

Chem. J. Chinese Universities ›› 2018, Vol. 39 ›› Issue (10): 2143.doi: 10.7503/cjcu20180224
• Articles:Inorganic Chemistry • Previous Articles Next Articles
Received:2018-03-22
Online:2018-09-29
Published:2018-09-29
Contact:
BEN Teng
E-mail:tben@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Yun,BEN Teng. Methane Storage Performace of Self-activated Seaweed-based Carbon Materials†[J]. Chem. J. Chinese Universities, 2018, 39(10): 2143.
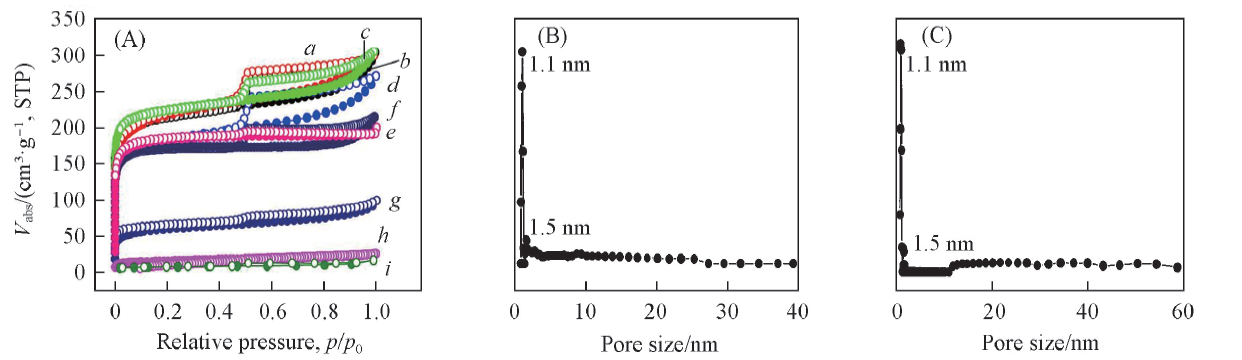
Fig.1 N2 sorption isotherms of different carbonized biomasses(A) and pore size distribution of SarCW-900-HF(B) and LamCW-900-HF(C)a. SarCW-900-HF; b. SarCW-850-HF; c. LamCW-900-HF; d. SarCW-950-HF; e. carbonized Walnut peel;f. LamCW-850-HF; g. LamCW-950-HF; h. carbonized passiflora edulia sims peel; i. Bentonite.

Fig.2 SEM images of Sar(A—E), Lam(F—J), SarCW-900-HF(K, L), LamCW-900-HF(O, P), TEM images of SarCW-900-HF(M, N) and LamCW-900-HF(Q, R)Insets of (N) and (R) are the SAED of the sample.
