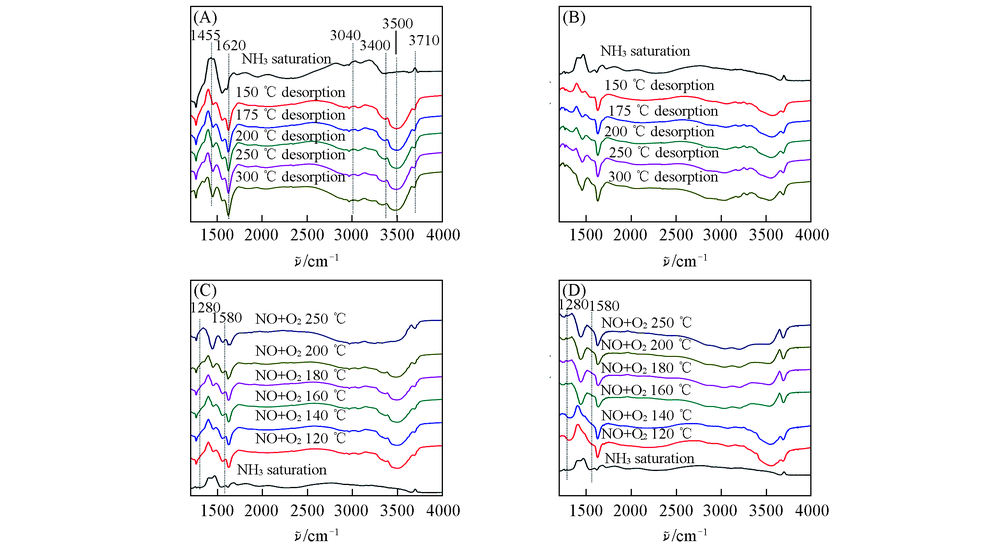
Fig.6 In situ DRIFTs spectra of catalysts(A) 3V6W/TiO2 adsorbed NH3 in 1%NH3-N2 and desorbed in N2; (B) 3Cr3V6W/TiO2 adsorbed NH3 in 1%NH3-N2 and desorbed in N2; (C) 3V6W/TiO2 adsorbed NH3 in 1%NH3-N2 then reacted in 0.1%NO+5%O2/N2; (D) 3Cr3V6W/TiO2 adsorption in 1%NH3-N2 then reacted in 0.1%NO+5%O2-N2.
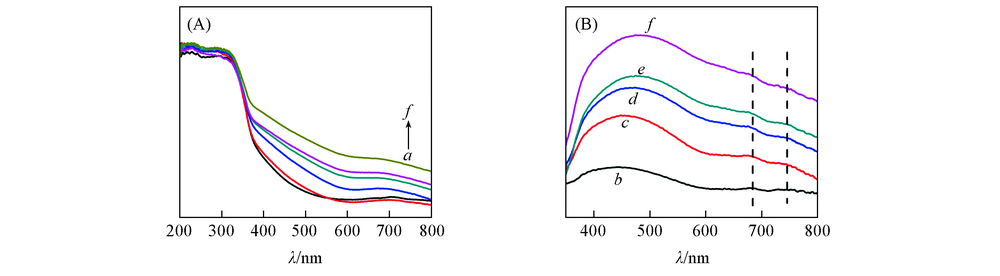
Fig.7 UV-Vis spectra of catalysts(A) 200—800 nm; (B) 350—800 nm(3V6W/TiO2 sample as background). a. 3V6W/TiO2;b. 1Cr3V6W/TiO2; c. 3Cr3V6W/TiO2; d. 5Cr3V6W/TiO2; e. 7Cr3V6W/TiO2; f. 9Cr3V6W/TiO2.