| Material | Surface area/ (m2·g-1) | Pore volume/ (cm3·g-1) | Condition | CH4 storage capacity (mass ratio,%) | Excess CH4 uptake/ (cm3·cm-3) | Heat of adsorption/ (kJ·mol-1) | Ref. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p/MPa | T/K | |||||||||||||||||
| Porous | COF-102 | 3620 | 1.55 | 3.5 | 298 | 15.8 | 120 | 8.6 | [ | |||||||||
| polymers | COF-103 | 3530 | 1.54 | 3.5 | 298 | 14.9 | 152 | 9.5 | [ | |||||||||
| HCP-1 | 1904 | 0.54 | 1.5 | 298 | 6.4 | [ | ||||||||||||
| HCP-4 | 1366 | 0.55 | 3.6 | 298 | 8.1 | [ | ||||||||||||
| PIM-1 | 850 | 0.349 | 1.5 | 293 | 3.8 | [ | ||||||||||||
| PPN-3 | 2840 | 1.7 | 3.5 | 295 | 12.2 | 15.2 | [ | |||||||||||
| PPN-4 | 6461 | 3.04 | 5.5 | 295 | 21.5 | [ | ||||||||||||
| MOFs | Cu2(sbtc)[PCN-11] | 1931 | 0.91 | 2.5 | 298 | 14.0 | 171 | 14.6 | [ | |||||||||
| Ni(dhtp)[NiMOF-74,CPO-27-Ni] | 1027 | 0.54 | 3.5 | 298 | 11.9 | 206 | 21.5—22 | [ | ||||||||||
| Zn4O(bdc)3[MOF-5] | 1870 | 3.6 | 13.5 | 110 | 12.2 | [ | ||||||||||||
| LIFM-82 | 1624 | 0.71 | 3.5 | 298 | 15.2 | 196 | 17.5 | [ | ||||||||||
| MOF-905 | 3490 | 1.34 | 3.5 | 298 | 18.8 | 145 | 11.7 | [ | ||||||||||
| HKUST-1 | 1850 | 0.78 | 3.5 | 298 | 18.4 | 227 | 17.0 | [ | ||||||||||
| NU-125 | 3120 | 1.29 | 3.5 | 298 | 16.5 | 182 | 15.1 | [ | ||||||||||
| PCN-14 | 2000 | 0.829 | 3.5 | 298 | 17.4 | 202 | 17.6 | [ | ||||||||||
| UTSA-76 | 2820 | 1.06 | 3.5 | 298 | 15.4 | 211 | 15.4 | [ | ||||||||||
| UTSA-20 | 1620 | 3.5 | 298 | 18.1 | 184 | [ | ||||||||||||
| NOTT-102 | 3342 | 1.268 | 3.5 | 298 | 16.5 | 181 | 16.0 | [ | ||||||||||
| NOTT-103 | 2958 | 1.157 | 3.5 | 300 | 15.6 | 193 | 15.9 | [ | ||||||||||
| MAF-38 | 2022 | 0.808 | 3.5 | 298 | 14.1 | 226 | 21.6 | [ | ||||||||||
| ZJU-35 | 2899 | 1.156 | 3.5 | 300 | 14.4 | 177 | [ | |||||||||||
| MIL-100(Cr) | 1900 | 1.1 | 3.5 | 11.7 | 144 | 19 | [ | |||||||||||
| Zeolites | CaX | 0.36 | 3.2 | 298 | 7.5 | 24.68 | [ | |||||||||||
| Porous | CMK-3 | 950 | 0.87 | 3.5 | 298 | 7.5 | 19.78 | [ | ||||||||||
| carbon | K-PAF-1-750 | 2926 | 3.5 | 298 | 17.1 | 17.9 | [ | |||||||||||
| material | Maxsorb A | 3100 | 4.0 | 298 | 16.4 | 152 | [ | |||||||||||
| KUA31752 | 3355 | 4.0 | 298 | 15.6 | 155 | [ | ||||||||||||
| PC | 1220 | 0.47 | 2.0 | 298 | 10.2 | 141 | [ | |||||||||||
| Saran A carbon 36X | 1650 | 3.2 | 298 | 11.5 | [ | |||||||||||||
| KF-1500 | 1500 | 3.2 | 298 | 7.6 | [ | |||||||||||||
| UlaC-950-HF | 1476 | 0.61 | 3.5 | 298 | 17 | 232 | 22.4 | [ | ||||||||||
Table 1 Summary of methane storage capacities of some porpus materials
| Material | Surface area/ (m2·g-1) | Pore volume/ (cm3·g-1) | Condition | CH4 storage capacity (mass ratio,%) | Excess CH4 uptake/ (cm3·cm-3) | Heat of adsorption/ (kJ·mol-1) | Ref. | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| p/MPa | T/K | |||||||||||||||||
| Porous | COF-102 | 3620 | 1.55 | 3.5 | 298 | 15.8 | 120 | 8.6 | [ | |||||||||
| polymers | COF-103 | 3530 | 1.54 | 3.5 | 298 | 14.9 | 152 | 9.5 | [ | |||||||||
| HCP-1 | 1904 | 0.54 | 1.5 | 298 | 6.4 | [ | ||||||||||||
| HCP-4 | 1366 | 0.55 | 3.6 | 298 | 8.1 | [ | ||||||||||||
| PIM-1 | 850 | 0.349 | 1.5 | 293 | 3.8 | [ | ||||||||||||
| PPN-3 | 2840 | 1.7 | 3.5 | 295 | 12.2 | 15.2 | [ | |||||||||||
| PPN-4 | 6461 | 3.04 | 5.5 | 295 | 21.5 | [ | ||||||||||||
| MOFs | Cu2(sbtc)[PCN-11] | 1931 | 0.91 | 2.5 | 298 | 14.0 | 171 | 14.6 | [ | |||||||||
| Ni(dhtp)[NiMOF-74,CPO-27-Ni] | 1027 | 0.54 | 3.5 | 298 | 11.9 | 206 | 21.5—22 | [ | ||||||||||
| Zn4O(bdc)3[MOF-5] | 1870 | 3.6 | 13.5 | 110 | 12.2 | [ | ||||||||||||
| LIFM-82 | 1624 | 0.71 | 3.5 | 298 | 15.2 | 196 | 17.5 | [ | ||||||||||
| MOF-905 | 3490 | 1.34 | 3.5 | 298 | 18.8 | 145 | 11.7 | [ | ||||||||||
| HKUST-1 | 1850 | 0.78 | 3.5 | 298 | 18.4 | 227 | 17.0 | [ | ||||||||||
| NU-125 | 3120 | 1.29 | 3.5 | 298 | 16.5 | 182 | 15.1 | [ | ||||||||||
| PCN-14 | 2000 | 0.829 | 3.5 | 298 | 17.4 | 202 | 17.6 | [ | ||||||||||
| UTSA-76 | 2820 | 1.06 | 3.5 | 298 | 15.4 | 211 | 15.4 | [ | ||||||||||
| UTSA-20 | 1620 | 3.5 | 298 | 18.1 | 184 | [ | ||||||||||||
| NOTT-102 | 3342 | 1.268 | 3.5 | 298 | 16.5 | 181 | 16.0 | [ | ||||||||||
| NOTT-103 | 2958 | 1.157 | 3.5 | 300 | 15.6 | 193 | 15.9 | [ | ||||||||||
| MAF-38 | 2022 | 0.808 | 3.5 | 298 | 14.1 | 226 | 21.6 | [ | ||||||||||
| ZJU-35 | 2899 | 1.156 | 3.5 | 300 | 14.4 | 177 | [ | |||||||||||
| MIL-100(Cr) | 1900 | 1.1 | 3.5 | 11.7 | 144 | 19 | [ | |||||||||||
| Zeolites | CaX | 0.36 | 3.2 | 298 | 7.5 | 24.68 | [ | |||||||||||
| Porous | CMK-3 | 950 | 0.87 | 3.5 | 298 | 7.5 | 19.78 | [ | ||||||||||