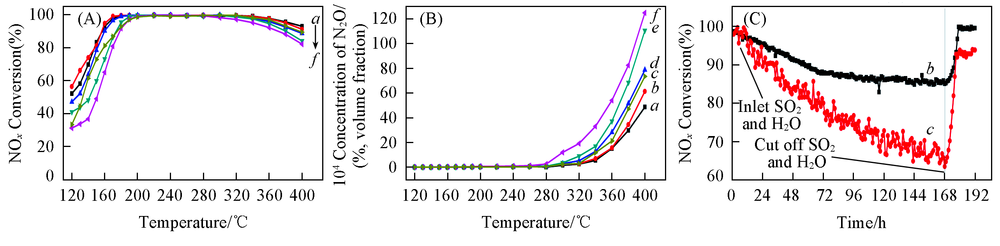
Fig.8 NH3-SCR activity(A), N2 selective(B) and SO2 resistance(C) of catalystsa. 1Cr3V6W/TiO2; b. 3Cr3V6W/TiO2; c. 3V6W/TiO2; d. 5Cr3V6W/TiO2; e. 7Cr3V6W/TiO2; f. 9Cr3V6W/TiO2.
| [1] | Zhang Q. X., Preparation and Characterization of Chromium Doped Catalysts and Study on NO Properties of Catalytic Oxidation, Nanjing University of Science and Technology, Nanjing, 2013 |
| (张婧欣. 铬基催化剂的制备、 表征及其催化氧化NO性能的研究, 南京: 南京理工大学, 2013) | |
| [2] | Ren X.G., Ren C. L., Fu X.,Li P., Song Y. J., Chem. Eng. J., 2012, 6(9), 3219—3222 |
| (任晓光, 任超李, 富霞, 李鹏, 宋永吉. 环境工程学报, 2012, 6(9), 3219—3222) | |
| [3] | Zhuang F. L., Liu S. G., Lin W. S., Chen Y.,Chen C. W., Environ. Eng., 2016, 34(12), 98—102 |
| (庄凤来, 刘少光, 林文松, 陈焱, 陈成武. 环境工程, 2016, 34(12), 98—102) | |
| [4] | Luo Y., Study on Low Temperature Selective Catalytic Reduction of Nitrogen Oxide Chromium Cerium Catalyst, South China University of Technology, Guangzhou, 2016 |
| (罗洋. 低温选择性催化还原氮氧化物铬铈催化剂研究, 广州: 华南理工大学, 2016) | |
| [5] | Zhuang S. L., Wang X. T., Energy Technology and Management, 2012, (5), 1—3 |
| (庄沙丽, 王学涛. 能源技术与管理, 2012, (5), 1—3) | |
| [6] | Duan J.W., Wang X. T., Zhang F. S., Liu J. G., Chen J. T., Journal of Functional Materials, 2018, 49(3), 3103—3108 |
| (段景卫, 王学涛, 张福顺, 刘剑光, 陈江涛. 功能材料, 2018, 49(3), 3103—3108) | |
| [7] | Sun X. L., Hong H., Su Y. C., Yan J. F., Song L. Y., Qiu W. G., Chem. J. Chinese Universities, 2017, 38(5), 814—822 |
| (孙向丽, 何洪, 苏垚超, 闫京芳, 宋丽云, 邱文革. 高等学校化学学报, 2017, 38(5), 814—822) | |
| [8] | Schneider H., Maciejewski M., Kohler K., Wokaun A., Baiker A., Kohler K., J. Catal., 1995, 157(2), 312—320 |
| [9] | Yang R., Huang H. F., Chen Y. J.,Zhang X. X., Lu H. F., Chinese J. Catal., 2015, 36(8), 1256—1262 |
| [10] | Karami A., Salehi V., J. Catal., 2012, 292, 32—43 |
| [11] | Shen K., Zhang Y.P., Wang X. L., Xu H. T., Sun K. Q., Zhou C. C., J. Energy Chem., 2013, 22(4), 617—623 |
| [12] | Huang H.F., Jin L. L., Lu H. F., Yu H., Chen Y. J., Catal. Commun., 2013, 34, 1—4 |
| [13] | Liu F. D., Shan W. P.,Shi X. Y.,Zhang C. F., He H., Chinese J. Catal., 2011, 32(7), 1113—1128 |
| (刘福东, 单文坡, 石晓燕, 张长斌, 贺泓. 催化学报, 2011, 32(7), 1113—1128) | |
| [14] | Paniego A. R., Anales De Quimica Serie A-Quimica Fisica Y Quimica Tecnica, 1989, 85(3), 386—399 |
| [15] | Farokhbag M., Jan B., Morteza S., Mostafa F., Mehri S., Appl. Catal.B: Environ., 2003, 46(1), 65—76 |
| [16] | Li Q., Yang H. S., Nie A. M.,Fan X. Y., Zhang X. B., Catal. Lett., 2011, 141(8), 1237—1242 |
| [17] | Thirupathi B., Smirniotis P. G., Appl. Catal. B: Environ., 2011, 110, 195—206 |