| carbon | K-PAF-1-750 | 2926 | 3.5 | 298 | 17.1 | 17.9 | [ | |||||||||||
| material | Maxsorb A | 3100 | 4.0 | 298 | 16.4 | 152 | [ | |||||||||||
| KUA31752 | 3355 | 4.0 | 298 | 15.6 | 155 | [ | ||||||||||||
| PC | 1220 | 0.47 | 2.0 | 298 | 10.2 | 141 | [ | |||||||||||
| Saran A carbon 36X | 1650 | 3.2 | 298 | 11.5 | [ | |||||||||||||
| KF-1500 | 1500 | 3.2 | 298 | 7.6 | [ | |||||||||||||
| UlaC-950-HF | 1476 | 0.61 | 3.5 | 298 | 17 | 232 | 22.4 | [ | ||||||||||
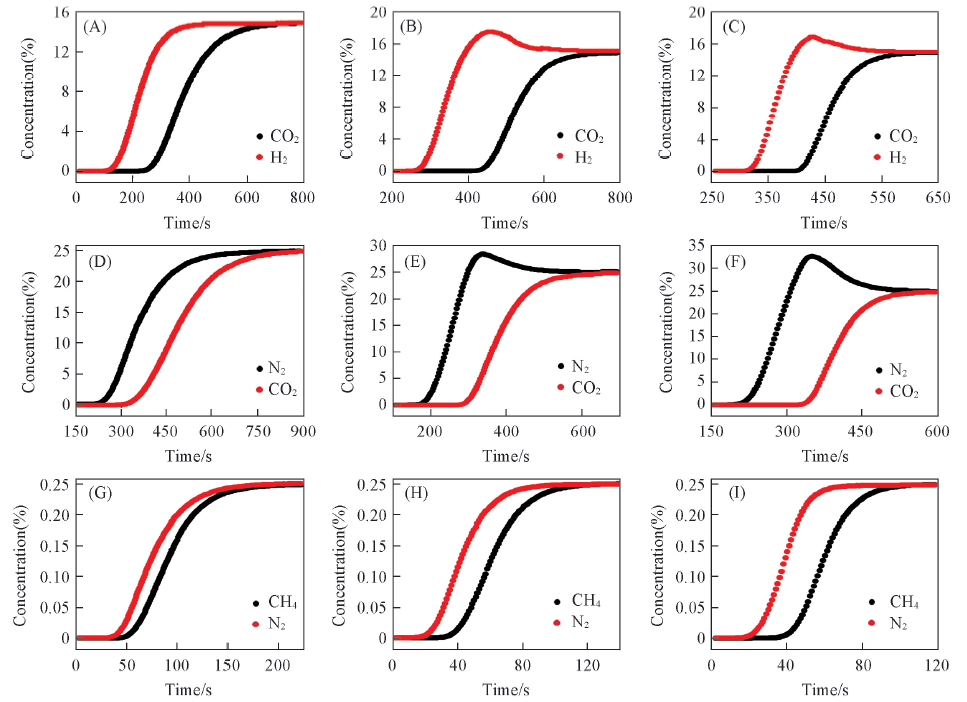
Fig.11 Breakthrough curves of LamCW-900-HF for CO2/H2/He(A—C), N2/CO2/He(D—F) and CH4/N2/He(G—I) mixture with ratio of 1:1:2 at 299 K(A), (D), (G): 100 kPa; (B), (E), (H): 200 kPa; (C), (F), (I): 300 kPa.
| [1] | |
| [2] | Piñero E.R., Leroux F., Béguin F., Adv. Mater., 2006, 14(18), 1877—1882 |
| [3] | Laine J., Calafat A., Labady M., Carbon, 1989, 27(2), 191—195 |
| [4] | Laine J., Calafat A., Carbon, 1991, 29(7), 949—953 |
| [5] | Ahmadpour A., Do D.D., Carbon, 1997, 35(12), 1723—1732 |
| [6] | Pandolfo A.G., Amoli M. A., Killingley J. S., Carbon, 1994, 32(5), 1015—1019 |
| [7] | Fuertes A.B., Ferrero G. A., Sevilla M., J. Mater. Chem.A, 2014, 2(35), 14439—14448 |
| [8] | Deng J., Xiong T.Y., Xu F., Li M. M., Han C. L., Gong Y. T., Wang H. Y., Wang Y., Green Chem., 2015, 7, 4053—4060 |
| [9] | Wang Z., Tan Y.T., Yang Y. L., Zhao X. N., Liu Y., Niu L. Y., Tichnell B., Kong L. B., Kang L., Liu Z., Ran F., J. Power Sources, 2018, 378, 499—510 |
| [10] | Laine J., Simoni S., Calles R., Chem. Eng. Commun., 1991, 99(1), 15—23 |
| [11] | Bergius F., Z. Angew. Phys. Chem., 1913, 20, 260—260 |
| [12] | Sevilla M., Antonio J., Antonio M.A., Fuertes B., Biomass Bioenerg., 2011, 35(7), 3152—3159 |
| [13] | Biswal M., Banerjee A., Deo M., Ogale S., Energ. Environ. Sci., 2013, 6(4), 1249—1259 |
| [14] | Lu W.J., Huang S. Z., Miao L., Liu M. X., Zhu D. Z., Li L. C., Duan H., Gan Z. H., Chinese Chem. Lett., 2017, 28(6), 1324—1329 |
| [15] | Zhong Y., Xia X.H., Deng S. J., Zhan J. Y., Fang R. Y., Xia Y., Wang X. L., Zhang Q., Tu J. P., Adv. Energy Mater., 2018, 8(1), 1701110 |
| [16] | Caturla F., Molina-Sabio M., Rodríguez-Reinoso F., Carbon, 1991, 29(7), 999—1007 |
| [17] | Gong Y., Li D., Luo C., Fu Q., Pan C., Green Chem., 2017, 19(17), 4132—4140 |
| [18] | Paskaleva E.E., Lin X. D., Duus K., Mc Sharry J. J., Veille J. C. L., Thornber C., Liu Y. Z., Lee D. Y. W., Canki M., Virol. J., 2008, 5(1), 8 |
| [19] | Zhu Y., Murali S., Stoller M.D., Ganesh K. J., Cai W., Ferreira P. J., Pirkle A., Wallace R. M., Cychosz K. A., Thommes M., Su D., Stach E. A., Ruoff R. S., Science, 2011, 332(6037), 1537—1541 |
| [20] | Konstas K., Osl T., Yang Y.X., Batten M., Burke N., Hill A. J., Hill M. R., J. Mater. Chem., 2012, 22(33), 16698—16708 |
| [21] | Wood C.D., Tan B., Trewin A., Su F. B., Rosseinsky M. J., Bradshaw D., Sun Y., Zhou L., Cooper A. I., Adv. Mater., 2008, 20(10), 1916—1921 |
| [22] | Larsen G.S., Lin P., Siperstein F. R., Colina C. M., Adsorption, 2011, 17(1), 21—26 |
| [23] | Makal T.A., Li J. R., Lu W. G., Zhou H. C., Chem. Soc. Rev., 2012, 41(23), 7761—7779 |
| [24] | Yuan D.Q., Lu W. G., Zhao D., Zhou H. C., Adv. Mater., 2011, 23(32), 3723—3725 |
| [25] | Zhou W., Chem. Rec., 2010, 10(3), 200—204 |
| [26] | Chen C.X., Wei Z. W., Jiang J. J., Zheng S. P., Wang H. P., Qiu Q. F., Cao C. C., Fenske D., Su C. Y., J. Am. Chem. Soc., 2017, 139(17), 6034—6037 |
| [27] | Jiang J., Furukawa H., Zhang Y.B., Yaghi O. M., J. A.Chem. Soc., 2016, 138(32), 10244—10251 |
| [28] | Peng Y., Krungleviciute V., Eryazici I., Hupp J.T., Farha O. K., Yildirim T., J. Am. Chem. Soc., 2013, 135(32), 11887—11894 |
| [29] | Wilmer C.E., Farha O. K., Yildirim T., Eryazici I., Krungleviciute V., Sarjeant A. A., Snurr R. Q., Hupp J. T., Energ. Environ. Sci., 2013, 6(4), 1158—1163 |
| [30] | Li B., Wen H.M., Wang H., Wu H., Yildirim T., Zhou W., Chen B. L., Energ. Environ. Sci., 2015, 8(8), 2504—2511 |
| [31] | Li B., Wen H.M., Wang H., Wu H., Tyagi M., Yildirim T., Zhou W., Chen B., J. Am. Chem. Soc., 2014, 136(17), 6207—6210 |
| [32] | Guo Z.Y., Wu H., Srinivas G., Zhou Y. M., Xiang S. C., Chen Z. X., Yang Y. T., Zhou W., O’Keeffe M., Chen B. L., Angew. Chem. Int. Ed., 2011, 50(14), 3178—3181 |
| [33] | He Y., Zhou W., Yildirim T., Chen B., Energ. Environ. Sci., 2013, 6(9), 2735—2744 |
| [34] | Lin J.M., He C. T., Liu Y., Liao P. Q., Zhou D. D., Zhang J. P., Chen X. M., Angew. Chem. Int. Ed., 2016, 55(15), 4674—4678 |