| [18] | Huong T.D., Pullur A. K., Young E. J., Heon P. H., Res. Chem. Intermediates, 2016, 42(1), 155—169 |
| [19] | Li J.J., Mu Y., Yang J., Yu J., Duan Z. K., Xu G. W., Xiong B. C., CIESC Journal, 2013, 64(4), 1249—1255 |
| (李俊杰, 牟洋, 杨娟, 余剑, 段正康. 化工学报, 2013, 64(4), 1249—1255) | |
| [20] | Nagaveni K., Hegde M.S., Ravishankar N., Subbanna G. N., Giridhar M., Langmuir, 2004, 20(7), 2900—2907 |
| [21] | Zhang S., Ultrasonics Sonochemistry, 2012, 19(4), 767—771 |
| [22] | Huang H.F., Han Y., He D. F., Asico L. D., Jose P. A., Zeng C. Y., International Journal of Cardiology, 2009, 137, S71—S72 |
| [23] | Zhong L.,C ai W., Yu Y., Zhong Q., Appl. Surf. Sci., 2015, 325, 52—63 |
| [24] | Wang J., Yan Z., Liu L., Chen Y., Zhang Z., Wang X., Appl. Surf. Sci., 2014, 313, 660—669 |
| [25] | Centeno M. A. Carrizosa I., Odriozola J. A., Appl. Catal. B:Environ., 2001, 29(4), 307—314 |
| [26] | Liu H. D., Li W. M.,Huang Y. P., Chen Y. F., Chinese J. Inorg. Chem., 2013, 29(11), 2399—2404 |
| (刘海弟, 李伟曼, 黄亿苹, 陈运法. 无机化学学报, 2013, 29(11), 2399—2404) | |
| [27] | Shan W., Liu F., He H., Shi X., Zhang C., Appl. Catal.B: Environ., 2012, 115, 100—106 |
| [28] | Kumar S. G., Devi L. G., J. Phys. Chem. A, 2011, 115(46), 13211—13241 |
| [29] | Weckhuysen B.M., Spooren H. J., Schoonheydt R. A., Zeolites, 1994, 14(6), 450—457 |
| [30] | Shi D., Zhao Z. Xu C., Duan A., Liu J., Dou T., J. Molecular Catal. A: Chem., 2006, 245(1/2), 106—113 |
| [31] | Weckhuysen B.M., Verberckmoes A. A., de Baets A. R., Schoonheydt R. A., J. Catal., 1997, 166(2), 160—171 |
| [32] | Cimino A., Cordischi D., de Rossi S., Ferraris G., Gazzoli D., Indovina V., Occhiuzzi M., Valigi M., J. Catal., 1991, 127(2), 761—776 |
| [33] | Satsuma A., Shibata J., Shimizu K. I., Hattori T., Catalysis Surveys from Asia, 2005, 9(2),75—85 |
| [34] | Dzwigaj S., Shishido T., J. Phys. Chem. C, 2008, 112(15), 5803—5809 |
| [35] | Puurunen R. L., Weckhuysen B. M., J. Catal., 2002, 210(2), 418—430 |
| [36] | Gao Y., Luan T., Lü T., Cheng K., Xu H. M., Chinese J. Chem. Eng., 2013, 21(1), 1—7 |
| [37] | Chen C., Cao Y., Liu S., Chen J., Jia W., Chinese J. Catal., 2018, 39(8), 1347—1365 |
| [38] | Wu B. J., Xiao P., Liu X. Q., Chem. Res. Chinese Universities, 2010, 26(6), 1002—1006 |
| [39] | Yi T., Li J., Zhang Y., Yang X., Chem. Res. Chinese Universities, 2018, 34(4), 661—664 |
| [1] | ZHANG Ling,DUAN Hongchang,TAN Zhengguo,WU Qinming,MENG Xiangju,XIAO Fengshou. Recent Advances in the Preparation of 8MR Zeolites for the Selective Catalytic Reduction of NOx(NH3-SCR) in Diesel Engines † [J]. Chem. J. Chinese Universities, 2020, 41(1): 19. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||