| [35] | Kong G.Q., Han Z. D., He Y. B., Ou S., Zhou W., Yildirim T., Krishna R., Zou C., Chen B. L., Wu C. D., Chem-Eur. J., 2013, 19(44), 14886—14894 |
| [36] | Llewellyn P.L., Bourrelly S., Serre C., Vimont A., Daturi M., Hamon L., de Weireld G., Chang J. S., Hong D. Y., Hwang Y. K., Jhung S. H., Ferey G., Langmuir, 2008, 24(14), 7245—7250 |
| [37] | Zhang S.Y., Talu O., Hayhurst D. T., J. Chem. Phys., 1991, 95(4), 1722—1726 |
| [38] | Zhou H.S., Zhu S. M., Honma I., Seki K., Chem. Phys. Lett., 2004, 396(4), 252—255 |
| [39] | Li Y.Q., Ben T., Zhang B. Y., Fu Y., Qiu S. L., Sci. Rep-UK, 2013, 3(33), 2420 |
| [40] | Castelló D.L., Amorós D. C., Solano A. L., Energ.Fuel, 2002, 16(5), 1321—1328 |
| [41] | Menon V.C., Komarneni S., J. Porous Mater., 1998, 5(1), 43—58 |
| [42] | Wang Y., Ben T., Qiu S.L., Chem. J. Chinese Universities, 2016, 37(5), 801—809) |
| (王昀, 贲腾, 裘式纶. 高等学校化学学报, 2016, 37(5), 801—809) | |
| [43] | Lu J.L., Plant Nutrition, China Agricultural University Press, Beijing, 2003, 61—72 |
| (陆景陵. 植物营养学, 北京: 中国农业大学出版社, 2003, 61—72) | |
| [44] | Mendoza-Cortés J.L., Han S. S., Furukawa H., Yaghi O. M., Goddard III W. A., J. Phys. Chem.A, 2010, 114(40), 10824—10833 |
| [45] | Bojan M.J., Slooten R. V., Steele W., Sep. Sci. Technol., 1992, 27(14), 1837—1856 |
| [46] | Chen X.S., Mcenaney B., Mays T. J., Alcaniz-Monge J., Cazorla-Amoros D., Linares-Solano A., Carbon, 1997, 35(9), 1251—1258 |
| [47] | Lv Y.K., Gan L. H., Liu M. X., Xiong W., Xu Z. J., Zhu D. Z., Wright D. S., J. Power Sources, 2012, 209(209), 152—157 |
| [48] | Bhatia S.K., Myers A. L., Langmuir, 2006, 22(4), 1688—1700 |
| [1] | WANG Yun,BEN Teng. Preparation and Electrochemical Performance of Seaweed-based Heteroatom-containing Carbon Materials† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2627. |
| [2] | WANG Yun, BEN Teng, QIU Shilun. Self-activated Carex Meyeriana Kunth-based Porous Carbon Prepared by Direct Carbonization and Its Electrochemical Properties† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1042. |
| [3] | WANG Yun, BEN Teng, QIU Shilun. Performance of Gas Storage in Carex meyeriana Kunth-based Carbon† [J]. Chem. J. Chinese Universities, 2016, 37(5): 801. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
